Anti-Obesity Therapeutic Single Dose Toxicity Study Service
InquiryThe escalating global prevalence of obesity and its associated metabolic comorbidities, such as type 2 diabetes and non-alcoholic fatty liver disease (NAFLD), has intensified the search for novel pharmacotherapies.
Single-Dose Toxicity Assessment for Anti-Obesity Therapies
Protheragen provides specialized anti-obesity therapeutic single-dose toxicity study services designed to evaluate the safety and initial toxicological profile of emerging compounds. These preclinical studies are fundamental for identifying the maximum tolerated dose (MTD) and potential target organ toxicity before advancing to repeated-dose programs. By leveraging high-fat diet (HFD) induced models and advanced metabolic monitoring, we ensure that your drug candidate's safety margins are rigorously established to meet the highest scientific standards and ensure reliable translational data.
Core Technologies
To provide a comprehensive assessment of Anti-Obesity Candidates, Protheragen integrates cutting-edge bioanalytical and physiological monitoring technologies. Our core capabilities include:
- High-Resolution Metabolic Phenotyping
Utilizing indirect calorimetry and specialized cages to monitor energy expenditure, respiratory exchange ratio (RER), and physical activity immediately following dose administration.

- Multiplex Biomarker Profiling
Precise quantification of acute changes in adipokines (leptin, adiponectin) and inflammatory markers (IL-6, TNF-α) to detect early systemic responses.
- Stereoselective Pharmacokinetic (PK) Analysis
Crucial for chiral compounds like HSG4112 analogs, where enantiomer-specific metabolic stability significantly impacts safety and efficacy (published data).
- Quantitative Histopathology
Utilizing AI-driven image analysis for the rapid detection of acute lipid accumulation or cellular damage in the liver, pancreas, and kidneys.
Solution Scope
Protheragen offers a versatile range of single-dose toxicity assessments tailored to different therapeutic modalities:
Evaluation of gastrointestinal lipase inhibitors (similar to Orlistat) and central nervous system (CNS) appetite suppressants.
Assessment of GLP-1 Receptor Agonists and other incretin mimetics, focusing on acute tolerability and injection site reactions.
- Phytotherapeutic Extracts
Safety screening for botanical compounds, such as Viola mandshurica extracts, ensuring high-dose safety (up to 5000 mg/kg) as demonstrated in relevant literature (Published data).
- Metabolic Targeted Therapies
Evaluating compounds designed to increase energy expenditure or activate AMPK pathways without inducing acute cardiovascular or respiratory distress.
Explore Our Full Service Scope.
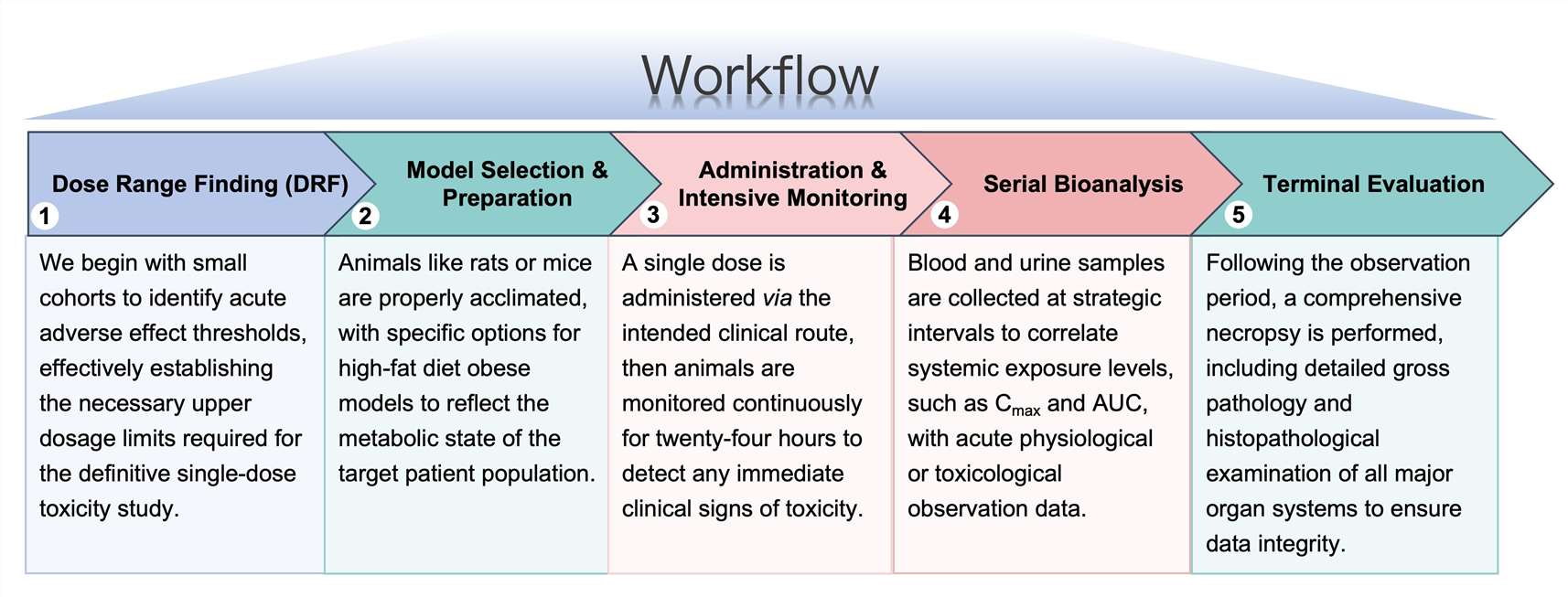
Workflow
Our workflow is engineered for speed and scientific rigor, ensuring that developers receive the data necessary to make critical go/no-go decisions.
