Adeno-associated Viru (AAV) Vector Development
InquiryOverview
Obesity is a complex, multifactorial disease with global implications. Recent advances in gene therapy, particularly the application of adeno-associated virus (AAV) vectors, have opened promising avenues for developing novel, long-lasting anti-obesity therapies. As a non-pathogenic, replication-deficient virus, AAV is renowned for its efficient delivery of therapeutic genes to diverse cell types with low immunogenicity. Its stable circular plasmid expression characteristics make it an ideal tool for long-term regulation of metabolic pathways.
AAV Innovation: Accelerating Anti-Obesity Gene Therapy Development
Protheragen provides a high-quality AAV vector development service designed to accelerate the advancement of Gene Therapies for obesity. Leveraging cutting-edge technology and deep expertise in metabolic biology, we deliver comprehensive end-to-end solutions. By utilizing AAV to Deliver Genes regulating appetite, energy expenditure, and fat metabolism, we enable the development of next-generation therapies with the potential for sustained weight management.
Our Vector Development platform integrates multiple core technologies to ensure the highest quality and therapeutic efficacy. We focus on optimizing every step, from vector design to final product purification.
Target Identification and Validation
Before vector design, our expert team identifies and validates novel therapeutic targets for obesity. Utilizing advanced bioinformatics and cellular models, we screen potential genes and pathways involved in metabolic regulation. This critical step ensures the genes delivered by your selected AAV vector possess therapeutic development potential, saving significant time and resources in subsequent stages.
Design to Production
- Advanced Vector Design
We utilize proprietary platforms to engineer AAV vectors with enhanced tissue specificity and superior transduction efficiency. These vectors are customized for specific insert sizes and expression requirements. Our platform encompasses multiple AAV serotypes and engineered variants for precise targeting of key tissues and cell types involved in metabolic regulation. We also provide tissue-specific promoters to drive and enhance gene expression in target cells. These promoters are suitable for various species, including mice, rats, etc., ensuring versatility across different experimental settings.
- High-Yield Production
We utilize optimized cell lines combined with advanced manufacturing processes and meticulous purification workflows to achieve high-titer AAV production with low empty capsid rates. Comprehensive testing for content, physicochemical properties, impurities, and potency ensures high quality to meet preclinical research requirements.
Preclinical Studies
Utilizing diverse obesity models, we administer vector injections and monitor key indicators to comprehensively assess AAV vector efficacy. This encompasses gene expression and function, phenotypic improvement, AAV biodistribution, shedding detection, etc., to accelerate preclinical development.
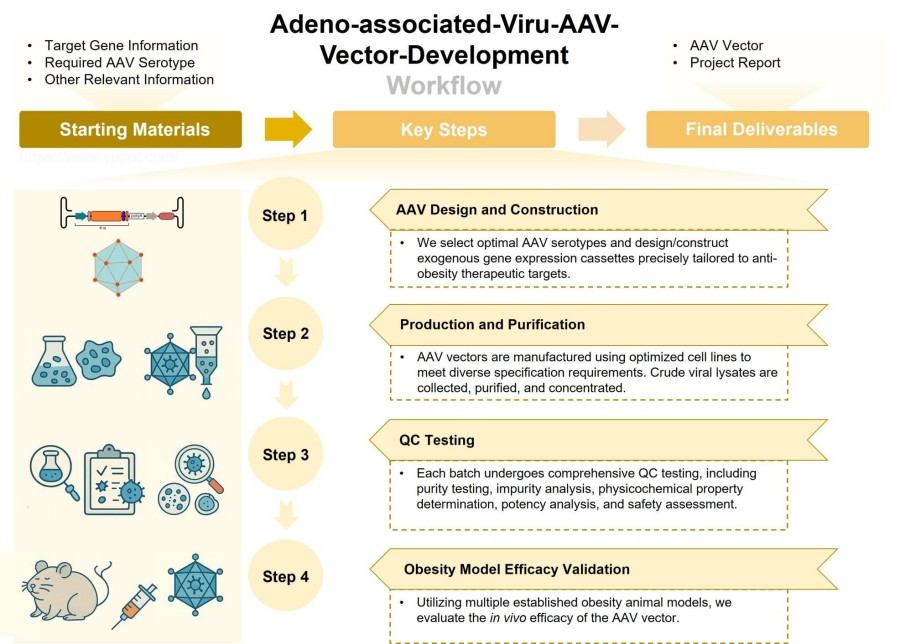
Workflow
Our meticulously refined AAV vector development workflow enables efficient project turnaround and end-to-end operational transparency. This systematic process serves as your project guide, precisely and smoothly steering your R&D initiatives from initial concept design through to the final delivery of ready-to-use, high-quality vectors.
