Targeting PCSK9 for Developing Anti-Obesity Therapeutics
InquiryOverview
Proprotein convertase subtilisin/kexin type 9 (PCSK9), a potent cholesterol metabolism regulator, is primarily synthesized and secreted by the liver. Its core mechanism involves mediating the endosomal degradation of low-density lipoprotein receptors (LDLR), thereby elevating circulating low-density lipoprotein cholesterol (LDL-C) levels. PCSK9 inhibitors have been developed to effectively lower LDL cholesterol. Deepening research reveals PCSK9's pivotal role in systemic metabolic dysregulation, encompassing chronic inflammation, mitochondrial homeostasis disruption, and widespread disturbances in lipid and cholesterol metabolism. These non-LDLR functions position PCSK9 as a potential therapeutic target for reversing metabolic dysfunction, such as obesity.
Precision Targeting of PCSK9: Reshaping Anti-Obesity Therapies
Protheragen is leveraging its established Anti-Obesity Therapeutic Development platform to pioneer a new generation of therapies focused on PCSK9's role in obesity-related pathways. Our core strategy involves screening for inhibitors that preferentially modulate this peripheral mechanism, achieving superior anti-obesity efficacy through regulation of metabolism and energy expenditure.
Therapy Development
Leveraging PCSK9's regulatory role in metabolism, we develop novel PCSK9 inhibitors, including small molecules, monoclonal antibodies, siRNAs, and other innovative therapies.
Leveraging cutting-edge multidisciplinary technologies, we rapidly and automatically screen millions of compound libraries via High-throughput Screening (HTS) to identify lead compounds that effectively inhibit PCSK9 activity or its interactions. Subsequently, Computer-aided Drug Design (CADD) techniques—such as molecular docking and pharmacophore modeling—optimize and modify these lead structures to enhance potency and selectivity. Crucially, during structural optimization, we concurrently perform ADMET Predictions (absorption, distribution, metabolism, excretion, toxicity) to early eliminate molecules with potential toxicity or adverse pharmacokinetics (PK). By comprehensively evaluating compounds' physicochemical properties, in vivo stability, and safety, we ensure selected candidates not only effectively target PCSK9 and exert anti-obesity effects but also possess strong potential for advancing into preclinical trials.
- Antibodies or Peptides
Beyond small-molecule drugs, we are actively developing high-affinity, highly specific therapeutic antibodies or optimized peptides. These biomolecular therapies precisely target PCSK9, aiming to achieve effective metabolic regulation and anti-obesity goals while minimizing off-target effects.
- siRNAs
siRNA-mediated suppression of PCSK9 gene expression represents a highly efficient and long-lasting gene silencing approach. It blocks PCSK9 protein synthesis at its source, offering a novel therapeutic strategy for obesity. However, siRNA molecules are prone to degradation in vivo and face challenges in efficiently penetrating target cells. We are also dedicated to developing novel nanoparticles (NPs) as smart delivery systems to encapsulate and protect siRNA. These nanoparticles, engineered with surface modifications and targeted ligands, are designed to significantly enhance the tissue-specific delivery efficiency of siRNA.
Preclinical Studies
Our expertise in preclinical research enables a comprehensive evaluation of anti-obesity therapies under development.

We systematically monitor the effects of anti-obesity therapies on body weight, body fat percentage, food intake, energy expenditure, and biomarkers. Through precise studies, we provide detailed PD data to support further development.
Our services encompass: evaluating ADME processes and determining concentration-time relationships across various tissues and body fluids. Through precise PK studies, we obtain critical data on candidate drugs' bioavailability, half-life, clearance, and steady-state plasma concentrations, providing a scientific basis for designing dosing regimens and optimizing dosages.

Our bioanalysis services form the cornerstone of pharmacokinetic and toxicological studies, delivering highly sensitive and specific quantitative analysis of drugs and their metabolites in biological samples. We employ advanced liquid chromatography-tandem mass spectrometry (LC-MS/MS) technology to develop and validate drug detection methods in biological samples such as plasma, urine, and tissues.

Our safety assessment services comprehensively identify and evaluate potential risks of PCSK9 inhibitors during the preclinical phase. Studies encompass acute toxicity, subchronic toxicity, genotoxicity research, etc., delivering comprehensive toxicological data and safety evaluation outcomes.
