Anti-Obesity Therapeutic Repeat Dose Toxicity Study Service
InquiryThe development of anti-obesity therapeutics is currently undergoing a paradigm shift, moving from simple appetite suppressants to complex multi-mechanistic agonists and RNA-based modalities. However, the history of weight-management drugs is fraught with safety challenges, ranging from cardiovascular complications to psychiatric adverse events and potential oncogenic risks. At Protheragen, we provide a specialized anti-obesity therapeutic repeat dose toxicity study service designed to de-risk your development pipeline through rigorous preclinical safety assessments.
Repeat-Dose Toxicity Evaluation for Anti-Obesity Therapies
Our studies are engineered to evaluate the toxicological profile of test compounds—including Small Molecules, peptides, and monoclonal antibodies—following repeated administration. By simulating chronic exposure, we identify potential target organs, characterize dose-response relationships, and assess the reversibility of adverse effects. Protheragen utilizes advanced translational models—such as diet-induced obese (DIO) rodents and non-human primates—to generate biologically relevant safety data that reflects real-world biological validity.
Core Technologies
To ensure the highest precision in safety assessment, Protheragen integrates several cutting-edge technologies into our repeat dose toxicity protocols:
- Precision Metabolic Phenotyping
Utilizing automated systems to monitor energy expenditure, respiratory exchange ratios (RER), and locomotor activity in real-time during toxicity dosing.
- High-Resolution Digital Pathology
Advanced AI-driven histological analysis to detect subtle changes in adipose tissue, liver steatosis, and target organ morphology.
- Multi-Omics Integration
Combining transcriptomics and metabolomics to identify early molecular biomarkers of toxicity before clinical signs appear.
- Telemetric Cardiovascular Monitoring
Continuous monitoring of heart rate and blood pressure in non-rodent species to address the specific cardiovascular safety concerns inherent to metabolic drugs.
Sensitive liquid chromatography-tandem mass spectrometry (LC-MS/MS) and enzyme-linked immunosorbent assay (ELISA)-based systems for precise toxicokinetic (TK) profiling of parent compounds and metabolites.
Solution Scope
Protheragen offers a comprehensive suite of repeated dose toxicity configurations tailored to the obesity therapeutic landscape:
- Sub-acute Studies (14-28 Days)
Ideal for initial safety signal detection and dose-range finding (DRF) to support preclinical research.
- Sub-chronic Studies (90 Days)
Research requirement to support mid-stage development and evaluate longer-term metabolic effects.
- Chronic Toxicity Studies (6-9 Months)
Essential for therapeutics intended for long-term weight maintenance, providing deep insights into cumulative toxicity.
- Recovery Studies
Specialized arms to evaluate the persistence or reversibility of toxicological effects after treatment cessation.
- Specialized Modality Testing
Custom protocols for GLP-1/GIP Receptor Agonists, amylin analogs, and gene-silencing (siRNA) therapies.
Explore Our Full Service Scope.
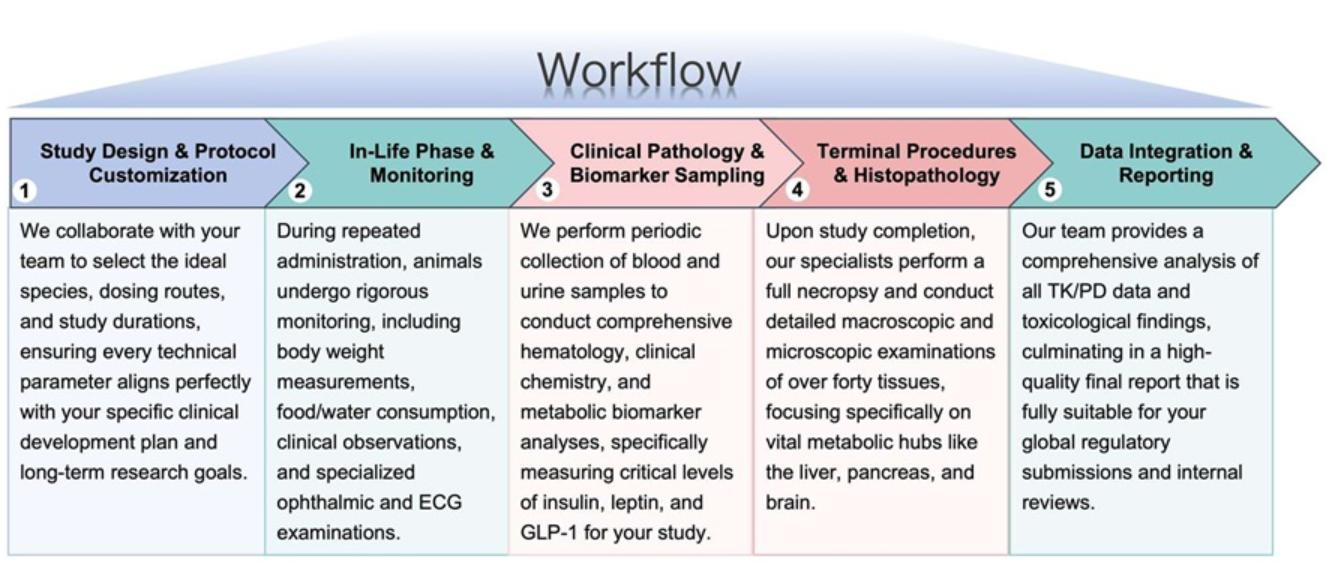
Workflow
Our service follows a structured, milestone-driven process to ensure data integrity and timely delivery.