AAV Design and Construction
We select optimal AAV serotypes and design/construct exogenous gene expression cassettes precisely tailored to anti-obesity therapeutic targets.
Production and Purification
AAV vectors are manufactured using optimized cell lines to meet diverse specification requirements. Crude viral lysates are collected, purified, and concentrated via advanced techniques like affinity chromatography to remove impurities.
QC Testing
Each batch undergoes comprehensive QC testing, including purity testing, impurity analysis, physicochemical property determination, potency analysis, and safety assessment.
Obesity Model Efficacy Validation
Following vector development and quality control, we provide the critical step of internal vector efficacy validation. Utilizing multiple established obesity animal models, we evaluate the in vivo efficacy of the AAV vector.
Applications
Leveraging AAV's precise and sustained delivery of therapeutic genes, we develop high-quality AAV vectors as powerful tools for creating novel anti-obesity therapies.
AAV vectors may also be applicable for developing therapeutic approaches targeting various obesity-related metabolic disorders, such as type 2 diabetes and non-alcoholic steatohepatitis.
Given obesity's strong association with cardiovascular disease, AAV vectors are being utilized to deliver genes that improve vascular function or reduce chronic inflammation, thereby developing treatments for complications like hypertension and atherosclerosis.
Advantages
Unparalleled Expertise
Our team possesses a deep and nuanced understanding of AAV biology and its application in obesity research. We are committed to your success, leveraging robust technical platforms and unmatched expertise to help you overcome challenges in gene therapy R&D.
High-Efficiency Production Capabilities
Our meticulously refined AAV production platform ensures rapid project delivery while offering highly competitive vector manufacturing services.
Extensive Practical Experience
Comprehensive experimental data robustly demonstrate that our AAV vectors exhibit exceptional purity and the highest titer of activity. These high-performance vectors have been validated across multiple in vivo models, consistently delivering stable and superior gene delivery and expression efficiency.
At Protheragen, our core value extends far beyond the simple manufacturing of gene delivery vectors. We focus on building highly integrated "delivery-analysis" closed-loop solutions to comprehensively advance your obesity research and development. By seamlessly integrating with advanced multidimensional technologies, we deeply dissect the complex biological networks underlying obesity.

Publication Data
Title: AAV-mediated gene therapy as a strategy to fight obesity and metabolic diseases
Journal: EMBO Molecular Medicine, 2018
DOI: https://doi.org/10.15252/emmm.201708791
Summary: This study introduces a potential AAV-mediated gene therapy for obesity. Researchers engineered an AAV vector carrying the FGF21 gene (AAV-FGF21) to mediate sustained long-term production of FGF21 protein in animal models. The vector was injected into obese mice to evaluate its therapeutic potential against obesity. Findings revealed that a single injection maintained FGF21 levels for up to one year, accompanied by reductions in body weight, adipocyte size, and validated fat mass. Additionally, energy expenditure and glucose homeostasis improved, demonstrating favorable anti-obesity effects. This study validates the therapeutic efficacy of AAV-mediated FGF21 gene therapy and provides data supporting the application of AAV-mediated gene therapy in obesity treatment.
