Custom Gene Delivery Development
InquiryThe persistently high global obesity rate poses a severe challenge to public health. Given the inherent limitations of existing therapies, gene therapy has emerged as the next revolutionary approach to treating obesity. This therapy holds promise for achieving a one-time curative intervention, shifting from mere chronic symptom management to directly targeting the disease's genetic root cause. Its core lies in a deep understanding of the genetic and molecular pathways regulating energy homeostasis and fat storage. By utilizing efficient delivery systems to precisely target therapeutic genes to specific cells, it aims to restore and maintain metabolic balance. This approach not only avoids the limitations of targeting systemic hormonal pathways alone but also demonstrates immense potential in anti-obesity treatment.
Customized Vector Engineering Services to Accelerate Your Obesity Therapy Development
The foundation of gene therapy lies in developing effective delivery systems to transport genetic material to target cells. With years of deep expertise in obesity research, Protheragen offers diversified services for Anti-obesity Gene Therapy Development and gene delivery system design. We design, optimize, and manufacture a full range of gene delivery vectors, significantly advancing our clients' gene therapy development efforts.
Custom Viral Vector Development
Traditional gene delivery often faces challenges of low efficiency and transient expression. We utilize customized viral vectors (such as AAV and lentivirus) to cleverly bypass cellular defense mechanisms, efficiently delivering therapeutic DNA into the cell nucleus. This system offers the advantage of long-lasting, stable gene expression, accelerating the entire process from target discovery to functional validation.
Lentiviral vectors serve as powerful tools for developing anti-obesity gene therapies, particularly suited for studies requiring long-term stable gene expression. Our lentiviral vector development services focus on constructing customized vectors that are safe, efficient, and capable of integrating into the host cell genome. This ensures that delivered therapeutic DNA—such as genes encoding metabolic hormones, appetite regulators, or thermogenesis promoters—maintains sustained expression once introduced into target cells. We design and optimize vectors for targeted delivery to adipocytes, hepatocytes, hypothalamic neurons, and other cell types, providing high-titer, high-purity lentiviral particles as required. This system is ideal for studying gene function, in vivo mechanisms of action, and establishing stably expressing cell lines, thereby significantly advancing the development of obesity treatment strategies.
AAV vectors are the preferred tool due to their non-integrating nature and excellent in vivo safety profile. Our AAV vector development services are tailored for anti-obesity gene therapy, focusing on delivering highly efficient, low-immunogenicity gene delivery solutions. This encompasses customized vector engineering, including selection of optimal AAV serotypes and design of tissue-specific promoters to ensure therapeutic gene expression is confined to target organs. The final delivered vectors guarantee ultra-high titers and minimal empty capsid rates, maximizing effective doses while minimizing immunotoxicity. This accelerates the translation of your anti-obesity therapies from R&D to preclinical studies.
Custom Non-viral Gene Delivery System Development
Parallel to viral vectors, non-viral delivery methods are emerging as more promising and often safer alternatives. These systems offer multiple significant advantages: simpler and more cost-effective large-scale production, low host immunogenicity, and virtually no size restrictions for delivered therapeutic genes. We specialize in designing and optimizing delivery systems based on lipid nanoparticles (LNPs), polymeric nanoparticles, and other non-viral platforms. Unlike viral vectors, non-viral systems lack viral proteins, significantly reducing immune responses and potential toxicity risks. We tailor the particle size, surface charge, and ligand modifications of the delivery system based on the type of nucleic acid you wish to deliver (e.g., plasmid DNA, mRNA, or siRNA) and the target site (e.g., liver, adipose tissue). This ensures efficient cellular uptake and targeted delivery, enabling precise regulation of key pathways such as appetite, energy expenditure, or fat metabolism.
Custom Oligonucleotide and Nucleic Acid Synthesis
Any successful gene therapy relies on high-quality nucleic acid payloads. Protheragen provides comprehensive services for the custom synthesis and modification of these critical components. Our platform employs advanced chemical techniques and automated solid-phase synthesis to produce nucleic acids ranging from simple primers to complex therapeutic constructs. Services cover siRNA, miRNA, mRNA, and more. Each batch undergoes rigorous quality control to ensure high purity and exceptional batch-to-batch consistency.
Workflow
Protheragen specializes in the customized production of a full range of viral vectors for gene delivery. Our expertise spans multiple vector types, including lentivirus, AAV vectors, LNP, etc. We also meticulously design multi-step purification strategies tailored to diverse downstream applications, whether for research-grade materials or high-purity formulations for in vivo use.
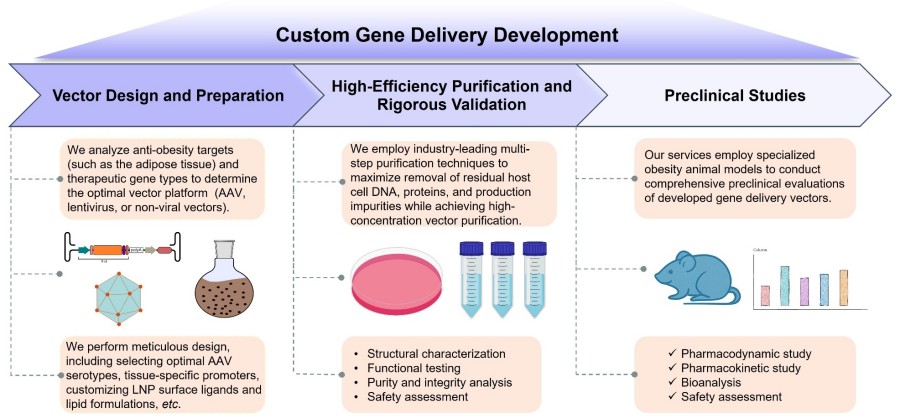
Vector Design and Preparation
We first analyze anti-obesity targets (such as the hypothalamus, adipose tissue, and liver) and therapeutic gene types to determine the optimal vector platform (AAV, lentivirus, or non-viral vectors). Relevant gene targets include those involved in circadian rhythm regulation, thermogenesis, lipolysis, adipocyte differentiation, food intake regulation, etc. Based on this, we perform meticulous design, including selecting optimal AAV serotypes, tissue-specific promoters, customizing LNP surface ligands and lipid formulations, etc. Following vector design completion, we utilize methods such as cell line transfection (for AAV/lentivirus) and microfluidic technology (for non-viral LNPs) to prepare high-quality vectors.