Fields of Application
Our single-dose toxicity studies support various stages and types of anti-obesity research:
- Lead Optimization: Comparing the acute safety profiles of several structural analogs to select the most promising candidate.
- Pre-First-in-Human Safety Profiling: Delivering the critical acute safety data and toxicological assessments necessary to transition a candidate from the laboratory to early-phase clinical development.
- Mechanism of Action Studies: Identifying if acute toxicity is a direct result of the primary pharmacology or an off-target effect.
- Formulation Screening: Assessing how different delivery vehicles affect the acute tolerability and absorption kinetics of the active ingredient.
Advantages
Choosing Protheragen provides your development team with unmatched precision.
Metabolic Expertise
Unlike general CROs, we specialize in metabolic diseases, ensuring our pathologists recognize subtle acute changes in adipose and hepatic tissues.
Reliable Safety Benchmarking
We utilize historical data from established anti-obesity agents to provide context for your candidate's performance. For instance, while Orlistat is effective, its potential for acute kidney injury through oxalate precipitation is a known risk; we specifically monitor for such markers (published data).
Tailored Protocol Design and Rapid Turnaround
We adjust washout periods and observation windows based on the predicted half-life and mechanism of your specific molecule. Our streamlined internal logistics allow for the delivery of preliminary safety reports within weeks of study completion.
Contact Our Team for More Information and to Discuss Your Project.
Publication Data
Title: Anti-obesity activity, acute toxicity, and chemical constituents of aqueous and ethanol V. mandshurica extracts
Journal: BMC Complementary and Alternative Medicine, 2017
DOI: https://doi.org/10.1186/s12906-017-1810-4
Summary: This study investigated the effectiveness of V. mandshurica (VM), a perennial herb traditionally used as an anti-inflammatory and diuretic, in treating obesity. Researchers compared two types of extracts—ethanol (VME) and aqueous (VMA)—to determine their impact on high-fat diet (HFD)-induced obese mice over seven weeks. The study specifically focused on how low doses of these extracts affect body weight, fat accumulation, and metabolic markers, such as AMPK (adenosine monophosphate-activated protein kinase), which regulates energy balance. Additionally, the researchers performed acute toxicity tests to ensure the safety of the plant extracts for potential therapeutic use.
Key Findings
- Significant Weight and Fat Reduction: Daily oral administration of VME or VMA (at doses of 50, 100, or 200 mg/kg) led to a marked decrease in body weight gain, liver mass, and adipose (fat) tissue mass in obese mice.
- Improved Metabolic Markers: Both extracts significantly lowered serum levels of triglycerides, free fatty acids, total cholesterol, and "bad" LDL cholesterol. They also increased "good" HDL cholesterol and adiponectin, a hormone that helps regulate glucose and break down fatty acids.
- Activation of the AMPK Pathway: The extracts were found to activate the AMPK signaling pathway in both the liver and adipose tissue. This activation helps suppress the creation of new fats (lipogenesis) while promoting the burning of existing fat (fatty acid oxidation).
- Reduced Adipocyte Size: Microscopic analysis revealed that the treatments effectively inhibited the enlargement of fat cells (adipocytes), which is a hallmark of diet-induced obesity.
