Anti-Obesity Therapeutic Sub-acute Toxicity Study Service
InquiryThe global rise in metabolic disorders has driven a surge in the development of next-generation anti-obesity medications (AOMs), encompassing a range of approaches from GLP-1/GIP receptor agonists to novel peptide blends and small-molecule metabolic activators. However, transitioning from efficacy to safety is the most critical hurdle in the preclinical pipeline. Protheragen provides specialized anti-obesity therapeutic sub-acute toxicity study services designed to characterize the safety profile of candidate compounds over repeated-dose exposure.
Sub-Acute Toxicity Evaluation for Anti-Obesity Therapies
A sub-acute toxicity study (typically 14 to 28 days) is essential for identifying target organ toxicity, dose-response relationships, and potential adverse effects that may not be apparent in acute single-dose studies. Our service focuses exclusively on the preclinical stage, providing the rigorous data required to establish the no-observed-adverse-effect level (NOAEL) and guide subsequent chronic safety evaluations. By leveraging our deep expertise in metabolic biology, we help clients navigate the complexities of long-term dosing early in the development cycle.
Core Technologies
Protheragen employs a suite of advanced technologies to ensure the highest resolution of safety data for anti-obesity candidates.
- Automated Metabolic Monitoring Systems
We utilize high-precision telemetry and home-cage monitoring to track real-time changes in feed intake, water consumption, and physical activity during the dosing period, which is critical for AOMs that target the central nervous system or gut-brain axis.
- High-Resolution Histopathology & Digital Imaging
Our pathology suite is equipped with automated tissue processing and AI-assisted image analysis to detect subtle morphological changes in key metabolic organs, including the liver (steatosis assessment), pancreas, and adipose tissues (white vs. brown fat transformation).
- Multi-Analyte Clinical Chemistry Platforms
We provide comprehensive metabolic profiling, including markers for hepatic function, renal integrity, and lipid metabolism, ensuring that potential systemic toxicities are identified with high sensitivity.
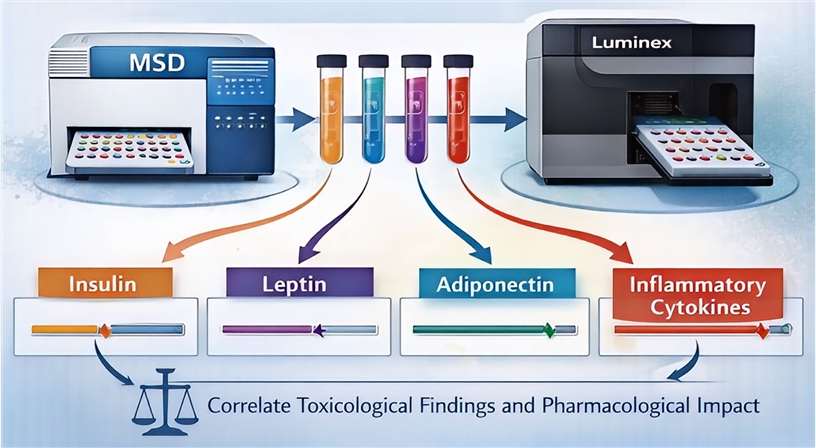
- Targeted Biomarker Immunoassays
Using multiplex platforms like Meso Scale Discovery (MSD), we quantify circulating levels of insulin, leptin, adiponectin, and inflammatory cytokines to correlate toxicological findings with the drug's pharmacological impact.
Solution Scope
Protheragen offers a flexible range of preclinical sub-acute toxicity evaluations for various therapeutic modalities:
- Repeated-Dose Toxicity (14/28 Days)
Determination of toxicity following daily administration, identifying target organs and dose-limiting toxicities.
- Dose Range Finding (DRF) Studies
Preliminary assessments to select the optimal doses for pivotal studies.
- Specific Organ Toxicity Panels
Specialized focus on hepatotoxicity (e.g., evaluation of NASH/NAFLD regression vs. toxic insult) and cardiotoxicity.
- Recovery Studies
Assessment of the reversibility or persistence of toxic effects following a treatment-free period.