Fields of Application
Our services are critical for the development of:
- Incretin Mimetics: Evaluating the safety of GLP-1, GIP, and glucagon receptor poly-agonists.
- Lipase Inhibitors: Assessing gastrointestinal and nutritional safety over chronic exposure.
- Centrally Acting Agents: Identifying potential neurotoxicity or behavioral side effects.
- Novel Peptides & Biologics: Characterizing immunogenicity and metabolic stability in repeated-dose settings.
- Combination Therapies: Testing the synergistic safety profiles of co-formulated metabolic drugs.
Advantages
Choosing Protheragen provides your program with distinct competitive edges.
High Standards
Our study protocols are meticulously designed to meet the most current industry standards and scientific best practices for weight-management therapies, ensuring that every project aligns with global safety expectations and modern pharmacological requirements.
Translational Model Excellence
We utilize validated DIO models that more accurately reflect the metabolic state of the target patient population compared to lean animals.
Multidisciplinary Expertise and Rapid Turnaround
Our team includes board-certified toxicologists and metabolic specialists with decades of experience in drug development. Optimized workflows and in-house bioanalytical labs significantly reduce the time from study initiation to final report delivery.
Secure Your Research Path with Industry-leading Safety Data.
Publication Data
Title: Single, 14-Day, and 13-Week Repeated Dose Toxicity Studies of Daily Oral Gelidium elegans Extract Administration to Rats
Journal: Molecules, 2018
DOI: https://doi.org/10.3390/molecules23010217
Summary: This paper evaluates the toxicity of G. elegans extract (GEE), a polyphenol-rich extract from red algae with antioxidant, anti-adipogenic, and anti-hyperglycemic effects, through single, 14-day, and 13-week repeated oral dose studies in Sprague-Dawley (CD) rats. No deaths or significant toxicological changes were observed across all studies; the median lethal dose (LD50) of GEE is >5000 mg/kg, and the no observed adverse effect level (NOAEL) is >2000 mg/kg/day. Minor observations (e.g., compound-colored stools, transient diarrhea, slight changes in some hematological/biochemical parameters) were non-toxicologically significant, as they were within normal ranges or resolved spontaneously. The study confirms GEE's safety as a dietary ingredient under experimental conditions.
Key Findings
- Acute Toxicity: A single oral dose of 5000 mg/kg GEE caused no mortality or long-term clinical signs in rats; transient compound-colored stools and diarrhea resolved within 2 days, leading to LD50 >5000 mg/kg.
- 14-Day Repeated Dose Toxicity: Doses of 500, 1000, and 2000 mg/kg/day GEE had no significant effects on body mass, food/water consumption, hematology, serum biochemistry, organ masses, or urinalysis in either sex.
- 13-Week Repeated Dose Toxicity: Consistent with short-term results, GEE at the same doses did not induce systemic toxicity; minor changes (e.g., lower water intake in males at 2000 mg/kg/day, slight hematological/electrolyte fluctuations) were within biological variation and unrelated to toxicity.
- Target Organ Safety: No pathological abnormalities were detected in vital organs (brain, liver, kidney, heart, etc.) via necropsy or histopathology.
