Anti-Obesity Drug Pharmacogenomics Service
InquiryThe global rise in obesity-related metabolic disorders has spurred the development of next-generation therapeutics, most notably GLP-1 and GIP receptor agonists. However, preclinical drug development often faces significant hurdles due to the high variability in drug response observed across different animal models and metabolic phenotypes. At Protheragen, we bridge this gap through our anti-obesity drug pharmacogenomics (PGx) service. By integrating genomic profiling into the preclinical phase, we empower researchers to identify genetic markers that dictate drug efficacy, metabolism, and potential safety risks before moving into clinical stages.
Preclinical Anti-Obesity Drug Pharmacogenomics Services
Our service focuses on the biological complexity of weight regulation, utilizing advanced multi-omics and genetic stratification to analyze how specific variations—such as those in GLP1R, ARRB1, MC4R, and FTO genes—influence the response to anti-obesity medications (AOMs). By partnering with Protheragen, drug developers can optimize lead candidate selection, refine dosing strategies in animal models, and maximize the translational success of their obesity pipeline.
Core Technologies
Protheragen leverages a suite of high-end technologies designed to uncover the molecular drivers of drug response:
- Next-Generation Sequencing (NGS) & High-Throughput Genotyping
We utilize specialized panels to identify single-nucleotide polymorphisms (SNPs) and copy number variations (CNVs) in genes associated with satiety signaling, lipid metabolism, and thermogenesis.
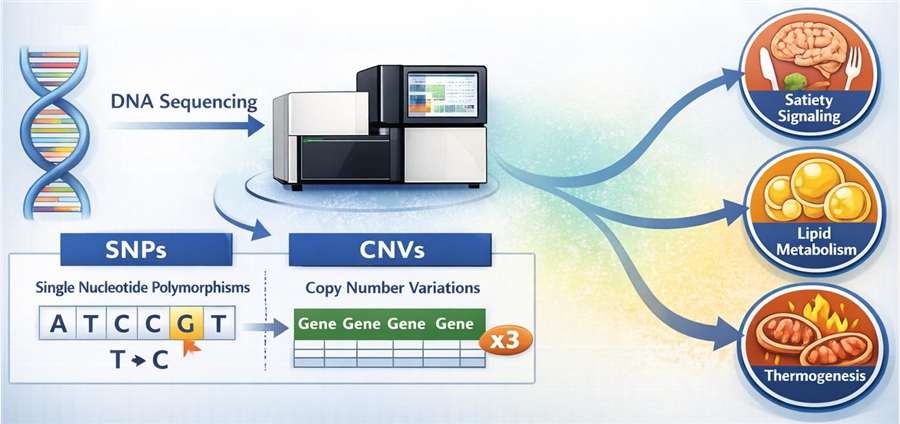
- Transcriptomic Profiling
Our platform analyzes mRNA expression in key metabolic tissues—including the hypothalamus, liver, and adipose tissue—to evaluate how drug candidates modulate gene pathways in response to specific genetic backgrounds.
- Targeted Pathway Analysis
We focus on the pharmacodynamics of centrally and peripherally acting pathways, specifically examining the G-protein coupled receptor (GPCR) signaling efficacy and the recruitment of beta-arrestin (e.g., ARRB1 variants).
- AI-Driven Predictive Modeling
By integrating polygenic risk scores (PRS) with metabolic phenotyping, our bioinformatics team predicts the probability of response ("responders" vs. "non-responders") for novel Small Molecules and biologics.
Solution Scope
Protheragen provides a broad range of pharmacogenomic services tailored specifically for obesity research:
- Marker Identification for GLP-1/GIP Agonists
Analysis of variants like GLP1R (rs6923761) and ARRB1 to predict weight loss magnitude and glycemic response.
- Metabolic Phenotyping Support
Utilizing genetic scores to classify models into "hungry brain" (satiety-deficient) or "hungry gut" (gastric emptying-related) phenotypes.
- Adverse Effect Prediction
Screening for genetic markers associated with common AOM side effects, such as gastrointestinal distress or muscle mass loss, to improve lead optimization.
Investigating variations in drug-metabolizing enzymes and transporters that may alter the bioavailability of Small-Molecule Anti-Obesity Agents.
Explore Our Full Service Scope.