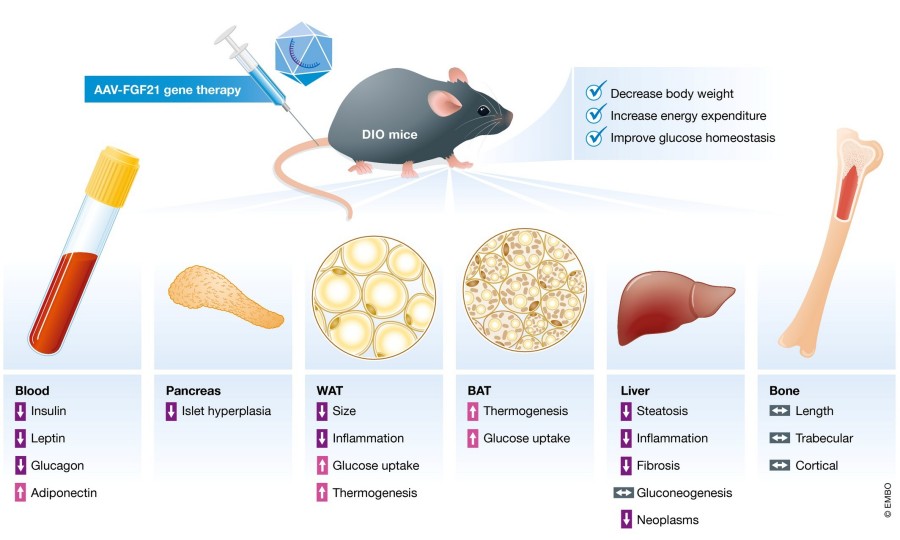 Fig.1 AAV-FGF21 gene therapy improves obese mouse traits. (Sponton & Kajimura, 2018)
Fig.1 AAV-FGF21 gene therapy improves obese mouse traits. (Sponton & Kajimura, 2018)
Customer Review
Trusted Partner
"Protheragen's high-purity AAV vectors form the cornerstone of our in vivo experimental success. More importantly, throughout the development process, their technical support team has not only been responsive but also provided invaluable guidance on biodistribution analysis. This end-to-end professional collaboration makes them our most trusted partner."— D. C., Scientific Associate
Specific Promoter Design
"The Protheragen team's expertise in complex promoter design is truly impressive. They successfully engineered a highly precise liver-specific promoter, strictly confining therapeutic gene expression to the target organ. This perfectly addressed our critical concern regarding off-target toxicity."— A. Z., Innovation Project Lead
Frequently Asked Questions
-
What advantages does AAV offer compared to other viral vectors for anti-obesity therapies?
AAV is the preferred choice due to its exceptional safety profile, low immunogenicity, and long-lasting gene expression characteristics. Unlike retroviruses or lentiviruses, AAV vectors typically exist as circular DNA and do not integrate into the host genome, thereby minimizing the risk of insertion mutagenesis.
-
Can you assist in selecting the optimal AAV serotype for target tissues?
Certainly. Our experts are well-versed in AAV tropism mechanisms and can guide you in selecting the most effective serotype for precise delivery of therapeutic payloads to target cell types.
-
How do you ensure the quality of AAV vectors?
We employ a rigorous multi-stage quality control process, including titer determination, purity analysis (e.g., sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), high-performance liquid chromatography (HPLC)), sterility testing, physicochemical property determination, and endotoxin testing. All operations are conducted in GMP-compliant facilities.
At Protheragen, we are dedicated to helping you transform scientific discoveries into breakthrough therapies. With a rigorous scientific approach and exceptional service, we serve as your trusted partner in AAV vector development. Please contact us for more information and to discuss your project requirements.
Reference
- Sponton, C.H.; Kajimura, S. AAV-mediated gene therapy as a strategy to fight obesity and metabolic diseases. EMBO molecular medicine. 2018, 10(8): e9431. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.