- Route-Specific Assessments
Expertise in various administration routes, including subcutaneous injection (common for peptides) and oral gavage (Small Molecules).
Explore Our Full Service Scope.
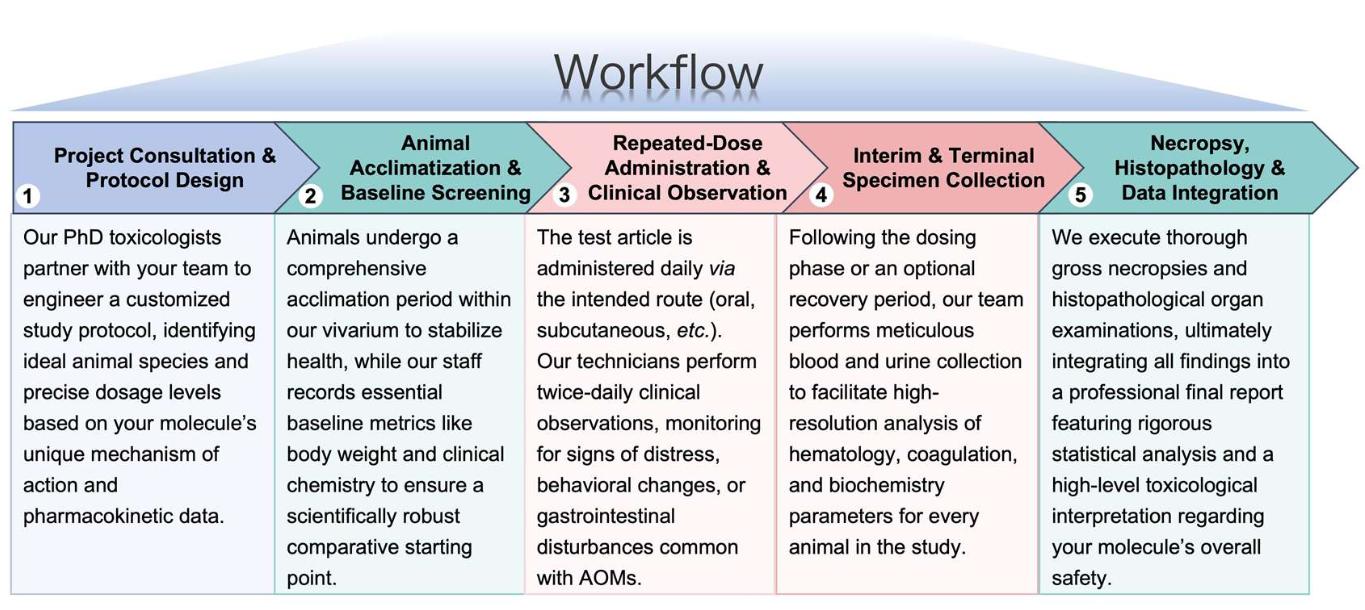
Workflow
Our streamlined service process is designed for transparency and scientific rigor.
Fields of Application
Our sub-acute toxicity services are essential for researchers and companies developing:
- Incretin Mimetics: GLP-1, GIP, and Glucagon Receptor Agonists (single, dual, or triple).
- Metabolic Activators: Sirtuin activators, AMPK Agonists, and mitochondrial uncouplers.
- Nutrient Absorption Blockers: Lipase inhibitors and carbohydrate absorption modulators.
- CNS Appetite Suppressants: MC4R agonists and other neuropeptide modulators.
- Novel Phytomedicines: Standardized botanical extracts and polyherbal formulations targeting weight management.
Advantages
Choosing Protheragen for your anti-obesity safety studies provides distinct competitive benefits:
Specialized Metabolic Expertise
Unlike generalist CROs, we understand the nuances of anti-obesity drug discovery. We can distinguish between pharmacological weight loss (efficacy) and toxicological cachexia (safety).
Accelerated Timelines
Our optimized workflow and dedicated metabolic vivarium allow for rapid study initiation and data turnaround, keeping your program on track.
Advanced Histopathology Suite
We specialize in the complex pathology of metabolic tissues, providing insights into white adipose tissue browning and liver lipid clearance that go beyond standard toxicity checks.