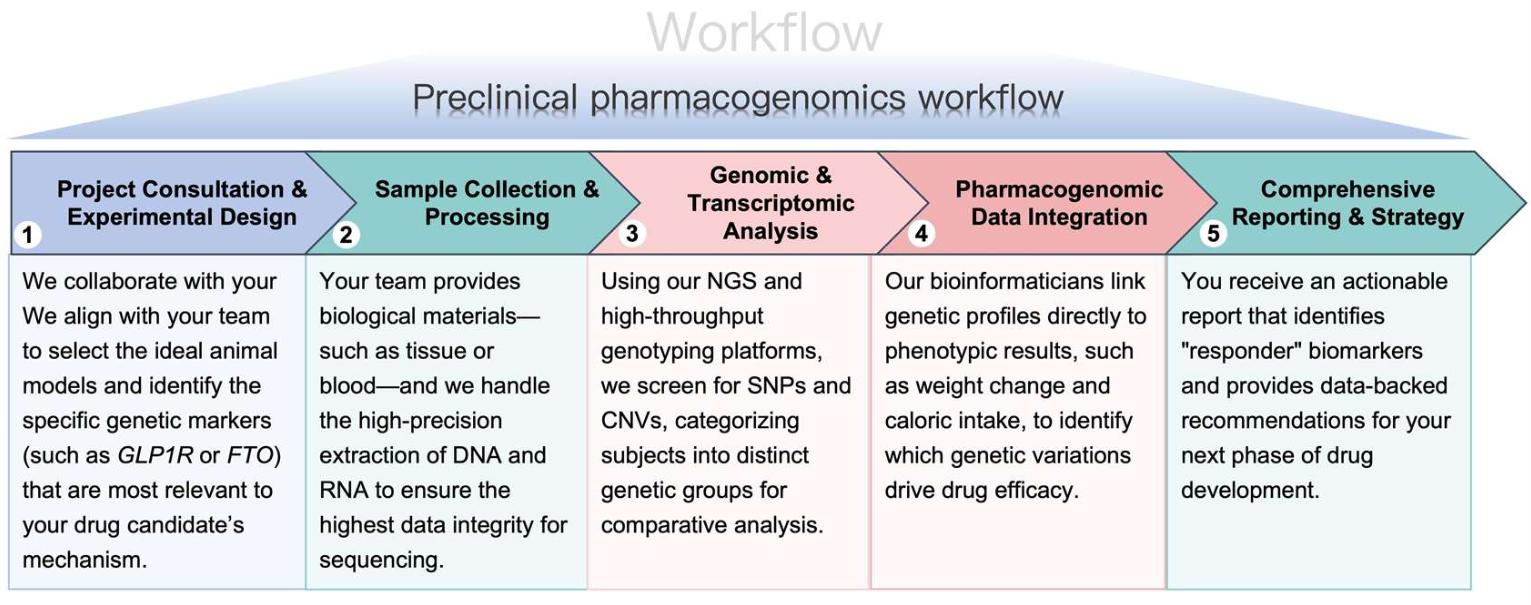
Workflow
Our preclinical pharmacogenomics workflow is designed to provide seamless integration into your existing R&D pipeline.
Fields of Application
The pharmacogenomics services provided by Protheragen are essential for developers looking to navigate the biological complexities of metabolic regulation across diverse therapeutic contexts.
- Novel Drug Discovery: Validating new targets within the leptin-melanocortin pathway or thermogenic adipocytes.
- Repurposing Studies: Evaluating how existing metabolic drugs perform when stratified by specific genetic risk scores.
- Combination Therapy Development: Assessing the synergistic effects of dual or triple agonists across diverse genetic backgrounds.
- Bioequivalence & Biosimilar Research: Ensuring that biosimilar candidates maintain consistent efficacy profiles across varying genotypes.
Advantages
Choosing Protheragen for your preclinical pharmacogenomics needs offers distinct competitive edges:
Enhanced Translational Precision
By identifying responders at the preclinical stage, you can significantly reduce the risk of late-stage attrition. Published data indicate that incorporating genetic stratification can increase the predictability of weight loss outcomes by up to 30%.
Optimized Resource Allocation
Avoid wasting resources on non-responsive models. Our services allow you to "fail fast" or pivot to more promising genetic subgroups early in development.
