Anti-Obesity Therapeutic General Toxicity Study Service
InquiryAs the global prevalence of metabolic disorders reaches unprecedented levels, the development of anti-obesity therapeutics has shifted from simple calorie-restriction mimetics to complex hormonal analogs, such as GLP-1, GIP, and glucagon receptor agonists. However, the rapid advancement of these modalities brings significant safety concerns, including gastrointestinal distress, thyroid C-cell hyperplasia risks, and potential cardiovascular complications.
Toxicity Testing Service for Anti-Obesity Therapies
Protheragen provides a specialized anti-obesity therapeutic general toxicity study service designed to de-risk your drug candidates during the preclinical phase. We offer a comprehensive suite of in vivo and in vitro assessments to evaluate the safety profile of novel weight-loss agents, ensuring that dose-limiting toxicities are identified long before clinical entry. Our focus remains on providing high-resolution data regarding repeated-dose toxicity, metabolic stability, and organ-specific pathology to streamline your path to regulatory submission.
Core Technologies
Protheragen utilizes a multi-dimensional technological platform to assess the safety of anti-obesity agents. We integrate high-throughput metabolic cages with advanced histopathological imaging to monitor the physiological impact of long-term administration.
- Continuous Metabolic Monitoring Systems
Real-time tracking of oxygen consumption (VO2), CO2 production (VCO2), and respiratory exchange ratio (RER) to detect metabolic shifts that might indicate systemic toxicity.
- Multiplex Biomarker Profiling
Evaluation of circulating levels of leptin, insulin, ghrelin, and inflammatory cytokines (IL-6, TNF-α) to observe the endocrine disruption potential of the therapeutic.
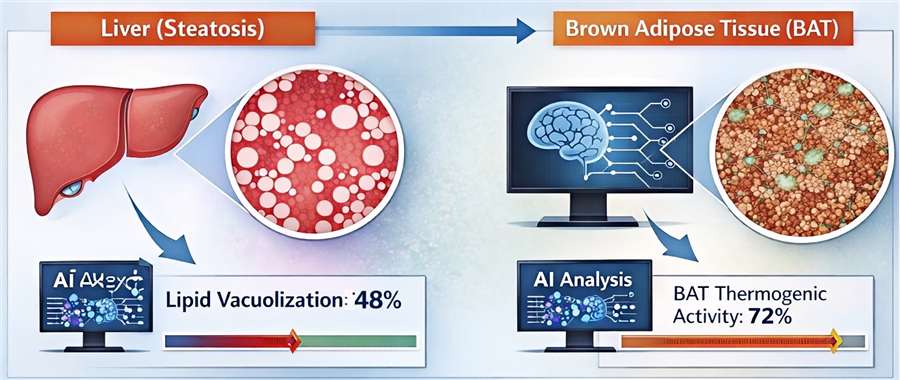
- Quantitative Digital Pathology
Utilizing AI-driven imaging to quantify lipid vacuolization in the liver (steatosis) and brown adipose tissue (BAT) thermogenic activity, providing objective data on tissue-specific toxicity.
- Cardiovascular Telemetry Integration
Monitoring heart rate and blood pressure in conscious models to address the known sympathetic activation risks associated with certain anti-obesity medications.
Solution Scope
The Protheragen toxicity platform covers a broad spectrum of preclinical safety evaluations tailored for the metabolic market:
Initial dose-ranging studies to evaluate the immediate toxicity of a single therapeutic dose.
Assess the toxicity associated with repeated administration over a set period.
Focused on identifying immediate toxic effects following short-term administration.
Evaluates the impact of therapeutic agents when administered over weeks to determine medium-term safety.
Conducted over longer periods, this study identifies any emerging toxic effects as the therapeutic is used over months.
Long-term studies assessing the safety and potential adverse effects when anti-obesity therapeutics are used over extended periods.
A study that helps define the safe therapeutic dose range, including determining maximum tolerable dose (MTD) and no-observed-adverse-effect-level (NOAEL).
Connect with a Protheragen Expert to Discuss Your Research Goals and Technical Strategy.
