Anti-Obesity Therapeutic Acute Toxicity Study Service
InquiryAs the global prevalence of obesity reaches unprecedented levels, the demand for safe and effective anti-obesity therapeutics has intensified. For pharmaceutical innovators, the transition from drug discovery to clinical evaluation is a high-stakes journey where safety remains the primary gatekeeper. Protheragen provides an industry-leading anti-obesity therapeutic acute toxicity study service, meticulously designed to characterize the safety profiles of novel compounds in the earliest stages of development.
Acute Toxicity Evaluation for Anti-Obesity Therapies
Our service focuses on identifying the toxicological threshold of potential anti-obesity agents—including small molecules, peptides, and herbal extracts—through rigorous acute and sub-acute testing protocols. By determining parameters such as the median lethal dose LD50 and the no-observed-adverse-effect level (NOAEL), we provide the critical data necessary to mitigate risk and refine dosage strategies for subsequent efficacy trials. At Protheragen, we bridge the gap between benchtop innovation and safety with high-precision preclinical assessments.
Core Technologies
To ensure the highest precision in safety assessment, Protheragen integrates several cutting-edge technologies into our repeat dose toxicity protocols:
- Targeted Metabolic Phenotyping
Utilization of state-of-the-art metabolic cages to monitor real-time energy expenditure, respiratory exchange ratios (RER), and precise food/water intake during toxicity monitoring.
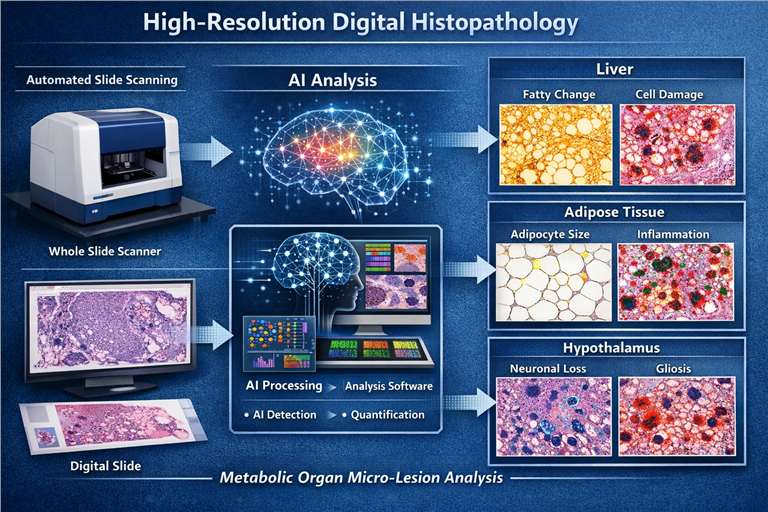
- High-Resolution Digital Histopathology
Automated slide scanning and AI-assisted tissue analysis for detecting subtle micro-lesions in metabolic organs such as the liver, adipose tissue, and hypothalamus.
- Automated Chemistry & Hematology
Integrated bioanalytical platforms for rapid profiling of lipid metabolism markers, glucose levels, and systemic inflammation indicators.
- Advanced Telemetry & Behavioral Tracking
Non-invasive monitoring of locomotor activity and physiological parameters to detect early-onset neurotoxicity or behavioral abnormalities following high-dose administration.
Solution Scope
Our expertise spans a wide range of anti-obesity modalities and study designs:
- Molecule Types
Small Molecule Inhibitors, GLP-1/GIP Receptor Agonists, polyherbal formulations, and novel gut-brain axis modulators.
- Study Durations
Single-dose acute studies (14-day observation) and repeat-dose sub-acute studies (28-day duration).
Primarily Sprague Dawley rats and C57BL/6 mice, with options for high-fat diet (HFD) induced obese models to assess safety in a disease-relevant phenotype.
- Routes of Administration
Specialized capabilities in oral gavage, subcutaneous injection, and intravenous infusion.
Explore Our Full Service Scope.