Success Stories
By providing clear and actionable NOAEL data, we have successfully supported the safety characterization of multiple GLP-1 Analogues and Dual Agonists, providing a robust scientific foundation for candidate progression beyond the lead optimization phase.
High-Fidelity Scientific Documentation
Protheragen adheres to the most rigorous international laboratory standards, ensuring that every study generates "published data" quality reports. These comprehensive dossiers are specifically structured to support deep-dive investor due diligence and provide a transparent, high-resolution record of your molecule's safety profile. By delivering data of this caliber, we ensure your project possesses the scientific credibility required for high-stakes decision-making and successful partnership transitions.
Contact Our Team for More Information and to Discuss Your Project
Publication Data
Title: Acute and 28-days subacute toxicity studies of Gαq-RGS2 signaling inhibitor
Journal: Laboratory Animal Research, 2021
DOI: https://doi.org/10.1186/s42826-021-00093-1
Summary: This study evaluated the acute and 28-day subacute toxicity of the synthetic Gαq-RGS2 signaling inhibitor (1-(5-chloro-2-hydroxyphenyl)-3-(4-(trifluoromethyl)phenyl)-1H-1,2,4-triazol-5(4H)-one) in rodents, following OECD guidelines 425 and 407, respectively. The compound, a triazolone ring-containing entity with potential therapeutic applications (e.g., for urinary incontinence, cardiac arrhythmia, cardiovascular diseases), was tested to assess its safety profile—a critical step before clinical development. Acute toxicity tests involved single oral doses up to 2000 mg/kg in mice, while subacute tests used repeated doses of 10 and 100 mg/kg in rats for 28 days. Key evaluations included mortality, body weight changes, hematological/biochemical parameters, organ damage markers, and histological examinations of essential organs. Results confirmed the compound's safety at the tested doses, supporting its potential for further preclinical and clinical advancement.
Key Findings
- Acute Toxicity Safety: A single oral dose of up to 2000 mg/kg in mice caused no mortality or adverse clinical signs (e.g., altered muscle/reflex/secretory activity) over 14 days. Per the Hodge and Sterner Scale, the compound is classified as slightly toxic.
- Subacute Toxicity Safety: Repeated 28-day administration of 10 and 100 mg/kg in rats resulted in no mortality or clinical abnormalities. Body weight gains were comparable to those of the control groups (normal and 1% DMSO vehicle) across the study period.
- Unchanged Hematological/Biochemical Parameters: No significant differences were observed in hematological indices (RBC, WBC, platelets, hemoglobin) or biochemical markers (cardiac: CK-MB, LDH; liver: ALT, AST; kidney: urea, creatinine; metabolic: glucose, cholesterol, triglycerides) between treated and control groups.
- No Organ Toxicity: Relative organ-to-body weight ratios (heart, kidney, liver, brain, lung) and histological examinations revealed no significant damage (e.g., inflammation, necrosis, atrophy) in essential organs of treated rats compared to controls.
- Gender Independence: Toxicity profiles showed no significant gender-related differences in hematological or biochemical parameters.
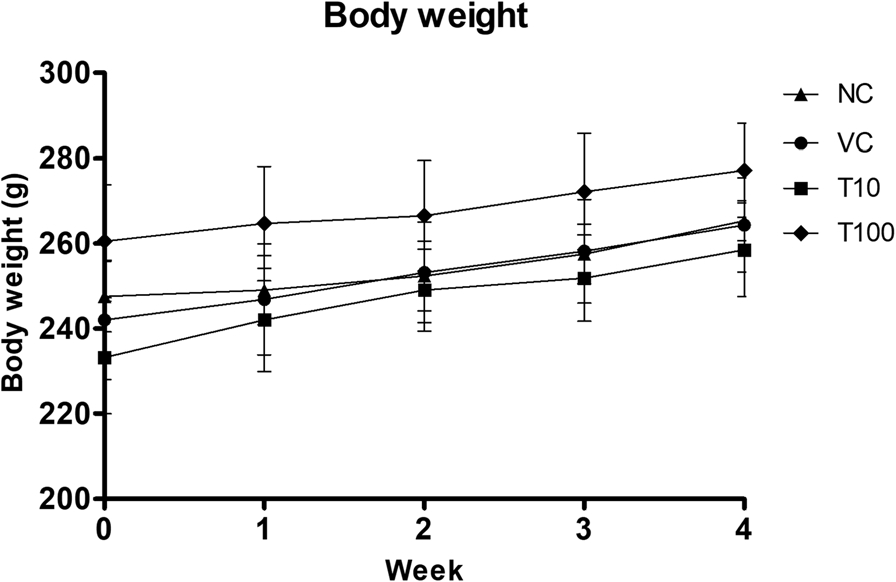 Fig.1 Effect of Gαq-RGS2 signaling inhibitor on body weight. (Beladiya, et al., 2021)
Fig.1 Effect of Gαq-RGS2 signaling inhibitor on body weight. (Beladiya, et al., 2021)
Customer Review
Precision in Metabolic Data Interpretation
"Working with Protheragen was a turning point for our lead peptide program. Their team didn't just provide raw data; they helped us interpret the subtle changes in liver enzymes that other labs might have overlooked. Their specialized knowledge in metabolic disease made the difference in our dose-selection strategy."