Mechanism-Specific Insights
Unlike generic genomic services, Protheragen specializes in metabolic pathways, providing deep expertise in how genetic variations affect receptor signaling and neuropeptide expression.
Data-Driven Dosing
Our insights help establish more accurate dose-response relationships by accounting for genetic variability in drug metabolism and target sensitivity.
Publication Data
Title: Pharmacotherapy for obesity: are we ready to select, tailor and combine pharmacotherapy to achieve more ambitious goals?
Journal: Front. Endocrinol., 2025
DOI: https://doi.org/10.3389/fendo.2025.1569468
Summary: Obesity is a complex, chronic disease influenced by a dynamic interplay of genetic, physiological, and environmental factors that disrupt energy homeostasis. While lifestyle modifications remain the foundational treatment, achieving and sustaining long-term weight loss is often challenging for many patients. Recent advancements have introduced a new generation of anti-obesity medications (AOMs), such as GLP-1 receptor agonists (e.g., semaglutide) and dual agonists (e.g., tirzepatide), which have demonstrated unprecedented weight loss efficacy in clinical trials. However, treatment outcomes remain highly variable, with some patients failing to respond to standard therapies. This review highlights the critical need for a precision medicine approach, where pharmacotherapy is tailored to an individual’s genetic markers, comorbidities, and specific obesity phenotypes (e.g., "hungry brain" vs. "hungry gut") to optimize treatment success.
Key Findings
- Unprecedented Efficacy of New Agents: Modern AOMs like semaglutide (14.9% weight loss) and tirzepatide (20.9% weight loss) have achieved double-digit weight reductions previously only seen with surgical interventions.
- Predictors of Success: An early response to treatment—defined as losing 3-5% of body weight within the first three to six months—is a strong predictor of long-term weight loss success.
- The Impact of Type 2 Diabetes: Patients with both obesity and type 2 diabetes often face greater challenges in weight loss. However, newer dual agonists like tirzepatide have shown significant promise for this population, reaching weight reductions of up to 14.7%.
- Pharmacogenomics & Genetic Markers: Genetic variations in the GLP-1 receptor (e.g., rs6923761) and ARRB1 may influence how an individual responds to specific weight-loss drugs. While clinical application is still emerging, these markers could eventually guide personalized drug selection.
- Phenotype-Driven Treatment: Classifying obesity into four subtypes—hungry brain (abnormal satiation), emotional hunger (hedonic eating), hungry gut (abnormal satiety), and slow burn (low metabolic rate)—allows for more effective, targeted therapy.
- Cardiovascular & Comorbidity Benefits: Beyond weight loss, AOMs like semaglutide have been proven to reduce the risk of major adverse cardiovascular events (MACE) by 20% in patients with obesity, independent of their effect on blood sugar.
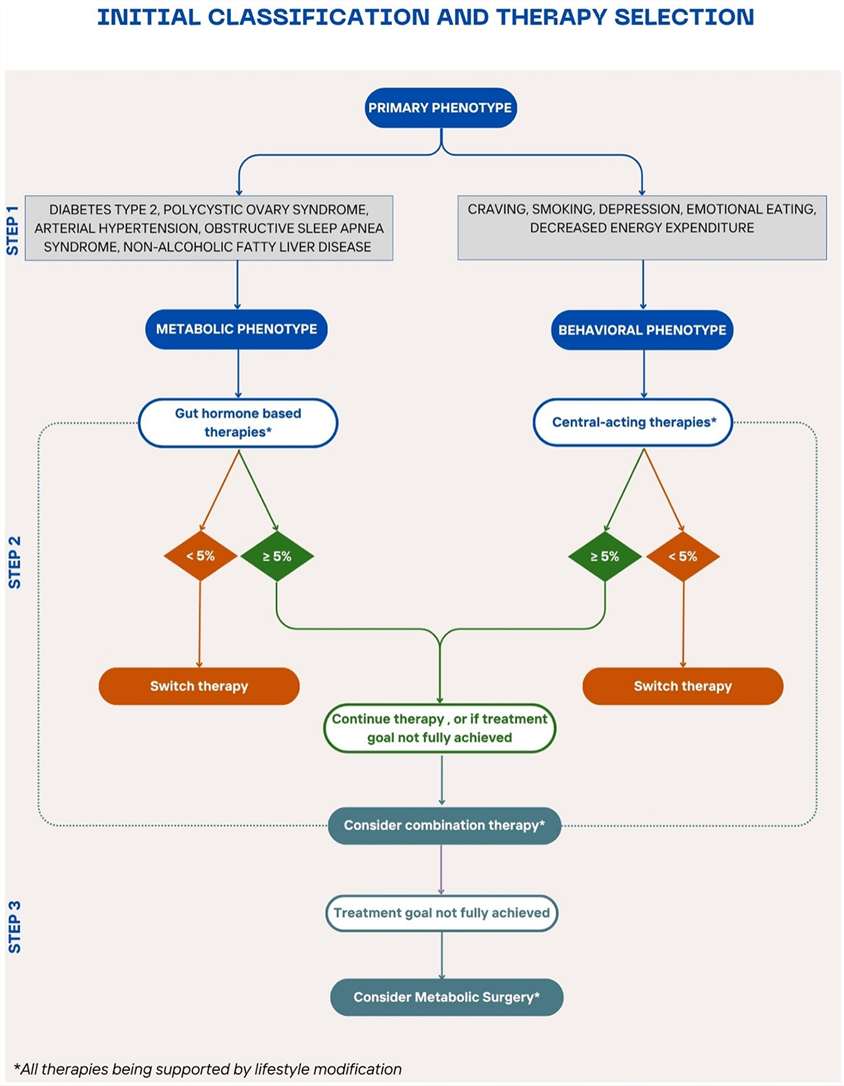 Fig.1 Stepwise algorithm for personalized obesity pharmacotherapy. (Steenackers, et al., 2025)
