Anti-Obesity Therapeutic Dose Range Study Service
InquiryThe global obesity epidemic has accelerated the demand for next-generation pharmacotherapies that offer superior efficacy and safety profiles compared to historical standards. As the therapeutic landscape shifts from mono-agonists to complex multi-receptor modalities—such as GLP-1/GIP/glucagon triple agonists and novel small molecule sensitizers—the need for precise, high-fidelity preclinical data is critical.
Anti-Obesity Therapy: Dose Range Evaluation Service
Protheragen provides a dedicated anti-obesity therapeutic dose range study service engineered to assist developers in pinpointing the ideal therapeutic window during the early stages of discovery. We utilize translationally robust animal models, specifically the diet-induced obese (DIO) mouse, to accurately replicate human metabolic dysfunction. By systematically assessing efficacy across a spectrum of dosage levels, we deliver the comprehensive data packages necessary to validate your compound's potential and confidently de-risk your development pipeline.
Core Technologies
To deliver high-precision data, Protheragen integrates cutting-edge physiological monitoring with molecular profiling:
Utilization of indirect calorimetry systems (Oxymax/CLAMS) to measure oxygen consumption (VO2), CO2 production, and respiratory exchange ratio (RER) in real-time under varying dosage regimens.
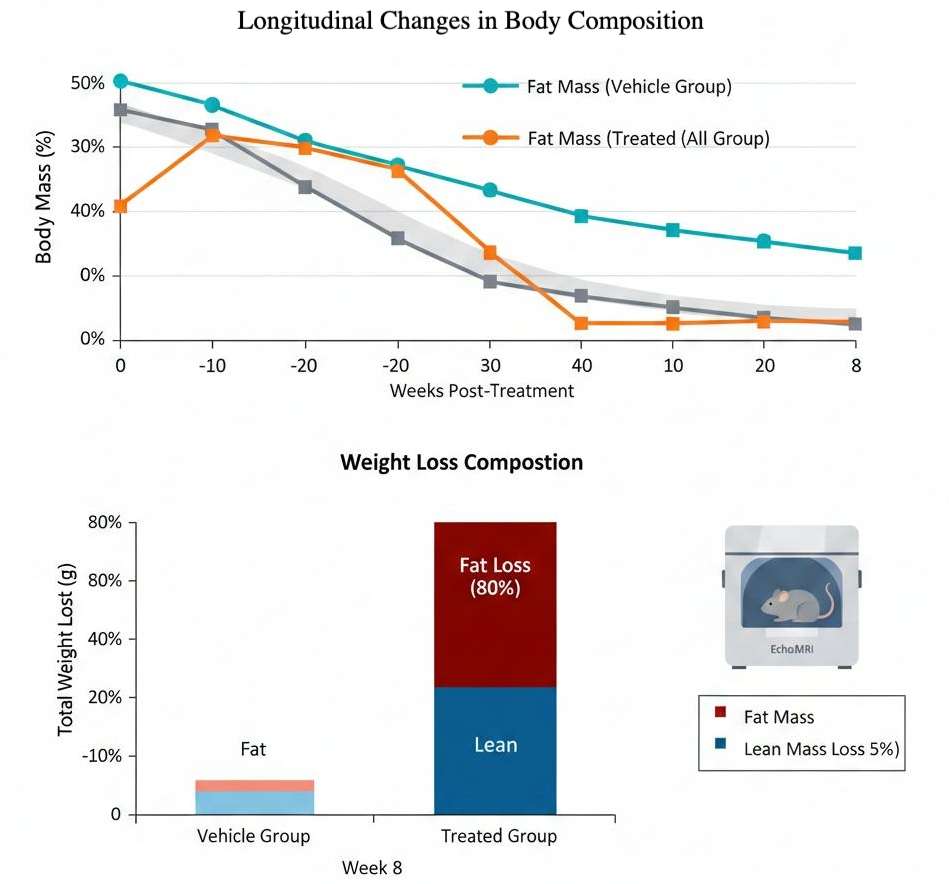
- Body Composition Analysis
Non-invasive EchoMRI or dual-energy X-ray absorptiometry (DXA) to quantify longitudinal changes in fat mass versus lean mass, ensuring weight loss is adipose-specific.
