Anti-Obesity Therapeutic Sub-chronic Toxicity Study Service
InquiryThe development of anti-obesity therapeutics is currently at the forefront of metabolic research, driven by the global obesity epidemic and the success of incretin-based therapies. However, because these drugs are often intended for long-term administration, identifying potential safety risks during the preclinical phase is paramount.
Sub-Chronic Toxicity Evaluation for Anti-Obesity Therapies
Protheragen offers a dedicated anti-obesity therapeutic sub-chronic toxicity study service, meticulously designed to evaluate the safety of repeated dosing over durations of 28 to 90 days. Our studies are engineered to identify target organ toxicity, establish no-observed-adverse-effect levels (NOAEL), and characterize the metabolic impact of your compound in both healthy and diet-induced obesity (DIO) models. By maintaining a strict focus on preclinical excellence, we provide the comprehensive toxicological data required to fully validate the safety and metabolic integrity of your therapeutic candidates.
Core Technologies
Our platform integrates traditional toxicology with advanced metabolic phenotyping to provide a multi-dimensional view of drug safety.
- Precision Metabolic Cage Systems
Automated monitoring of energy expenditure (VO2/VCO2), respiratory exchange ratio (RER), and fine-grained feeding patterns to detect subtle metabolic disturbances.
- High-Resolution Digital Pathology
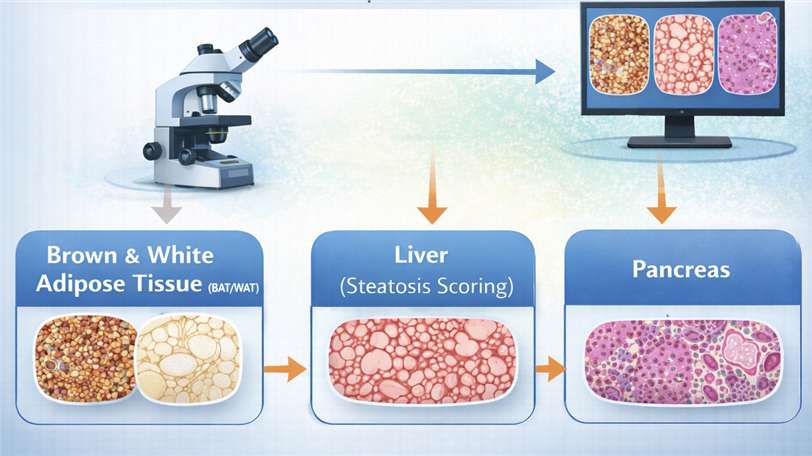
Quantitative histopathology of metabolic tissues, including brown and white adipose tissue (BAT/WAT), liver (steatosis scoring), and pancreas.
- Multi-Platform Biomarker Analysis
High-sensitivity detection of incretins (GLP-1, GIP), adipokines (leptin, adiponectin), and inflammatory markers using multiplexed ELISA and MSD platforms.
- Body Composition Profiling
Longitudinal tracking of lean vs. fat mass using non-invasive EchoMRI™ or dual-energy X-ray absorptiometry (DXA) to differentiate weight loss from muscle wasting or toxicity-induced cachexia.
Solution Scope
Protheragen provides a versatile range of sub-chronic toxicity assessment options tailored to various drug modalities:
- Standard Rodent Sub-chronic Studies
28-day and 90-day repeated dose studies in rats or mice.
Evaluation of candidates in DIO Models to identify safety profiles in the intended disease state.
- Combination Therapy Safety Assessment
Specialized protocols to evaluate the toxicity of co-administered agents (e.g., GLP-1 agonists combined with SGLT2 inhibitors).
Simultaneous blood sampling for PK/TK modeling to correlate systemic exposure with observed toxicological findings.
- Immunotoxicity Screening
Assessment of cytokine release and immune cell population changes, particularly for peptide and biologic-based anti-obesity agents.
Workflow
Our workflow is designed for transparency and scientific rigor, ensuring that every study meets the highest regulatory standards:

Fields of Application
Our specialized sub-chronic toxicity study services are designed to support a diverse array of therapeutic modalities, ensuring that every promising anti-obesity intervention is backed by rigorous safety data tailored to its specific biological mechanisms.
- Small Molecule Discovery: Evaluation of novel CNS-acting anorectics or peripheral metabolic modulators.