Fig.1 Stepwise algorithm for personalized obesity pharmacotherapy. (Steenackers, et al., 2025)
Customer Review
Precision Stratification for Enhanced Efficacy
"Working with Protheragen has transformed how we approach our dual-agonist program. Their ability to stratify our preclinical models based on GLP1R and ARRB1 variants allowed us to identify a specific sub-population where our lead candidate showed a 25% increase in efficacy. Their expertise in the biology of weight loss is unparalleled."
Principal Scientist, Biotech Innovation Group
Actionable Data for Crucial Development Milestones
"The team at Protheragen provided us with the clear, data-driven insights we needed to justify moving our AOM candidate into the next phase of development. Their reporting is incredibly detailed, bridging the gap between raw genomic data and actionable R&D strategy. We look forward to our continued collaboration on our upcoming triple-agonist study."
Director of Metabolic Research, Global Pharma Corp
Frequently Asked Questions
-
Does Protheragen provide clinical diagnostic testing for patients?
No, our services are strictly for preclinical research and drug development purposes only. We do not provide clinical services or diagnostic results for human patients.
-
What types of animal models are compatible with your PGx service?
We support a wide range of models, including standard inbred mouse/rat strains, DIO models, and custom transgenic or knockout lines.
-
Can you identify markers for non-GLP-1 pathways?
Yes, we analyze markers across the entire metabolic spectrum, including MC4R agonists, mitochondrial uncouplers, and amylin analogues.
-
How long does a typical preclinical PGx project take?
Timelines vary based on scope, but standard genotyping and correlation analysis usually take 4-6 weeks from sample receipt.
-
Can this service help predict gastrointestinal side effects?
While side effects are multifactorial, we can screen for genetic predispositions in pathways associated with gastric motility and nausea.
-
Is your technology compatible with small molecules and biologics?
Yes, our platform is designed to handle the pharmacogenomics of both small-molecule inhibitors and large protein-based agonists.
-
How do you ensure data security?
We maintain strict data encryption and confidentiality protocols to protect your proprietary drug discovery information.
-
How can I get a quote for my specific project?
Simply reach out through our inquiry portal with your project details, and our specialists will provide a customized proposal.
How to Contact Us
Protheragen is committed to advancing the frontier of anti-obesity drug development through precision pharmacogenomics. By identifying the right targets for the right models, we help you bring more effective and safer therapies to the market.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Steenackers, N.; et al. Pharmacotherapy for obesity: are we ready to select, tailor and combine pharmacotherapy to achieve more ambitious goals? Front. Endocrinol. 2025, 16:1569468. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

