Anti-Obesity miRNA Therapy Development
InquiryOverview
MicroRNAs (miRNAs) have emerged as potential therapeutic targets due to their regulatory roles in numerous biological processes, including transcriptional regulation of metabolism. These small non-coding RNA molecules act as master regulators of gene expression, coordinating various biological processes. In the field of metabolic health, miRNAs play a critical role in the key functions of adipose tissue, including adipocyte development and energy expenditure regulation through thermogenesis. Numerous studies have revealed hundreds of differentially expressed miRNAs in metabolic tissues of obese individuals, validating their potential as therapeutic targets for restoring metabolic homeostasis.
miRNA Precision, Targeting Obesity at the Source
Protheragen specializes in the development of Anti-obesity Therapies, leveraging its forward-looking insights into the molecular mechanisms of obesity and innovative breakthroughs in novel delivery systems to establish a comprehensive miRNA therapy development platform spanning the entire chain from "target discovery to drug design to preclinical validation." We are dedicated to developing innovative weight-loss therapies through precise regulation of miRNA activity and accelerating the advancement of anti-obesity miRNA therapies toward safety, efficacy, and personalization.
Therapy Development
- Our core strategy revolves around two main treatment approaches: miRNA mimics and anti-miRNAs. By restoring miRNA function or binding to target miRNAs and inhibiting their function, one enhances metabolism, reduces fat mass, and achieves weight loss effects. Concurrently, we employ chemical modifications to enhance miRNA stability and binding affinity to targets, thereby improving treatment efficacy.
- Precise delivery of miRNA therapy to the correct cells and tissues is one of the challenges. We are also dedicated to developing efficient delivery systems that protect miRNA molecules from degradation and safely and effectively deliver them to cells, thereby improving R&D efficiency.
Preclinical Studies
In the development of anti-obesity therapies, preclinical research serves as a critical bridge between basic research and human trials. Its core objective is to validate the safety, efficacy, and mechanism of action of candidate molecules through a multidimensional experimental system, providing a scientific basis for subsequent trials. We possess extensive Obesity Cell and Animal Models and conduct detailed pharmacodynamic studies, pharmacokinetic studies, safety assessments, and bioanalytical analyses for miRNA therapies.
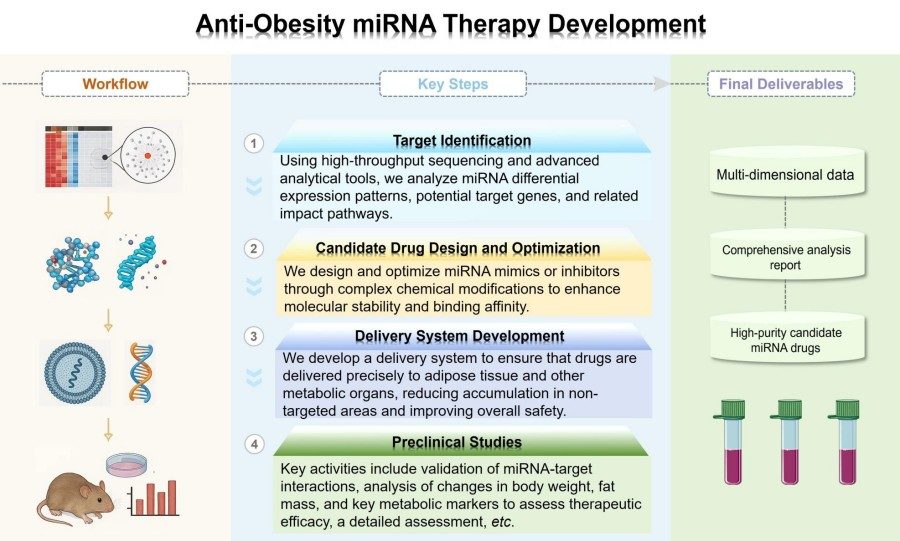
Workflow
Our services include a carefully designed end-to-end process aimed at reducing the risks associated with each stage of anti-obesity miRNA therapy development.