- Peptide and Biologic Development: Safety assessment of long-acting GLP-1, GIP, and Glucagon Receptor Agonists.
- Nutraceutical and Functional Food Testing: Screening of botanical extracts or bioactive compounds for sub-chronic safety before market entry.
- RNA Therapeutics: Specialized toxicity and biodistribution studies for ASO or siRNA-based metabolic interventions.
Advantages
Choosing Protheragen means partnering with a leader in metabolic preclinical research. Our advantages include:
Disease-Relevant Background Data
We maintain extensive "published data" and internal historical databases on DIO models, allowing us to distinguish between drug-induced toxicity and background metabolic fluctuations.
Metabolic Specialization
Unlike generalist CROs, our pathologists and toxicologists specialize in metabolic disorders, ensuring that subtle changes in insulin sensitivity or adipocyte morphology are correctly interpreted.
Methodological Precision, Superior Surgical and Dosing Capability
We offer specialized expertise in complex dosing regimens, including continuous subcutaneous infusion via osmotic pumps for peptide therapeutics. Our study protocols meet the highest global benchmarks for scientific integrity, delivering the rigor and consistency essential for high-stakes drug development decisions.
Inquire Now to Speak with Our Toxicologists.
Customer Review
Precision in Differentiating Efficacy from Adverse Effects
"Working with Protheragen was a turning point for our lead peptide program. Their expertise in DIO models allowed us to clearly differentiate our compound's weight-loss efficacy from potential off-target gastrointestinal effects. The level of detail in their histopathology reports was exceptional, providing us with the confidence to move into IND-enabling studies."
Senior Director of Pharmacology, Mid-sized Biotech
Proactive Scientific Partnership and Adaptive Problem-Solving
"What impressed us most was the Protheragen team's proactive communication. When our compound showed unexpected metabolic changes in the pilot phase, their toxicologists immediately suggested a modified biomarker panel that helped us identify a specific compensatory mechanism rather than a toxic effect. We look forward to our next collaboration on our dual-agonist candidate."
Principal Scientist, Metabolic Research Institute
Frequently Asked Questions
-
Why is a 90-day study preferred over a 28-day study for anti-obesity drugs?
While a 28-day study provides initial safety signals, a 90-day study is often required to observe cumulative effects on organ systems. It allows for a more comprehensive assessment of metabolic adaptation and potential chronic toxicity.
-
Can you perform these studies in high-fat diet (HFD) fed animals?
Yes. In fact, evaluating toxicity in the intended disease model (obese animals) is often more predictive than testing in lean animals, as the underlying metabolic stress can alter drug metabolism and organ sensitivity.
-
How do you differentiate between weight loss due to efficacy and weight loss due to toxicity?
We use EchoMRI to monitor body composition. Efficacy-driven weight loss typically targets fat mass while preserving lean mass, whereas toxicity-induced weight loss (cachexia) often involves significant loss of lean muscle and organ weight.
-
What clinical chemistry parameters are included in your standard panel?
Our panel includes standard markers (ALT, AST, BUN, Cr) plus metabolic-specific markers such as fasting glucose, HbA1c, total cholesterol, HDL/LDL, and triglycerides.
-
What is the typical turnaround time for a 90-day study report?
Including the in-life phase, histopathology, and data analysis, a final audited report is typically delivered within 4–6 months from the start of the in-life phase.
-
Can we include "satellite groups" in the study design?
Absolutely. We highly recommend satellite groups to observe the persistence or reversal of toxic effects after a 14- or 28-day recovery period.
-
How do you handle the high viscosity of some long-acting peptide formulations?
Our technical team is experienced in handling diverse formulations and uses specialized needles and dosing techniques to ensure accurate delivery of viscous or suspension-based therapeutics.
-
Is the histopathology of the brain included?
Yes, given that many anti-obesity drugs target the CNS, we perform detailed histopathological exams of the hypothalamus and other relevant brain regions.
-
How can I get a quote for my specific compound?
Simply click our inquiry link. We will set up a technical consultation to understand your compound's modality and dosing requirements to provide a tailored proposal.
How to Contact Us
Protheragen provides an industry-leading preclinical platform for the comprehensive safety evaluation of anti-obesity therapeutics. By integrating rigorous sub-chronic toxicity assessments with advanced metabolic monitoring and specialized pathology, we deliver the high-quality data necessary to fully understand the biological impact and safety profile of your metabolic drug candidates.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

