Anti-Obesity Therapeutic Chronic Toxicity Study Service
InquiryAs the global landscape of metabolic disease management shifts toward long-term pharmacological interventions, the demand for rigorous safety assessment of anti-obesity therapeutics has never been higher. At Protheragen, we provide a specialized anti-obesity therapeutic chronic toxicity study service focused on uncovering the long-term biological impact of sustained weight-loss interventions. Our expertise lies in characterizing how metabolic candidates interact with complex physiological systems over extended durations, ensuring a profound understanding of a compound's safety profile and systemic tolerability well before it advances to the next stage of development.
Long-Term Toxicity Assessment for Anti-Obesity Therapies
With over 20 years of experience in metabolic research, our team understands that anti-obesity agents—ranging from GLP-1 receptor agonists and dual/triple incretin mimetics to lipase inhibitors—require sophisticated monitoring of nutritional status, cardiovascular health, and endocrine balance. Our services focus exclusively on the preclinical phase, utilizing advanced animal models to characterize the safety profile of your candidate before it enters clinical development.
Core Technologies
To provide high-fidelity safety data, Protheragen integrates cutting-edge technologies that go beyond standard toxicology:
Utilizing automated monitoring systems to track real-time food intake, energy expenditure, and locomotor activity in chronic study cohorts.
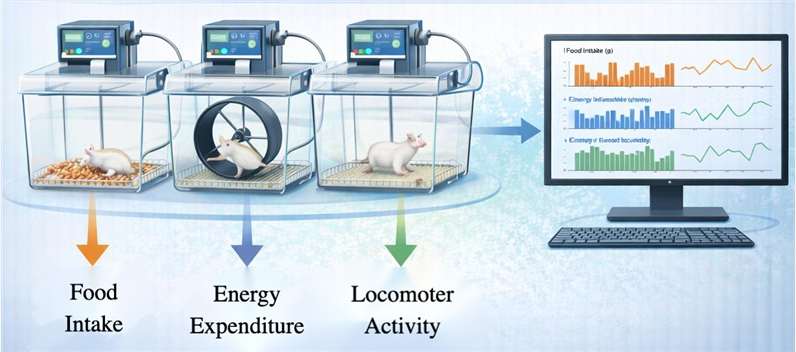
- Advanced Biomarker Analytics
Multiplexed assays for monitoring gut hormones (GLP-1, GIP, PYY), lipid profiles, and glycemic markers over extended durations.
- High-Resolution Histopathology
Specialized tissue processing for evaluating longitudinal changes in adipose tissue morphology, liver steatosis, and gastrointestinal integrity.
- Telemetry-Integrated Safety Pharmacology
Continuous monitoring of blood pressure and heart rate in non-rodent models to detect subtle cardiovascular signals associated with weight-loss agents.
Solution Scope
Our chronic toxicity service covers a broad spectrum of anti-obesity modalities:
- Incretin Mimetics
Long-term safety profiles for GLP-1, GIP, and Glucagon Receptor Agonists.
- Centrally Acting Agents
Evaluation of neurotoxicity and behavioral changes for drugs targeting satiety centers in the hypothalamus.
- Metabolic Modulators
Studies on mitochondrial uncouplers and thermogenic agents.
- Gastrointestinal Agents
Chronic assessment of lipase inhibitors and nutrient absorption blockers, focusing on GI-tract integrity and vitamin absorption.
- Peptide & Small Molecule Therapeutics
Tailored study designs for both biological and chemical entities.
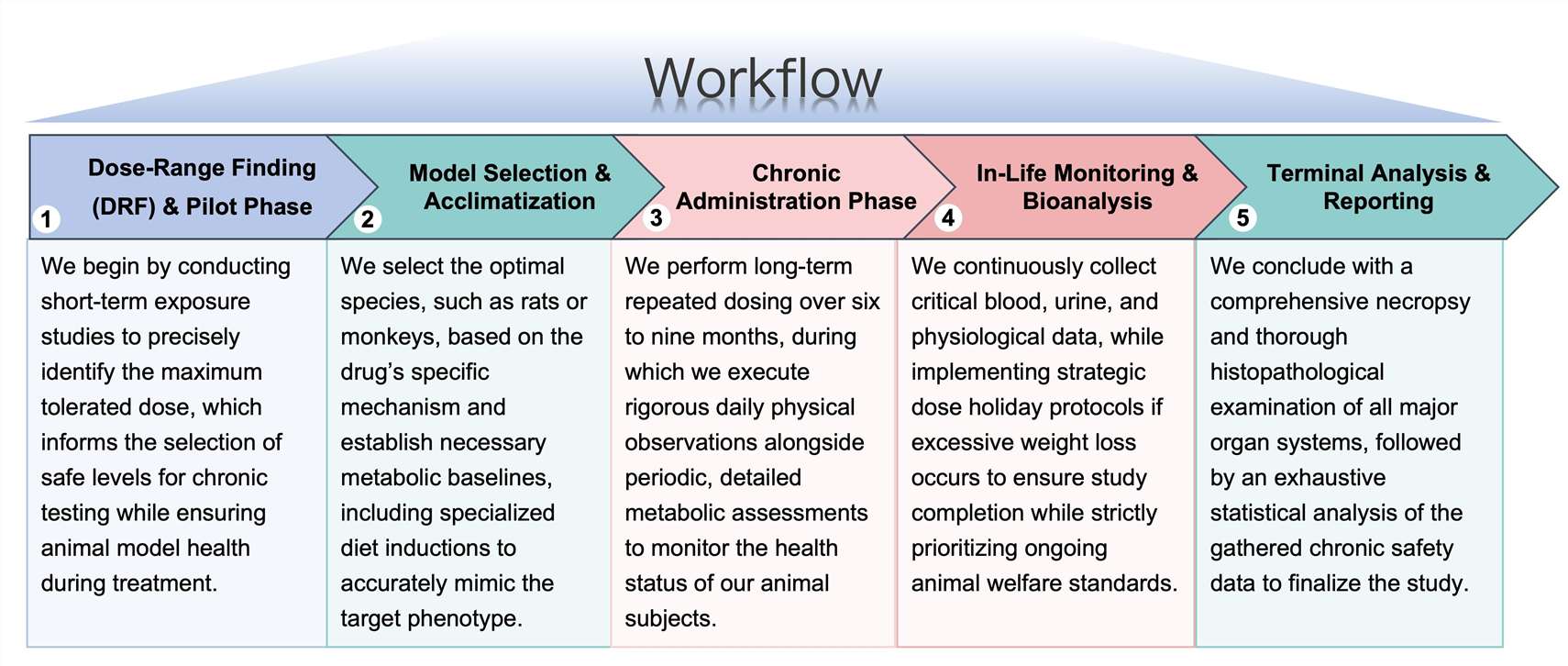
Workflow
Our service is meticulously structured to deliver profound scientific depth and biological clarity through a comprehensive, multi-stage analytical process.