- Multiplex Incretin Profiling
High-sensitivity assays to measure endogenous insulin, leptin, adiponectin, and ghrelin levels, providing a holistic view of the compound's impact on the endocrine system.
- Continuous Glucose Monitoring (CGM) Integration
Wireless telemetry for 24/7 glucose tracking is essential for assessing the glycemic stability of dual or triple-agonist candidates.
Solution Scope
Protheragen offers a modular approach to dose-range studies, adaptable to various therapeutic modalities:
- Peptide & Biologic Titration
Evaluation of long-acting acylated peptides and monoclonal antibodies.
- Small Molecule Pharmacokinetics
Assessment of oral bioavailability and dose-proportionality in metabolic tissues.
- Combination Therapy Synergism
Specialized studies investigating the additive or synergistic effects of dual-targeting agents (e.g., GLP-1/glucagon co-agonists).
- Chronic vs. Acute Dosing
Comparative studies between single-dose "washout" kinetics and chronic 28-day steady-state evaluations.
Unlock the Full Potential of Your Metabolic Pipeline. Inquire about Study Design and Pricing.
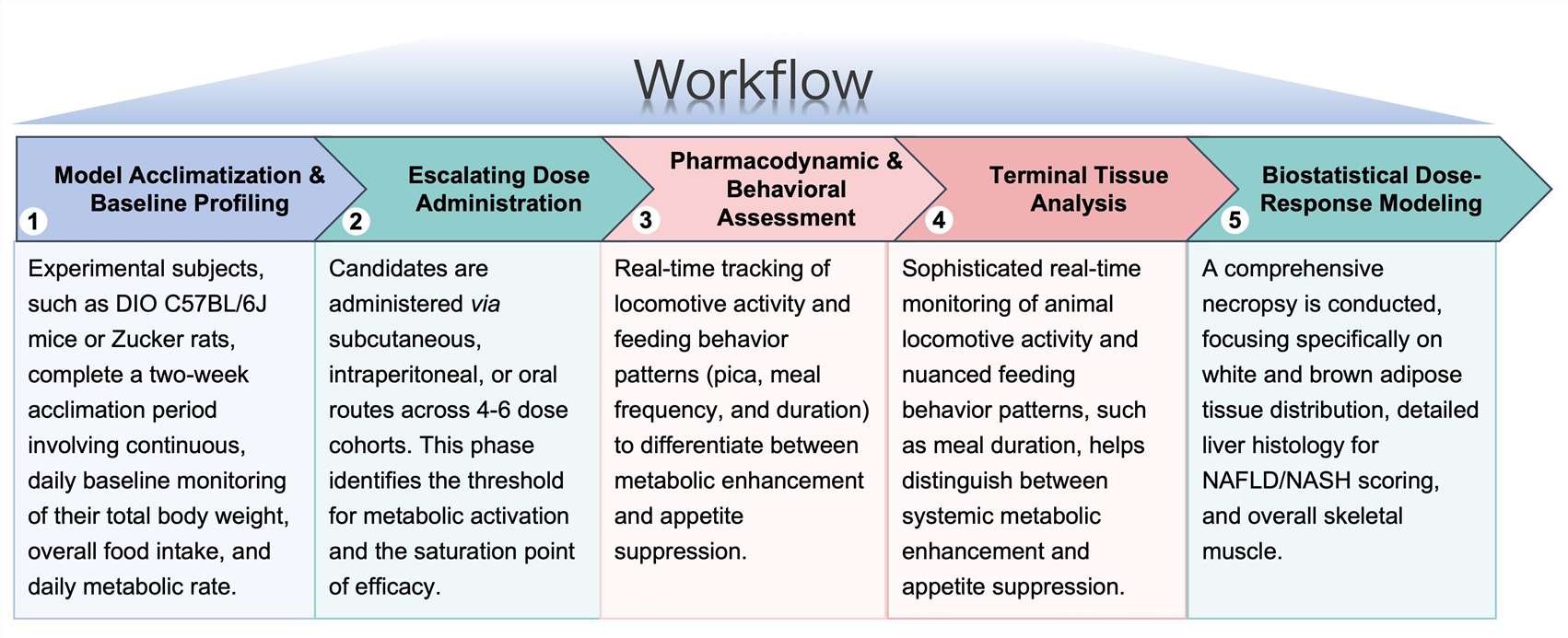
Workflow
Our streamlined preclinical workflow is designed for reproducibility and rapid data turnaround:
Fields of Application
The specialized dose-range studies conducted by Protheragen serve as a pivotal bridge in the development of diverse metabolic therapies, providing the foundational pharmacodynamic data required to transition novel compounds from initial discovery into rigorous preclinical validation.