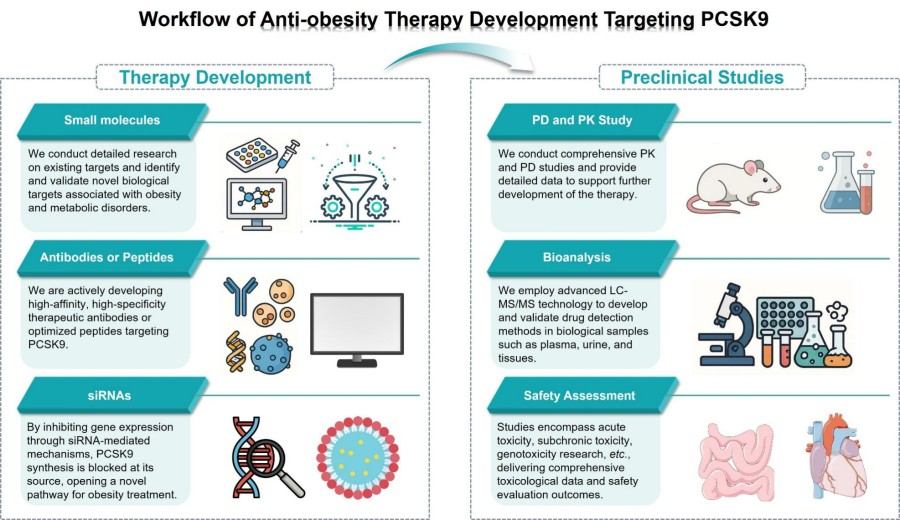
Workflow
Protheragen employs a rigorous, multidisciplinary anti-obesity therapeutic development process. By integrating high-throughput screening, computer-aided design, and integrated preclinical evaluation, we ensure maximum efficiency and quality at every stage.
Applications
Advantages
Deep Expertise
Leveraging years of experience focused on complex core metabolic targets, we guide PCSK9-targeting candidates from initial discovery through preclinical studies.
Diverse Therapeutic Development
We offer development services for multiple anti-obesity drug modalities—including small molecules, antibodies, peptides, and siRNA—providing comprehensive support for all research phases.
Preemptive Risk Mitigation
During lead optimization, we integrate ADMET prediction, specificity screening, and safety assessment. This strategy eliminates poorly druggable or potentially toxic molecules early, significantly reducing late-stage R&D risks and costs.
Beyond providing core PCSK9-targeted therapeutic development services, we are committed to empowering your obesity research with integrated, multidimensional testing and analytical solutions. Our goal is to delve into the complex biological mechanisms of obesity, delivering precise scientific insights and robust decision support.
Publication Data
Title: Deciphering the genetic landscape of obesity: a data-driven approach to identifying plausible causal genes and therapeutic targets
Journal: Journal of Human Genetics, 2023
DOI: https://doi.org/10.1038/s10038-023-01189-3
Summary: In this study, researchers utilized genome-wide association study (GWAS) datasets to screen and compile an extensive list of 28,787 obesity-associated single-nucleotide polymorphisms (SNPs). Subsequently, genes with significant computational simulation evidence were prioritized, ultimately refining a list of 74 key genes closely associated with obesity. These genes not only exhibit high mutual association but also show significant enrichment in multiple core biological processes contributing to obesity, particularly energy expenditure and metabolic homeostasis. Through in-depth drug-gene interaction analysis, researchers further identified 23 key genes with potential associations to existing therapies.
Given the pivotal role of hub genes in disease networks and their immense potential as drug development targets, the researchers further analyzed hub genes associated with metabolic indicators like LDL cholesterol (e.g., PCSK9). These hub genes represent not only attractive targets for novel drug development but also provide a robust scientific rationale for repurposing existing drugs to treat obesity.
This work integrates data from multiple cutting-edge knowledge repositories, providing clear potential therapeutic targets to address the challenge of target selection in obesity treatment.