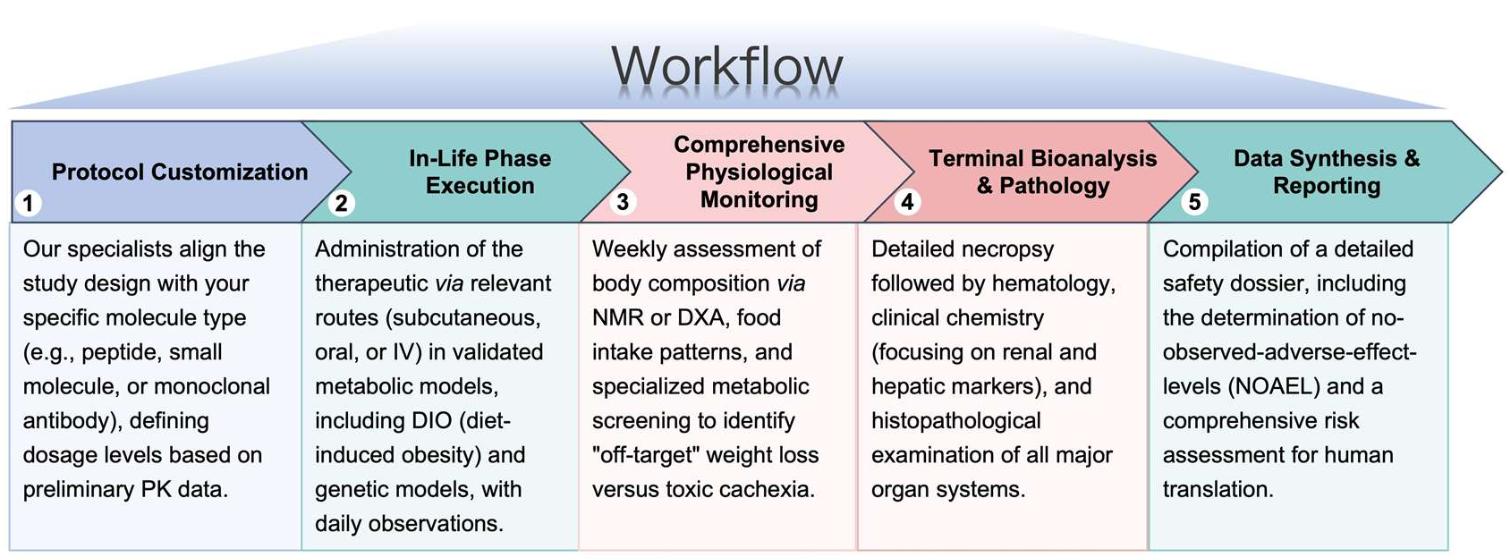
Workflow
Our service is structured to provide rigorous oversight from initial protocol design to the final safety report, ensuring every preclinical milestone is met with precision.
Fields of Application
Our toxicity studies are essential for a variety of therapeutic approaches within the weight management space.
- Incretin Mimetics: Safety profiling for GLP-1, GIP, and glucagon receptor multi-agonists.
- Centrally Acting Agents: Evaluating the neurotoxicity and behavioral safety of appetite suppressants.
- Nutrient Absorption Inhibitors: Assessing the gastrointestinal integrity and long-term malabsorption risks.
- Thermogenic Activators: Monitoring cardiovascular strain and hyperthermia risks associated with mitochondrial uncouplers or BAT activators.
Advantages
Choosing Protheragen ensures your drug candidate undergoes the most rigorous metabolic safety screening available in the preclinical sector.
Metabolic-Specific Expertise
Unlike general CROs, we focus specifically on the nuances of metabolic disease, understanding the difference between therapeutic efficacy and toxicological weight loss.
Advanced Model Selection and Rapid Turnaround
We utilize specialized models that better mimic the human obese phenotype, providing higher predictive value for clinical safety. Our optimized workflow allows for the delivery of preliminary safety data weeks faster than industry standards.
Quality Standards and Scientific Integrity
Our data packages are developed to satisfy the most rigorous international benchmarks for preclinical excellence. We ensure that every report provides the high-fidelity evidence and detailed documentation necessary for clear, confident decision-making during the drug development process.
Inquire Now to Speak with a Protheragen Specialist About Your Project Strategy.
Customer Review
Distinguishing Therapeutic Efficacy from Systemic Toxicity with Precision
"The team at Protheragen provided us with critical safety data that allowed us to pivot our lead optimization strategy early. Their ability to distinguish between target-related GI effects and systemic toxicity was pivotal for our last round of funding. We plan to utilize their full toxicity suite for our upcoming IND-enabling studies."
Director of Pharmacology, Biopharma Startup
Seamless Collaboration and High-Resolution Metabolic Insights
"Working with Protheragen has been a seamless experience. Their expertise in metabolic cages and continuous monitoring gave us a level of detail we couldn't find at larger, more generalized CROs. They are our primary partner for all metabolic safety assessments."
Senior Scientist, Global Pharmaceutical Firm
Frequently Asked Questions
-
How do you differentiate between weight loss from efficacy and weight loss from toxicity?
We utilize pair-feeding studies and metabolic cage data to distinguish between reduced caloric intake and systemic malaise or metabolic dysfunction.
-
Can you accommodate biologics as well as small molecules?
Yes, Protheragen has dedicated platforms for both, including specialized immunogenicity testing for peptides and proteins.
-
Which animal models do you typically use?
We primarily utilize diet-induced obesity (DIO) rodents, but we can customize the model based on the mechanism of action of your drug.
-
Do your studies include cardiovascular monitoring?
Absolutely. Given the history of anti-obesity drugs, we offer integrated telemetry to monitor for tachycardia or hypertension.
-
What is the typical duration of a general toxicity study?
We offer a range from 7-day pilot studies to 6-month chronic toxicity assessments.
-
How do you assess liver safety in the context of obesity?
We use specialized histopathology to differentiate between pre-existing NAFLD/NASH and drug-induced liver injury (DILI).
-
Can we add specific biomarkers to the study?
Yes, our multiplex platform is highly flexible; we can include specific hormonal or inflammatory markers upon request.
-
How do I start a project with Protheragen?
Simply reach out via our contact portal; we can provide a preliminary study design and quote within 48 hours.
How to Contact Us
Protheragen is committed to advancing the next generation of safe and effective anti-obesity therapeutics. Our specialized expertise in metabolic toxicity ensures that your drug candidate is tested with the highest scientific rigor.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