- Incretin Mimetic Development: Determining the optimal dose for GLP-1, GIP, and oxyntomodulin analogs.
- Thermogenic Activators: Assessing the efficacy of UCP1 activators and brown adipose tissue (BAT) stimulants.
- Lipid Metabolism Modulators: Investigating PPAR agonists and THR-β selective ligands.
- CNS-Targeted Anorectics: Evaluating dose-dependent appetite suppression and potential neurobehavioral side effects.
Advantages
Choosing Protheragen ensures your drug development program is anchored by scientific excellence and operational efficiency.
Unparalleled Predictive Accuracy and Rapid Cycle Times
Our DIO Models closely mimic human metabolic syndrome, significantly increasing the translatability of preclinical ED50 values. Our specialized facility layout allows for the simultaneous execution of multiple dose cohorts, shortening your development timeline by weeks.
Lean Mass Preservation Focus
Unlike generic studies, we prioritize the "quality of weight loss," providing "published data" demonstrating our ability to distinguish between lean mass wasting and healthy adiposity reduction.
Comprehensive Technical Reporting
Every study concludes with an exhaustive data dossier featuring high-resolution visualization of metabolic trends and rigorous statistical validation, ensuring your team possesses a complete and audit-ready pharmacological profile of your lead candidate.
Contact Us to Design a Customized Dose-range Study that Defines Your Compound's Therapeutic Window.
Publication Data
Title: BI 456906: Discovery and preclinical pharmacology of a novel GCGR/GLP-1R dual agonist with robust anti-obesity efficacy
Journal: Boehringer Ingelheim Pharma, 2022
DOI: https://doi.org/10.1016/j.molmet.2022.101633
Summary: This paper focuses on the discovery and preclinical pharmacology of BI 456906, a novel once-weekly injectable GCGR/GLP-1R dual agonist developed for obesity, type 2 diabetes, and non-alcoholic steatohepatitis (NASH). Through in vitro and in vivo studies (including cell-based assays, animal models of DIO, and transcriptional analysis), the research demonstrates that BI 456906 exerts robust anti-obesity efficacy by balancing activation of both receptors: it reduces food intake and gastric emptying via GLP-1R agonism, and increases energy expenditure via GCGR agonism. Compared with the selective GLP-1R agonist semaglutide, BI 456906 achieves superior bodyweight reduction in DIO mice while maintaining glycemic control. Additionally, transcriptional and biomarker analyses reveal its potential hepatoprotective effects, supporting its clinical development for NASH.
Key Findings
- Molecular & Dosing Advantage: BI 456906 is a 29-amino-acid acylated peptide with a C18 fatty acid, enabling once-weekly dosing. It potently activates both human GCGR (EC50=0.52 nM) and GLP-1R (EC50=0.33 nM) in vitro.
- Superior Weight Loss Efficacy: In DIO mice, 30 nmol/kg BI 456906 reduced bodyweight by 32% over 30 days—more than semaglutide's maximum effect (25–27%).
- Dual Mechanism of Action: It cuts food intake and slows gastric emptying (via GLP-1R) while boosting energy expenditure (via GCGR), with energy expenditure increases independent of bodyweight.
