Custom Viral Vector Development
InquiryOverview
Obesity and its associated metabolic disorders, such as type 2 diabetes, are emerging as an increasingly severe global health crisis. Traditional interventions face challenges in achieving sustained long-term efficacy. This limitation has paved the way for the emergence of a breakthrough new therapy—gene therapy. Leveraging efficient delivery systems (such as viral vectors), gene therapy holds promise for providing more fundamental and potentially curative solutions by precisely targeting the underlying genetic and metabolic drivers of disease.
Enabling Precision Delivery for Anti-obesity Gene Therapy Development
Customized viral vector development is key to unlocking the full potential of anti-obesity gene therapy. Protheragen specializes in designing, developing, and manufacturing customized viral vectors to meet the unique and complex demands of Anti-obesity Gene Therapy. This comprehensive service encompasses plasmid production, molecular and process development, upstream/downstream manufacturing, drug filling, and a full suite of in-house analytical testing and release services. Extensive development experience ensures each constructed vector is optimized for its specific therapeutic target.
Vector Design
Built upon a flexible proprietary platform, our customizable viral vectors include adeno-associated virus (AAV) and lentivirus.
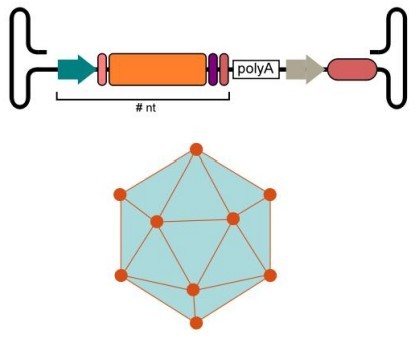
AAV serves as the cornerstone of in vivo gene therapy due to its exceptional safety profile. This vector is non-pathogenic and induces the lowest immune response compared to other viral vectors, making it ideal for achieving sustained, long-term gene expression within living tissues. We select appropriate serotypes and engineered variants to achieve precise targeted delivery.
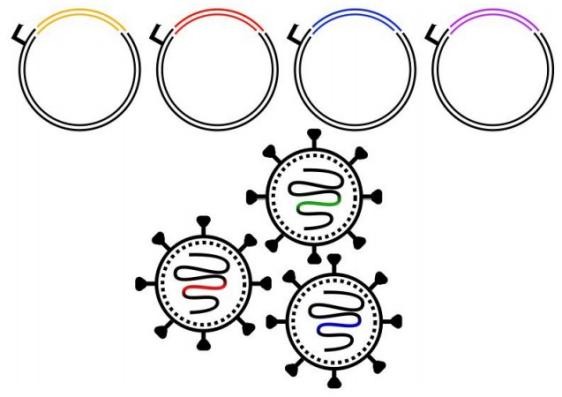
Lentiviral vectors are valued as versatile tools for delivering genetic material to diverse cell types, including both proliferating and non-proliferating cells. Their core advantage lies in the stable integration of therapeutic gene payloads into the host cell genome, enabling long-term stable expression. This characteristic makes them particularly suitable for ex vivo gene therapy applications. With a packaging capacity of up to 8 kb, lentivirus carries large genes or multiple gene cassettes, offering flexibility for designing complex therapeutic regimens.
Preclinical Studies
Through a series of processes including production, purification, and quality control, we obtain viral vectors with the highest purity, titer, and consistency. Subsequently, we evaluate the efficacy of these vectors in treating obesity using cells and obesity animal models, providing reliable support for therapeutic development.