- High Safety Profile: In acute toxicity studies, a single massive oral dose of 5,000 mg/kg of either extract resulted in no mortality or clinical signs of toxicity over a 14-day observation period.
- Active Compounds Identified: Using high-performance liquid chromatography (HPLC), the researchers identified two primary bioactive compounds in the extracts: esculetin and schaftoside, both of which are known to have anti-obesity and fat-lowering properties.
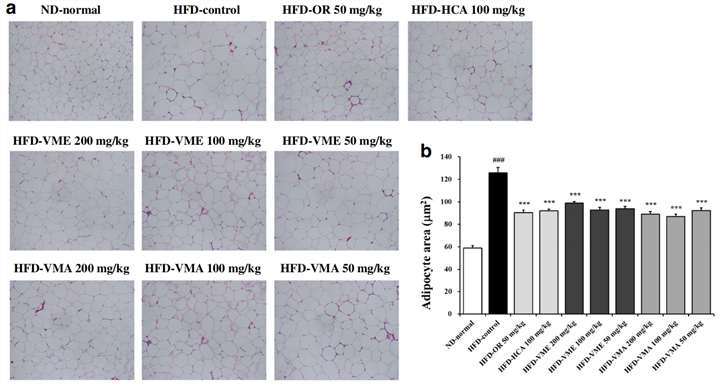 Fig.1 Effects of VME and VMA on histology of adipose tissue in HFD-induced obese mice. (Sung, et al., 2017)
Fig.1 Effects of VME and VMA on histology of adipose tissue in HFD-induced obese mice. (Sung, et al., 2017)
Customer Review
Strategic Insight into Metabolic Interactions
"Working with Protheragen was a pivotal step for our lead compound. Their team suggested using a high-fat diet model for our single-dose study, which revealed a unique metabolic interaction we wouldn't have seen in lean animals. Their reporting was exceptionally thorough and provided the comprehensive scientific evidence needed to confidently validate our data and advance the project to the next stage."
Senior VP of Preclinical Development, Biotech Firm
Precision Analytical Excellence in Isomer Differentiation
"The precision of Protheragen's stereoselective PK analysis was impressive. They helped us differentiate the safety profiles of our enantiomers, allowing us to move forward with the safer isomer with full confidence. We look forward to our upcoming 28-day study with their team."
Head of Toxicology, Pharmaceutical Innovation Lab
Frequently Asked Questions
-
Why is a single-dose study necessary if we plan for chronic administration?
It establishes the MTD and identifies target organs of toxicity, providing the safety ceiling for all subsequent multi-dose and chronic studies.
-
Can you conduct these studies in obese animal models?
Yes. While standard toxicity studies use healthy animals, we often recommend HFD-induced models for anti-obesity drugs to observe the drug's interaction with a compromised metabolic state.
-
What species are typically used?
Rats and mice are the most common; however, we can scale to non-rodent species if the drug's mechanism or PK requires it.
-
How do you monitor for CNS-related side effects common in anti-obesity drugs?
We utilize functional observational batteries (FOB) to monitor tremors, gait, and behavioral changes immediately post-dosing.
-
What markers do you use to detect acute liver injury?
We analyze a full panel of hepatic enzymes (ALT, AST, ALP) and perform histopathological scoring for acute steatosis or necrosis.
-
Does your service include pharmacokinetic (PK) analysis?
Yes, we offer integrated PK/TK (toxicokinetic) services to correlate dosing levels with actual blood concentrations.
-
How do you handle compounds with low solubility?
Our formulation experts can develop specialized vehicles to ensure optimal bioavailability for the toxicity assessment.
-
Can you detect potential for kidney stones, similar to the risks seen with some lipase inhibitors?
Yes, we include renal function markers and specialized staining (e.g., Pizzolato's) to detect calcium oxalate crystals in the renal tubules.
-
What is the typical duration of a single-dose toxicity study?
The observation period is usually 14 days post-administration to ensure any delayed toxicity is captured.
-
How do I start a project with Protheragen?
Simply reach out through our contact portal; our specialists will schedule a technical consultation to define your study parameters.
How to Contact Us
Protheragen is your dedicated partner in navigating the complex landscape of metabolic drug safety. Our anti-obesity therapeutic single-dose toxicity study service provides the rigorous data and scientific insight required to transform your innovative molecules into safe, viable therapies.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Sung, Y.Y.; et al. Anti-obesity activity, acute toxicity, and chemical constituents of aqueous and ethanol Viola mandshurica extracts. BMC Complement Altern Med. 2017, 17(1):297. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