- Glycemic & Metabolic Benefits: Unlike standalone GCGR agonists, it maintains normal blood glucose, lowers liver triglycerides/cholesterol, and reduces plasma insulin/leptin in DIO mice.
- GCGR-Specific Biomarkers: It increases liver NNMT mRNA and plasma FGF-21, and reduces plasma amino acids (serine, glutamine)—key signs of GCGR activation.
- Hepatoprotective Potential: It regulates 93 liver genes linked to anti-fibrosis and metabolism, with gene expression patterns opposing those in human NASH fibrosis.
- Dual Receptor Engagement: In transgenic mice, it activates both GLP-1R (pancreas) and GCGR (liver) simultaneously, while semaglutide (GLP-1R-only) and LA-GCG (GCGR-only) act on single receptors.
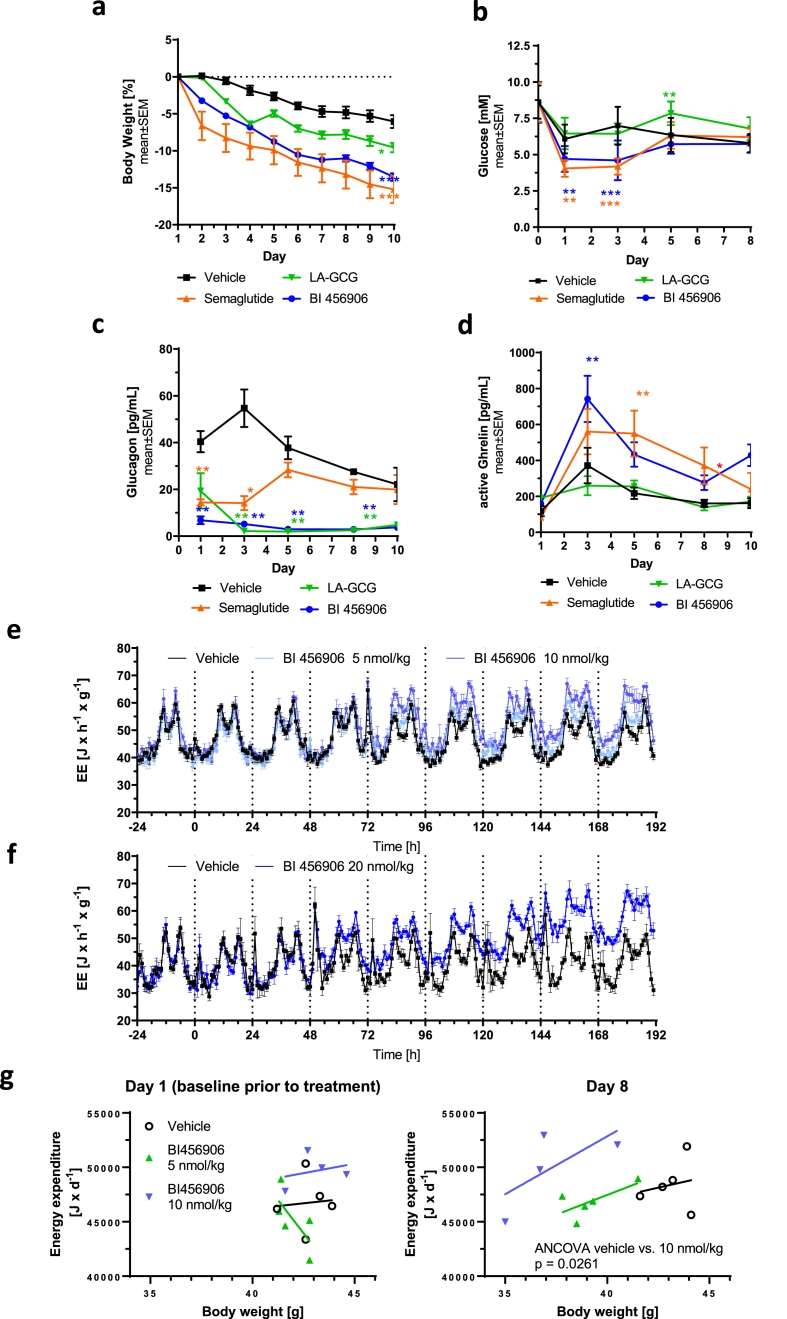 Fig.1 BI 456906 demonstrates significantly greater bodyweight-lowering efficacy compared with maximally effective doses of semaglutide in DIO mice by engaging the glucagon receptor. (Zimmermann, et al., 2022)
Fig.1 BI 456906 demonstrates significantly greater bodyweight-lowering efficacy compared with maximally effective doses of semaglutide in DIO mice by engaging the glucagon receptor. (Zimmermann, et al., 2022)
Customer Review
Precision Engineering of Therapeutic Windows
"Partnering with Protheragen fundamentally transformed our lead optimization phase. We were struggling to find the precise balance between metabolic activation and appetite suppression for our dual-agonist candidate. The team at Protheragen provided a high-resolution dose-response map that clearly identified the optimal concentration for maximizing fat oxidation without triggering adverse gastrointestinal responses. Their expertise in indirect calorimetry allowed us to visualize real-time metabolic shifts that we simply couldn't capture in-house. We are already planning our next longitudinal study with them to explore chronic dosing effects."
Director of Metabolic Research, Mid-sized Biotech
Unmatched Data Fidelity for Competitive Benchmarking
"What sets Protheragen apart is its commitment to the 'quality' of weight loss. During our recent study, their body composition analysis provided the critical evidence we needed to prove that our compound preserved lean muscle mass significantly better than current market benchmarks. The data was so meticulously organized and statistically robust that it integrated seamlessly into our internal pharmacology presentations. They acted as a true extension of our scientific team, providing insights that helped us refine our dosing strategy for the next stage of development. We look forward to a long-term collaboration as our pipeline expands."