Target Identification
This stage involves systematically identifying key genes or signaling pathways associated with obesity as potential targets for miRNA regulation. Using high-throughput sequencing and advanced bioinformatics platforms, we analyze miRNA expression patterns to identify miRNAs that are differentially expressed between obese and normal metabolic tissues. We also utilize advanced tools to predict potential target genes of miRNAs and their associated signaling pathways.
Candidate Drug Design and Optimization
After identifying targets, we design and optimize therapeutic candidate drugs, namely miRNA mimics or inhibitors. Through complex chemical modifications, we enhance molecular stability and binding affinity. We also utilize proprietary platforms combining computational modeling and high-throughput screening to evaluate and optimize key pharmacologic properties, including efficacy and specificity.
Delivery System Development
The efficacy of therapeutic molecules depends on their delivery method. To this end, we have established specific steps for customizing and optimizing delivery system formulations. Through optimization, we ensure targeted delivery to adipose tissue and other metabolic organs, minimize non-targeted accumulation, and enhance overall safety.
Preclinical Research
After identifying candidate drugs, we conduct comprehensive preclinical validation. This includes in vitro and in vivo studies in relevant obesity models. Key activities include validation of miRNA-target interactions, analysis of changes in body weight, fat mass, and key metabolic markers to assess therapeutic efficacy, and a detailed assessment of the safety of the therapy.
Data Analysis
We systematically integrate multidimensional data from target discovery to preclinical studies and ultimately deliver highly detailed analytical reports and high-purity candidate miRNA drugs to support our clients' subsequent research.
Applications
The development of anti-obesity miRNA therapy aims to target molecular pathways involved in fat production, and other processes, improving a variety of complications at the same time.
miRNA therapy is used in combination with other weight loss drugs or technologies to develop combination therapies that work synergistically to improve weight loss results.
miRNA therapy research provides useful information for the development of personalized obesity intervention measures.
Advantages
Customized Experimental Protocols
We offer comprehensive, flexible, and integrated service models designed to meet the diverse needs of our clients. From basic discovery to preclinical research, our service solutions can be customized to meet client requirements.
Delivery System Development
We focus on developing highly efficient targeted delivery systems designed to significantly enhance the affinity of therapeutic molecules for their targets while precisely regulating their specificity and selectivity, thereby maximizing therapeutic efficacy.
Extensive Obesity Model Library
We have a rich collection of obesity models to explore miRNA expression differences, miRNA regulation of target genes, and physiological and pathological changes following drug administration.
Our team has been deeply involved in obesity mechanism research for many years and has built a full-chain technology platform covering risk prediction, pathology research, and personalized intervention analysis. By integrating multi-omics data and AI algorithms, we provide diversified service modules.
Publication Data
Title: Intravenous injection of microvesicle-delivery miR-130b alleviates high-fat diet-induced obesity in C57BL/6 mice through translational repression of PPAR-γ
Journal: Journal of Biomedical Science, 2015
DOI: https://doi.org/10.1186/s12929-015-0193-4
Summary: This study analyzed the effects of microvesicle (MV)-delivered miR-130b (miR-130b-MV) on mice induced by a high-fat diet. The results showed that after administration, the body weight and epididymal fat weight of the mice were significantly reduced, and glucose tolerance was partially restored. The expression of miR-130b in epididymal fat was significantly increased, and the lipolysis genes, hormone-sensitive lipase, monoglyceride lipase, and leptin were significantly upregulated. The protein content of its target gene, PPAR-γ, was significantly inhibited. The expression of miR-378a and miR-378-3p, which are involved in regulating fat deposition, was also increased. This study demonstrates that miR-130b-MV can reduce epididymal fat deposition and partially restore glucose tolerance by inhibiting PPAR-γ, providing data support for the development of anti-obesity therapies.