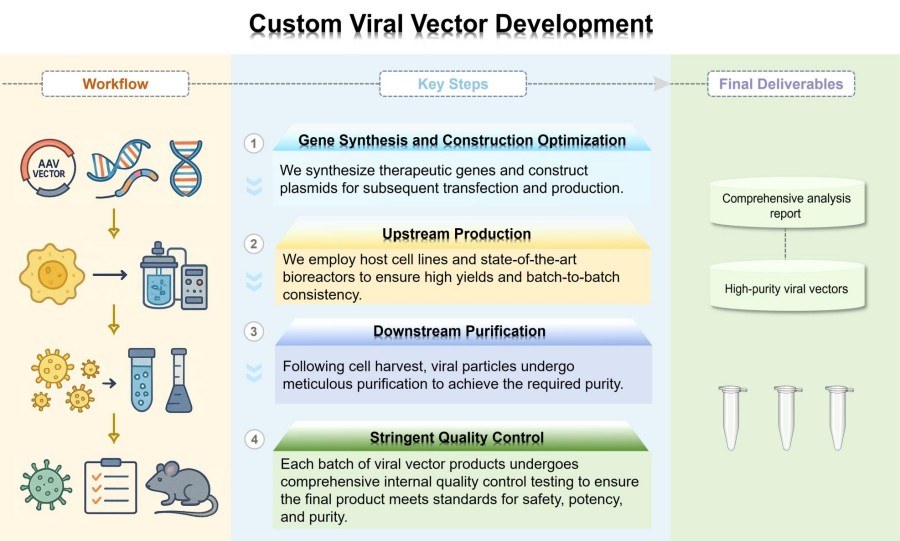
Workflow
Our services are built on scientific rigor, transparency, and operational efficiency, ensuring each phase is optimized for success while providing clients with real-time data and clear visibility.
Gene Synthesis and Construction Optimization
After defining research objectives, we screen for optimal viral vectors (AAV vector or lentiviral vector), designing vectors with careful consideration of promoters and other regulatory elements. Subsequently, we synthesize therapeutic genes and construct plasmids for subsequent transfection and production.
Upstream Production
Leveraging robust, scalable, and validated platforms, we employ host cell lines and state-of-the-art bioreactors to ensure high yields and batch-to-batch consistency.
Downstream Purification
Following cell harvest, viral particles undergo meticulous purification to achieve the required purity. We employ a combination of advanced techniques, including chromatographic purification, ultracentrifugation, etc., to remove cellular contaminants, residual plasmid DNA, and immunogenic proteins. This prevents toxicity to target cells or induction of immune responses in vivo.
Stringent Quality Control
Each batch of viral vector products undergoes comprehensive internal quality control testing to ensure the final product meets standards for safety, potency, and purity. Testing protocols include but are not limited to: titer determination (physical titer and infectivity titer), vector characterization, safety assessments (sterility, mycoplasma, endotoxin), and purity testing. Additionally, we utilize diverse obesity models for expression validation and efficacy evaluation.
Applications
Viral vectors can be utilized to deliver relevant genes, providing an efficient strategy for investigating pathological mechanisms such as metabolic balance, energy expenditure, and obesity.
The ability to precisely target specific tissues is a key advantage of viral vectors. Due to their low immunogenicity and capacity to drive long-term tissue-specific gene expression, they serve as critical tools for gene-targeted delivery and the development of effective anti-obesity therapies.
Beyond demonstrating therapeutic efficacy for obesity, viral vectors delivering metabolism-related genes may also offer therapeutic potential for other metabolic disorders.
Advantages
Unparalleled Scientific Expertise
With over 20 years of industry experience in obesity research, we possess a deep understanding of the complex biological and engineering challenges inherent in gene therapy. This expertise extends beyond foundational manufacturing to strategic scientific guidance in critical areas, including gene construct design, promoter selection, serotype screening, and delivery strategies.
Scalable and Flexible Production Platform
Engineered for efficiency and flexibility, our platform enables agile production workflows. This not only ensures rapid project advancement but also guarantees that the final delivered viral vectors exhibit industry-leading high titers and exceptional purity, meeting the most demanding research requirements.
Integrated End-to-End Services
We are committed to delivering a truly one-stop, end-to-end solution, comprehensively covering every critical step from high-quality vector manufacturing to rigorous in-house analysis and quality control. This ensures professionalism and reliability throughout the entire project lifecycle.
Our mission is to streamline obesity research by offering a complete, end-to-end solution. We guide your project from foundational studies to practical applications, with services that include pathological testing, microbiome analysis, risk prediction, and the creation of evidence-based weight management plans. We're here to help you fully grasp the multifaceted nature of obesity.