Senior Director of Discovery, Metabolic Therapeutics Startup
Comprehensive Safety Mapping for Novel Multi-Agonists
"Our team was developing a complex multi-mechanism agonist, and we were concerned about potential off-target effects in the hypothalamus. Protheragen provided an exceptional acute toxicity study that utilized high-resolution digital histopathology to give us absolute clarity. Their AI-assisted tissue analysis identified markers of inflammation early on, allowing us to pivot our chemical optimization before investing in larger trials. They are a true partner in metabolic safety."
Chief Scientific Officer, Metabolic Therapeutics Start-up
Frequently Asked Questions
-
Why is a 28-day study preferred over a 14-day study for anti-obesity drugs?
A 28-day study provides a more comprehensive view of the cumulative effects and potential target organ adaptation, which is vital for drugs intended for long-term chronic use in weight management.
-
Can you perform these studies in diet-induced obese (DIO) models?
Yes, while standard toxicity is often done in healthy animals, we can conduct sub-acute safety assessments in DIO models to observe how the drug interacts with existing metabolic comorbidities.
-
What is the typical turnaround time for a final report?
From the completion of the in-life phase, a draft report is typically available within 4–6 weeks, including full histopathology analysis.
-
Do you offer recovery groups in your sub-acute protocols?
Absolutely. We often recommend a 14-day recovery period to determine if any observed effects (like elevated liver enzymes) are reversible once dosing stops.
-
How do you handle high-potency peptides?
Our facility is equipped with specialized handling protocols and high-precision dosing equipment to ensure accurate administration of low-volume, high-potency anti-obesity peptides.
-
Is the histopathology of the brain included?
For AOMs that cross the blood-brain barrier or target the CNS, we include detailed neurohistopathology of the hypothalamus and brain stem.
-
Can you analyze biomarkers like GLP-1 or insulin during the study?
Yes, we can integrate pharmacodynamic biomarker analysis into the toxicity study to provide a holistic view of the drug's physiological impact.
-
What species do you typically use?
The Sprague-Dawley rat is the standard, but we also utilize various mouse strains depending on the client's previous efficacy data.
-
How do you distinguish between weight loss from the drug and weight loss from toxicity?
We monitor food efficiency ratios and specific clinical markers; pharmacological weight loss is usually accompanied by healthy metabolic shifts, whereas toxicity involves distress, lethargy, or organ dysfunction.
-
How can I start a project with Protheragen?
Simply reach out via our inquiry portal. We will schedule a technical call to discuss your molecule and provide a customized study design and quote.
How to Contact Us
Protheragen is a leading provider of anti-obesity therapeutic sub-acute toxicity study services, offering specialized preclinical expertise to ensure your metabolic drug candidates are both effective and safe. By combining advanced metabolic monitoring with rigorous toxicological analysis, we provide the data-driven insights necessary to advance your pipeline with confidence.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Beladiya, J.V.; et al. Acute and 28-days subacute toxicity studies of Gαq-RGS2 signaling inhibitor. Lab Anim Res. 2021, 37, 17. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