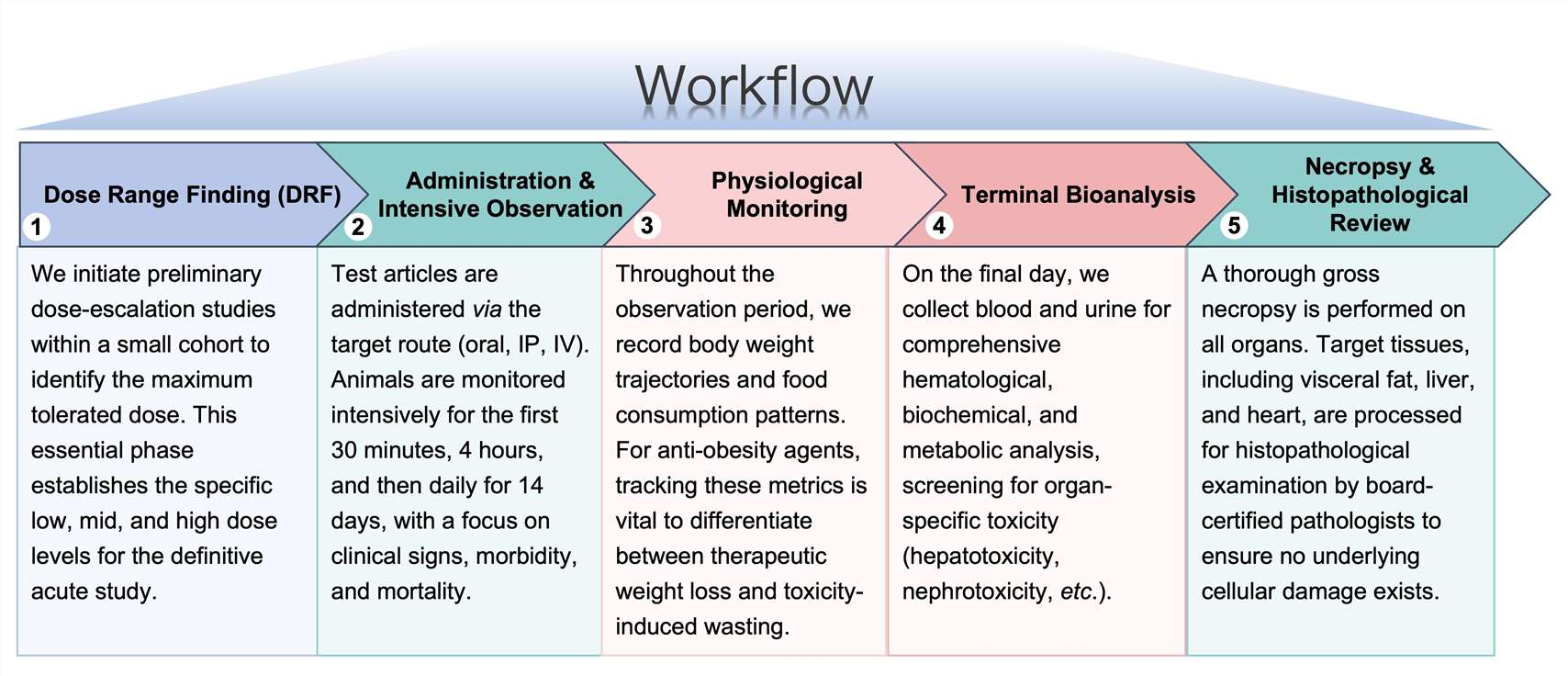
Workflow
Protheragen follows a rigorous, multi-stage workflow to ensure every acute toxicity study meets the highest scientific standards.
Fields of Application
The specialized toxicological insights provided by Protheragen serve as a critical catalyst across diverse sectors of metabolic research, enabling innovators to transition with confidence from initial discovery to advanced preclinical development.
- Early-Stage Drug Discovery: Rapidly eliminate candidates with unfavorable toxicity profiles before moving into expensive efficacy models.
- Natural Product Development: Safety validation for herbal and dietary supplements intended for weight management.
- Mechanism of Action Studies: Identifying target organs for potential toxicity to guide further chemical optimization.
- Preparation for enabling: Preparing essential safety data to support initial discussions and determine the appropriate dose selection.
Advantages
Protheragen stands at the forefront of preclinical safety assessment, offering unique benefits to our partners:
Disease-Centric Expertise
Unlike generic CROs, our toxicologists specialize in metabolic health, allowing them to distinguish between pharmacological weight reduction and adverse toxic effects.
Data Integrity & Scientific Rigor
We execute all study protocols according to the highest global scientific benchmarks, ensuring that every report delivers "published data" quality insights to empower your internal strategic decisions and candidate selection.
Tailored Study Design and Accelerated Timelines
We recognize that every compound is unique. We provide customized monitoring of specific biomarkers relevant to your drug's mechanism of action. Our optimized workflow allows for a 12-week turnaround from animal arrival to the delivery of an audited draft report.
Contact Protheragen Today to Consult with Our Toxicologists.
Publication Data
Title: Acute and sub-acute toxicity study of anti-obesity herbal granules in Sprague Dawley rats
Journal: Brazilian Journal of Biology, 2024
DOI: https://doi.org/10.1590/1519-6984.264320
Summary: This study evaluated the acute and 28-day sub-acute oral toxicity of anti-obesity polyherbal granules (PHG)—formulated with extracts of Camellia sinensis, Achyranthes aspera, Salacia reticulata, Phaseolus vulgaris, and Vitis vinifera—in Sprague Dawley rats following OECD guidelines 425 and 407, respectively. For acute toxicity, a single 2 g/kg dose of PHG caused no mortality, behavioral changes, or toxic signs over 14 days, confirming an LD50 greater than 2 g/kg. In the sub-acute study, rats received 0.3, 0.5, or 1 g/kg PHG daily for 28 days; no treatment-related mortality occurred, and hematological, biochemical, urine, necropsy, and histopathological analyses showed no significant toxic effects. Notably, PHG induced dose-dependent weight loss in both male and female rats (14–28 days) and reduced triglyceride levels without altering key organ function or structure. The no-observed-adverse-effect level (NOAEL) for PHG was determined to be 0.5 g/kg/day, demonstrating its favorable safety profile in animal models and supporting its potential as a clinical agent for obesity management.