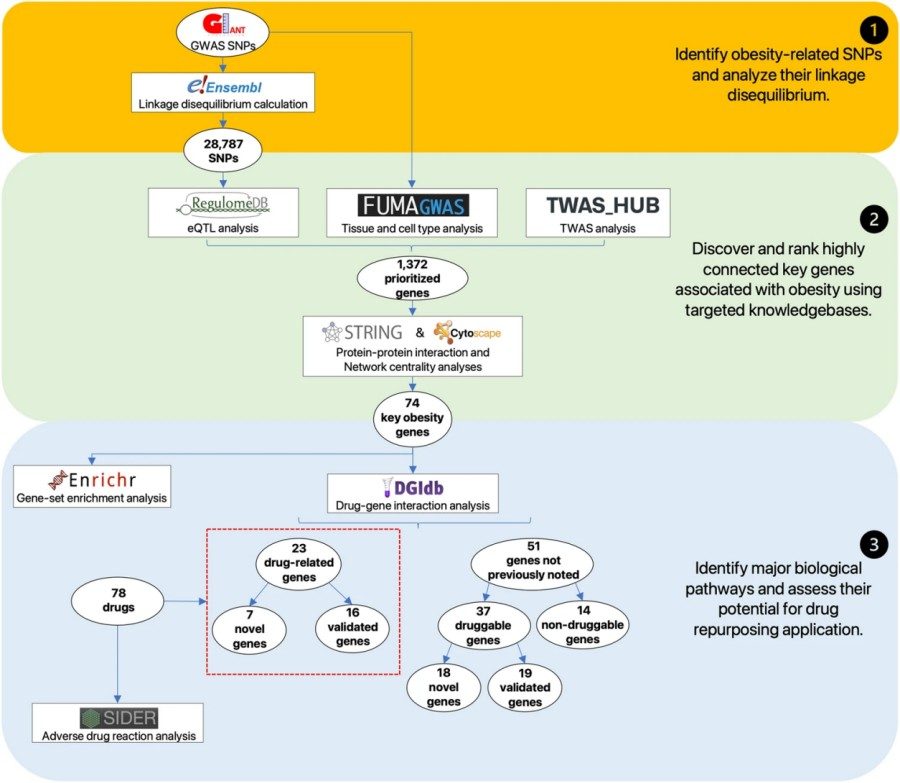 Fig. 1 Overview of the comprehensive data integration approach (Ang, et al., 2023)
Fig. 1 Overview of the comprehensive data integration approach (Ang, et al., 2023)
Customer Review
Precise Functional Screening
"Utilizing Protheragen's HTS service, we successfully identified PCSK9 inhibitor chemical scaffolds from their ultra-high-capacity compound library through precise functional screening. This significantly accelerated our PCSK9 inhibitor research timeline."— Dr. R*, Drug Discovery Senior Scientist
Comprehensive Lead Compound Evaluation
"Our collaboration with Protheragen was exceptionally smooth and efficient. Their evaluation during the lead optimization phase was remarkably thorough. This rigorous approach enabled us to eliminate several risky candidates early on, ultimately shortening the overall research timeline."— A. L., Director of Research
Frequently Asked Questions
-
How can drug specificity be ensured in the development of anti-obesity therapies?
We employ multiple technologies to guarantee high drug specificity:
- Structure-based design: For small molecules and peptides, we utilize high-resolution crystal structures for rational design, ensuring precise binding to the PCSK9 target.
- Differential screening: During lead optimization, we employ specificity screening techniques to exclude compounds with potential impacts on critical pathways, ensuring precise metabolic intervention.
-
How does our project interface with your development workflow?
We employ a systematic and efficient development process: from initial therapeutic design and high-throughput screening, through lead compound optimization, to comprehensive preclinical efficacy and safety evaluation. You may collaborate with us at any stage based on your project phase and requirements, and we will provide customized solutions.
Protheragen is the premier scientific partner for anti-obesity therapy development. Our unparalleled expertise, deep scientific insights, and proprietary technologies ensure the success of your leading candidates. Please contact us to discuss your PCSK9 program or receive a detailed scientific proposal for specific compounds.
Reference
- Ang, M.Y.; et al. Deciphering the genetic landscape of obesity: a data-driven approach to identifying plausible causal genes and therapeutic targets. Journal of Human Genetics. 2023, 68(12): 823-833. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