Principal Pharmacologist, Mid-sized Biotech
Frequently Asked Questions
-
What is the standard duration for an anti-obesity dose-range study?
Typically, acute studies last 7–14 days, while chronic efficacy studies targeting steady-state weight loss range from 28 to 56 days.
-
Can you accommodate oral gavage for small molecule testing?
Yes, we have dedicated teams proficient in daily oral dosing, as well as subcutaneous and intravenous administration.
-
How do you ensure the stability of peptide drugs during the study?
We utilize specialized cold-chain handling and can perform stability testing in the dosing vehicle before administration.
-
Is it possible to include a reference drug like Semaglutide?
Absolutely. We frequently include positive controls to provide a benchmark for your candidate's performance.
-
Do you provide histopathology services for the liver?
Yes, we offer full H&E and Oil Red O staining to evaluate the impact on hepatic steatosis.
-
What species are available for these studies?
We primarily utilize specialized rodent strains, including C57BL/6J DIO mice, ob/ob mice, and Sprague-Dawley rats.
-
How do you measure food intake accurately?
We use high-precision automated feeders that record consumption to the nearest 0.01g, accounting for spillage.
-
Can we customize the frequency of blood sampling?
Yes, we offer tail-vein sampling or cannulation for longitudinal PK/PD profiling without significant stress to the animal.
-
What happens if my compound shows toxicity at low doses?
We provide immediate "stop-study" notifications and preliminary necropsy findings to help you pivot your strategy.
-
Do you assist with the calculation of the Human Equivalent Dose (HED)?
While we focus on preclinical data, our reports provide the essential mg/kg metrics required for your clinical pharmacology team to calculate HED.
How to Contact Us
Protheragen is dedicated to advancing the science of weight management through rigorous preclinical excellence. Our team of expert biologists is ready to assist you in navigating the complexities of dose-response modeling and metabolic phenotyping.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Zimmermann, T.; et al. BI 456906: Discovery and preclinical pharmacology of a novel GCGR/GLP-1R dual agonist with robust anti-obesity efficacy. Mol Metab. 2022, 66: 101633. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