Publication Data
Title: Innovative gene therapy strategies for tackling obesity
Journal: Egyptian Journal of Medical Human Genetics, 2025
DOI: https://doi.org/10.1186/s43042-025-00686-8
Summary: This paper introduces obesity gene therapy, novel carriers, and delivery systems. Three methods regulate gene expression: viral vectors, non-viral vectors, and physical methods. Multiple carrier systems have been developed for gene delivery. Viruses can transfer their genes into host cells, making them powerful tools for gene transfer. Hydrodynamic delivery offers an efficient and straightforward alternative for intracellular DNA transfer in vivo. Ongoing research and technological innovation will yield more effective delivery systems to overcome existing challenges, ultimately translating gene therapy into a viable solution for treating obesity.
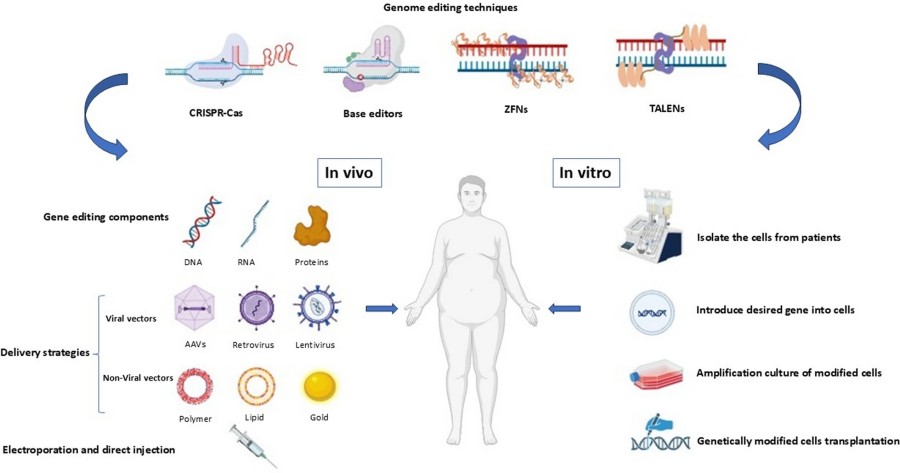 Fig. 1 Gene editing strategies for obesity treatment (Sheikh-Hosseini, et al., 2025)
Fig. 1 Gene editing strategies for obesity treatment (Sheikh-Hosseini, et al., 2025)
Customer Review
Satisfactory Scientific Collaboration
"Our partnership with Protheragen has been pivotal in advancing our anti-obesity therapy program. They resolved the instability and scalability limitations in our viral vector production, delivering AAV vectors capable of targeting adipose tissue, enabling us to progress smoothly to the next experimental phase."— E. M., Development Head
An Indispensable Partner
"Thanks to Protheragen's expert customization services, we obtained viral vectors with high purity, high titer, and exceptional expression efficiency. Your team not only strictly adhered to our design specifications but also provided invaluable advice that helped us optimize the vector architecture. We highly recommend this viral vector customization service."— Dr. S*, Laboratory Operations Director
Frequently Asked Questions
-
How do you assist us in selecting the appropriate viral vector for our project?
The vector selection process is collaborative. Protheragen's team of scientists works closely with each client to evaluate project-specific factors, including the size of the gene insertion fragment, desired expression duration, target tissues, and required safety features. This helps select the most suitable vector—such as AAV for long-term non-integrating expression or lentivirus for stable genomic integration—to align with the project's scientific objectives and requirements.
-
What is the minimum project size you can accommodate?
Protheragen offers flexible production scales to meet diverse needs. Our services range from small research-scale batches for proof-of-concept studies to large-scale manufacturing, ensuring every project receives the same level of expertise and quality assurance regardless of size.
Protheragen, a leading partner in custom viral vector development, offers unparalleled scientific expertise, scalable production platforms, and comprehensive analytical services tailored specifically for anti-obesity gene therapies. This end-to-end solution addresses critical industry challenges from meticulous scientific design to production support, paving a robust and efficient pathway for anti-obesity research. Please contact us to learn more, discuss your project, and obtain customized solutions that meet your specific needs and timelines.
Reference
- Sheikh-Hosseini, M.; et al. Innovative gene therapy strategies for tackling obesity. Egyptian Journal of Medical Human Genetics. 2025, 26(1): 58. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.