Fields of Application
Our anti-obesity therapeutic chronic toxicity study service is utilized across diverse research paradigms to characterize the long-term biological impact and safety margins of innovative metabolic interventions.
- Lead Optimization: Assessing the chronic safety margin of multiple candidates to select the most viable lead for development.
- Mechanism-of-Action Safety Profiling: Determining if adverse effects are related to the primary target or off-target interactions during long-term use.
- Combination Therapy Assessment: Evaluating the chronic safety of co-administered anti-obesity agents.
Advantages
Choosing Protheragen provides your development program with unmatched technical depth:
Specialized Expertise in Weight-Loss Pharmacology
We understand that "weight loss" is a pharmacological effect that can mask toxicity. We employ specialized nutritional support and paired-feeding designs to differentiate between target-mediated weight reduction and systemic toxicity.
Advanced Methodological Rigor and Data Integrity
Our studies are conducted using the highest standards of scientific precision and standardized laboratory protocols for chronic assessment, ensuring that the published data generated is of the highest caliber and provides a definitive biological profile of your candidate's long-term behavior.
Innovative Translational Models, Integrated PK/TK Analysis
Beyond standard lean animals, we offer Diet-Induced Obesity (DIO) models for chronic toxicity to better predict how your drug will interact with the physiological state of obesity. Simultaneous Toxicokinetic (TK) Evaluation allows for the correlation of long-term systemic exposure with observed toxicological endpoints.
Contact Our Team for a Consultation and Project Quote.
Customer Review
Achieving Biological Clarity in Complex Multi-Target Programs
"Working with Protheragen was instrumental for our dual-agonist program. Their expertise in managing the nutritional challenges of chronic weight-loss studies allowed us to obtain a clean safety profile. We look forward to our next collaboration on our oral peptide candidate."
V.P. of Toxicology, Mid-sized Biotech
Expert Differentiation Between Pharmacological Effects and Systemic Toxicity
"The team at Protheragen provided more than just data; they provided biological insight. Their ability to distinguish between pharmacological weight loss and true toxicological markers saved us months of re-testing. They are our go-to partner for metabolic safety."
Senior Director of Discovery, Pharmaceutical Global Lead
Frequently Asked Questions
-
Why is a chronic study necessary for anti-obesity drugs?
Anti-obesity drugs are intended for long-term use, often for years. Chronic studies (6–9 months in animals) are required to identify toxicities that only emerge with repeated exposure, such as organ accumulation or gradual metabolic shifts.
-
Do you use obese models for chronic toxicity?
Yes, Protheragen can utilize DIO models. Testing in an obese physiological state is often more translational, as obesity alters drug metabolism and organ sensitivity.
-
How do you handle excessive weight loss in the animals?
We implement strict welfare endpoints and can utilize "dose holiday" protocols or specialized high-calorie support to ensure that weight loss doesn't become a confounding factor for toxicity.
-
What species do you typically use?
We typically use one rodent (rat) and one non-rodent (non-human primate or mini-pig) species, as recommended by regulatory guidelines for chronic assessments.
-
Can you evaluate cardiovascular safety during the chronic study?
Absolutely. We can integrate telemetry or non-invasive blood pressure monitoring to assess the long-term cardiovascular impact of your compound.
-
What is the typical duration of a chronic study?
For drugs intended for chronic human use, the preclinical study duration is generally 6 months in rodents and 9 months in non-rodents.
-
How do you monitor for neurobehavioral effects?
For centrally acting drugs, we include specialized behavioral batteries (e.g., Irwin test or functional observational battery) to monitor for CNS side effects.
-
Do you provide histopathology services?
Yes, comprehensive histopathology of all major organs is a standard component of our chronic toxicity reports.
-
How can I start a project with Protheragen?
You can reach out via our inquiry form for a confidential discussion regarding your compound's mechanism and your specific regulatory goals.
How to Contact Us
Protheragen is a leading provider of anti-obesity therapeutic sub-acute toxicity study services, offering specialized preclinical expertise to ensure your metabolic drug candidates are both effective and safe. By combining advanced metabolic monitoring with rigorous toxicological analysis, we provide the data-driven insights necessary to advance your pipeline with confidence.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