Key Findings
- Acute Toxicity Safety: A single oral dose of 2 g/kg PHG showed no mortality, behavioral abnormalities, or toxic manifestations in rats over 14 days, with an LD50 exceeding 2 g/kg.
- Sub-Acute Toxicity Profile: Daily administration of PHG (0.3–1 g/kg) for 28 days caused no treatment-related mortality; hematological, hepatic, renal, and electrolyte parameters remained within normal ranges.
- Anti-Obesity Efficacy: PHG induced significant dose-dependent weight loss in male and female rats (vs. control) between 14–28 days, with no adverse impact on food consumption.
- Lipid Regulation: 0.5 g/kg and 1 g/kg PHG doses produced a significant dose-dependent reduction in serum triglyceride levels, supporting its lipid-modulating properties.
- NOAEL Determination: The no-observed-adverse-effect level for PHG was established at 0.5 g/kg/day, confirming its safety for repeated oral administration in rats.
- Organ and Tissue Safety: Necropsy and histopathological examinations revealed no structural alterations or toxic lesions in vital organs (liver, kidney, heart, lungs, etc.) across all PHG dose groups.
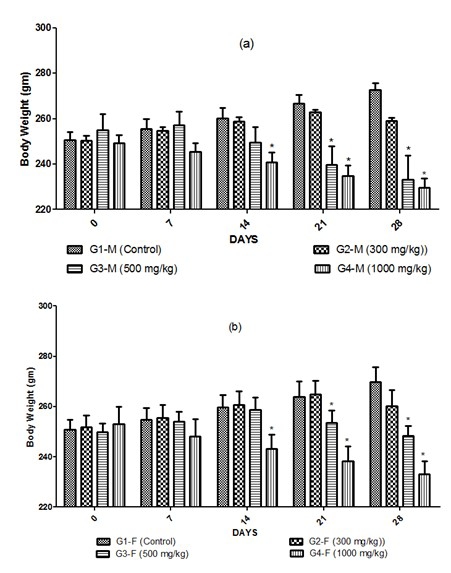 Fig.1 Initial and weekly mean body weight of (a) male rats and (b) female rats in the sub-acute dose study. (Patel, et al., 2024)
Fig.1 Initial and weekly mean body weight of (a) male rats and (b) female rats in the sub-acute dose study. (Patel, et al., 2024)
Customer Review
Precision in Metabolic Data Interpretation
"Working with Protheragen was a turning point for our lead peptide program. Their team didn't just provide raw data; they helped us interpret the subtle changes in liver enzymes that other labs might have overlooked. Their specialized knowledge in metabolic disease made the difference in our dose-selection strategy."
Senior Director of Discovery, Biotech Firm
Rapid Turnaround for Critical Project Milestones
"The efficiency of the Protheragen team is unmatched. We needed a rapid 28-day sub-acute study to meet an internal milestone, and they delivered a comprehensive report that exceeded our expectations. We look forward to our next collaboration on our upcoming GLP-1 agonist series."
Head of Pharmacology, Pharmaceutical Innovator
Frequently Asked Questions
-
Why is acute toxicity testing necessary for anti-obesity drugs?
It is the primary safety screen required to determine the toxic dose limits and identify target organ systems that may be sensitive to the compound.
-
How do you distinguish between weight loss from the drug and weight loss from toxicity?
We utilize pair-feeding controls and detailed behavioral monitoring to ensure weight loss is linked to metabolic modulation rather than malaise or illness.
-
Which animal species do you typically use?
We primarily use Sprague Dawley rats for acute studies due to their well-documented metabolic profiles, but we also offer mouse models.
-
Can you perform studies on natural extracts?
Yes, we have extensive experience in testing polyherbal granules and botanical fractions for safety.
-
Do you provide histopathology services?
Yes, complete histopathological analysis of all major organs is a standard component of our service.
-
How long does a sub-acute (28-day) study take?
Including acclimatization, dosing, and reporting, the entire process typically spans 14-16 weeks.
-
Can we monitor specific metabolic markers like insulin or leptin?
Absolutely. We can customize the clinical chemistry panel to include any relevant metabolic biomarkers.
-
How do I start a project with Protheragen?
Simply reach out via our inquiry form to schedule a technical consultation with our study directors.
How to Contact Us
Protheragen is dedicated to providing the precision, expertise, and reliability required for successful preclinical safety assessments. Our anti-obesity therapeutic acute toxicity study service ensures that your drug development path is built on a foundation of scientific integrity and safety.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Patel, C.; et al. Acute and sub-acute toxicity study of anti-obesity herbal granules in Sprague Dawley rats. Braz J Biol [Internet]. 2024, 84: e264320. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