High-Efficiency Purification and Rigorous Validation
We employ industry-leading multi-step purification techniques, including ion exchange, affinity chromatography, ultrafiltration, etc., to maximize the removal of residual host cell DNA, proteins, and production impurities while achieving high-concentration vector purification. Subsequently, purified vectors undergo rigorous quality control (QC) and characterization, encompassing: structural characterization, functional testing, purity and integrity analysis, and safety assessment. These stringent steps ensure the delivered vector product exhibits high purity, high activity, and high biosafety, fully meeting the preclinical research standards.
Preclinical Studies
Our services employ specialized Obesity Models to conduct comprehensive preclinical evaluations of developed gene delivery vectors. This includes biodistribution testing and shedding analysis using obesity models; assessment of vector efficacy in improving obesity phenotypes, including body weight, body fat percentage, food intake, and energy expenditure; and detailed toxicological analysis.
Applications
Utilizing highly targeted gene delivery systems to introduce key genes regulating appetite, fat metabolism, etc., into specific tissues or cell populations, thereby precisely manipulating gene function to reveal the underlying molecular mechanisms of obesity development.
We deploy optimized gene vectors into animal models to evaluate in vivo efficacy, biodistribution, and safety of candidate gene therapies, accelerating your treatment's transition from laboratory to preclinical stages.
Our gene delivery system development services offer exceptional flexibility and broad applicability. We are dedicated to addressing the complex metabolic disorders triggered by obesity, delivering precise and enduring treatment solutions.
Advantages
Decades of Scientific Expertise
Our elite team of PhD researchers and technical experts possesses a systematic and meticulous understanding of the entire process of gene delivery vectors. We excel at anticipating and resolving complex technical challenges that impact delivery efficiency and safety.
Scalable Production & Custom Solutions
We deliver truly precision-tailored solutions. Built upon rigorous and highly scalable process designs, we adapt with maximum flexibility to the scale and technical requirements of projects at every stage.
Precision Targeting & Specificity Design
Unlike basic services that merely provide vector skeletons, we are dedicated to developing next-generation delivery technologies. Our core advantage lies in enabling precise drug accumulation at the target site, ensuring maximum efficacy of therapeutic molecules.
At Protheragen, our core value extends far beyond the simple manufacturing of gene delivery vectors. We focus on building highly integrated "delivery-analysis" closed-loop solutions to comprehensively advance your obesity research and development. By seamlessly integrating with advanced multidimensional technologies, we deeply dissect the complex biological networks underlying obesity.

Publication Data
Title: Innovative gene therapy strategies for tackling obesity
Journal: Egyptian Journal of Medical Human Genetics, 2025
DOI: https://doi.org/10.1186/s43042-025-00686-8
Summary: Gene therapy represents a promising solution for obesity by regulating gene expression to promote energy expenditure and lipolysis, aiming to reduce fat accumulation and restore the body's energy homeostasis. This study introduces the current gene delivery methods, including viral vectors (such as adeno-associated virus AAV) and non-viral vectors (such as lipid nanoparticles LNP and hydrodynamic delivery). Researchers have successfully delivered key genes like FGF21 using these vectors, observing significant amelioration of high-fat diet-induced obesity in animal models. While this therapy holds immense potential for improving metabolic regulation and quality of life, the safety and long-term stability of delivery systems require further investigation. With ongoing research and technological innovation, these challenges are expected to be overcome, ultimately translating gene therapy into a viable solution for treating obesity.
Customer Review
Excellent Targeting and High Efficiency
"The AAV vectors developed with Protheragen demonstrated impressive precision targeting. Their vector design team is highly professional. Efficient delivery significantly accelerated our project timeline."— V*, Sourcing Manager
Complex Customization
"Protheragen truly delivered exceptional expertise. Leveraging their specialized lipid chemistry and nanoparticle surface modification techniques, they successfully customized LNPs to meet our exact requirements, ensuring seamless progression of our subsequent experiments."— S*, Technical Program Manager
Frequently Asked Questions
-
If I have specific safety or targeting requirements for the vector, do you offer customized services?
Absolutely. Our services are highly customized. We modify the vector according to your specific needs to enhance targeting, thereby meeting complex safety and delivery requirements in your research.
-
After the vector is developed, can you support subsequent preclinical evaluation?
Yes, we offer a one-stop service. Following vector development, we seamlessly transition into subsequent preclinical research services, including biodistribution studies, shedding analysis, pharmacodynamic assessments, comprehensive immunogenicity and toxicology analyses, and more.
Protheragen masters both viral and non-viral vector technologies to create truly customized solutions for your therapeutic vision. Welcome to contact us to explore with our R&D and business development teams how our tailored gene delivery platforms can accelerate obesity therapy research.
Reference
- Sheikh-Hosseini, M.; et al. Innovative gene therapy strategies for tackling obesity. Egyptian Journal of Medical Human Genetics. 2025, 26(1): 58. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.