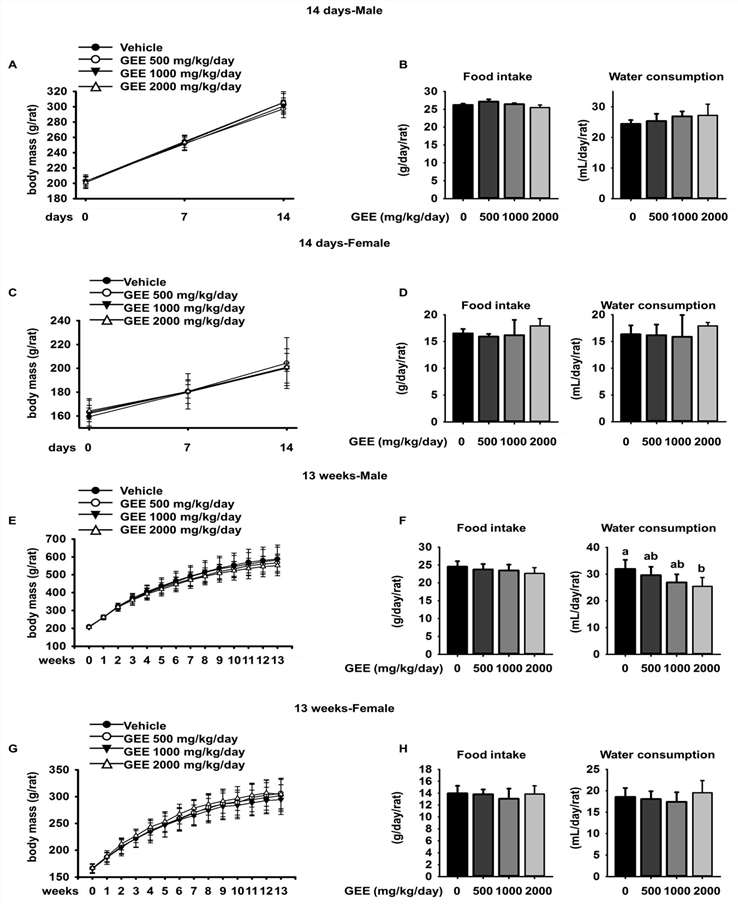 Fig.1 Effect of GEE on body mass, food intake, and water consumption of rats over 14 days and 13 weeks. (Choi, et al., 2018)
Fig.1 Effect of GEE on body mass, food intake, and water consumption of rats over 14 days and 13 weeks. (Choi, et al., 2018)
Customer Review
Strategic Dose Optimization and Safety Mapping for Novel Triple-Agonist Modalities
"The team at Protheragen was instrumental in the successful safety evaluation of our triple-agonist peptide. Their expertise in metabolic phenotyping allowed us to identify a narrow therapeutic window early on, which was critical for our dose selection."
Chief Scientific Officer, Mid-sized Biotech
Precision Toxicity Assessment for Long-Term Weight Maintenance Therapeutics
"We have partnered with Protheragen for three different anti-obesity programs. Their ability to manage long-term chronic studies with such high attention to detail in histopathology is unmatched. They don't just provide data; they provide the scientific insight necessary to move forward with confidence."
Director of Toxicology, Global Pharmaceutical Company
Frequently Asked Questions
-
What species are typically used for anti-obesity repeat dose studies?
Generally, one rodent (rat or mouse) and one non-rodent (often cynomolgus macaque or minipig) species are required to ensure pharmacological relevance.
-
Can you incorporate efficacy endpoints into a toxicity study?
Yes, we often include metabolic parameters like glucose tolerance or body composition analysis to provide a holistic view of the safety-efficacy window.
-
How do you handle the specific cardiovascular risks of anti-obesity drugs?
We employ continuous telemetry in non-rodents and specialized ECG protocols to detect any signals of QT prolongation or heart rate elevation.
-
How do you ensure the stability of the test article during a long-term study?
We conduct rigorous dose formulation analysis to ensure concentration, homogeneity, and stability throughout the dosing period.
-
Do you offer non-GLP pilot studies?
Yes, we recommend non-GLP pilot studies to optimize dose selection and refine parameters before committing to a pivotal GLP study.
-
How do you monitor for potential psychiatric side effects?
While complex, we utilize validated behavioral satiety sequences and observational batteries to detect signs of central nervous system distress.
-
Can you test RNA-based obesity therapeutics?
Yes, we have specialized platforms for the toxicological and TK assessment of siRNA and mRNA-based metabolic modifiers.
-
What sets Protheragen apart from larger CROs?
We provide a boutique experience with senior-level scientific oversight on every project, ensuring faster communication and more flexible study designs.
How to Contact Us
Protheragen provides end-to-end preclinical safety solutions for anti-obesity therapeutics. From initial dose-range finding to long-term chronic GLP studies, our expertise in metabolic diseases ensures your drug candidate is evaluated with the highest scientific standard.
Ready to advance your anti-obesity therapeutic? Contact our expert team for a detailed consultation and customized study plan.
Reference
- Choi, J.; et al. 14-Day and 13-Week Repeated Dose Toxicity Studies of Daily Oral Gelidium elegans Extract Administration to Rats. Molecules. 2018, 23(1):217. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