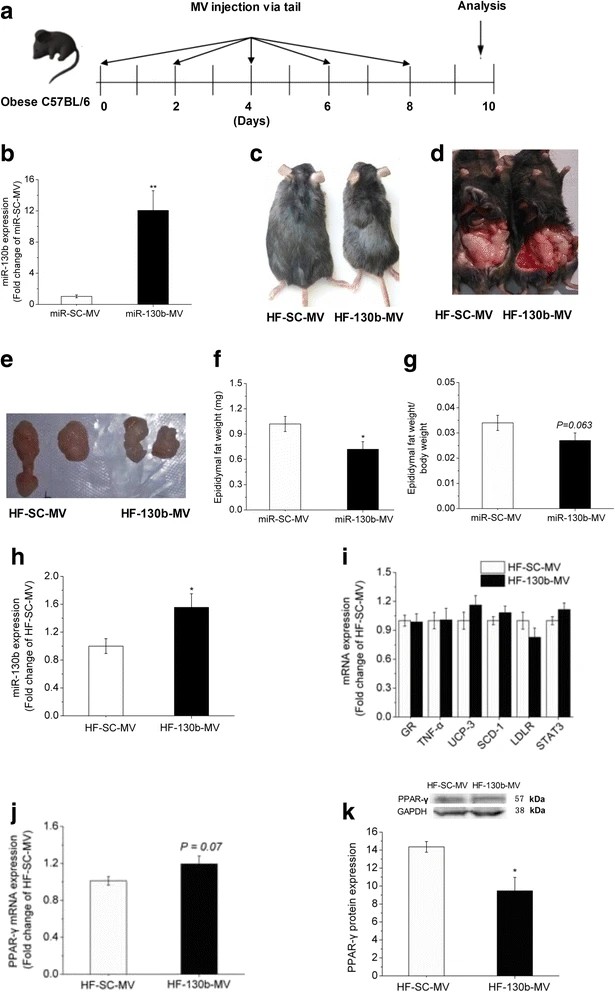 Fig. 1 Analysis after miR-130b administration. (Pan, et al., 2015)
Fig. 1 Analysis after miR-130b administration. (Pan, et al., 2015)
Customer Review
High-Quality Functional Testing
"Protheragen leveraged its in-house models to conduct in-depth functional testing on our customized miRNA, delivering highly reliable in vitro data. This efficient validation process significantly accelerated the validation of our anti-obesity targets."— A. C., Research Scientist
Strong Expertise
"Protheragen's team demonstrated compelling expertise in complex non-viral delivery systems. They successfully customized a liver-targeting LNP system for our miRNA, effectively ensuring efficient cellular uptake and functional release during in vivo experiments."— B. L., Technical Program Director
Frequently Asked Questions
-
How do you ensure that miRNA therapy is delivered specifically to adipose tissue rather than other organs?
Targeted delivery is at the core of our platform. We combine targeted delivery systems with optimization techniques to develop formulations with specific biodistribution characteristics. This ensures that therapeutic drugs are delivered precisely to the target site (such as adipose tissue) while avoiding non-targeted accumulation, which is a key differentiator in this field.
-
How do I start a project?
The first step in starting a project is to have a confidential consultation with our scientific team. We are happy to discuss your specific project goals and demonstrate how Protheragen can provide you with a clear and accelerated path to achieving them.
miRNA therapy represents a cutting-edge approach capable of regulating entire metabolic pathways, offering a comprehensive solution for obesity and its associated complications. Protheragen is at the forefront of this revolution and provides end-to-end obesity therapy development services. For more information about our obesity miRNA therapy development services or to discuss your specific project objectives, please contact us.
Reference
- Pan, S.; et al. Intravenous injection of microvesicle-delivery miR-130b alleviates high-fat diet-induced obesity in C57BL/6 mice through translational repression of PPAR-γ. Journal of biomedical science. 2015, 22(1): 86. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

