Anti-Obesity Therapeutic Local Toxicity Study Service
InquiryAs the global prevalence of obesity reaches pandemic proportions, the development of targeted anti-obesity therapeutics (AOTs) has shifted toward innovative delivery mechanisms, including subcutaneous injectables, transdermal microneedle patches, and site-specific metabolic modulators. Protheragen provides specialized preclinical local toxicity study services designed to evaluate the safety profile of these agents at the site of administration.
Advanced Local Tolerance Assessment for Metabolic Drug Candidates
Unlike systemic toxicity assessments, local toxicity studies focus on the morphological and functional integrity of tissues directly exposed to the therapeutic—such as skin, muscle, or adipose tissue—ensuring that potent metabolic efficacy does not come at the cost of injection site reactions, inflammation, or tissue necrosis.
Core Technologies
Protheragen integrates advanced histological imaging with molecular bioanalysis to provide a high-resolution view of tissue-drug interactions.
- High-Definition Histopathology
Our platform utilizes high-definition histopathology to detect micro-abscesses or subtle inflammatory infiltrates that standard screenings might miss.
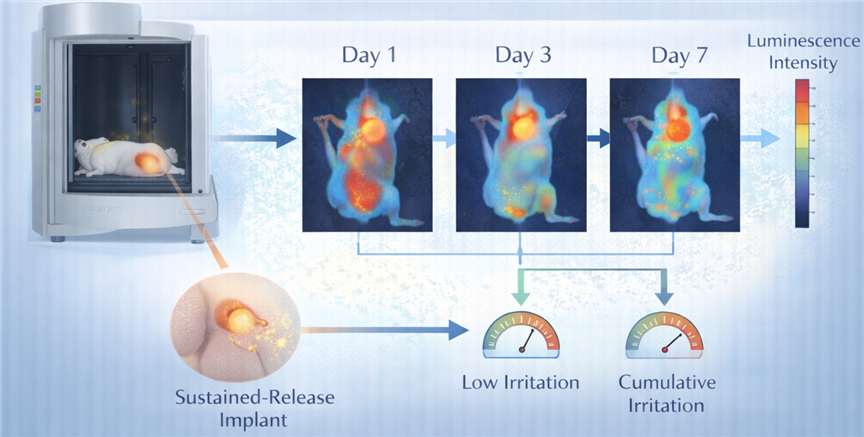
- In Vivo Bio-Imaging Systems (IVIS)
We employ IVIS to track the residence time of localized treatments, ensuring that sustained-release formulations do not cause cumulative irritation.
- Molecular Biomarker Profiling
Our molecular biomarker profiling assesses local cytokine expression (such as IL-6 and TNF-α) to quantify the sub-clinical inflammatory response, providing a data-driven safety margin for transition into later developmental stages.
Solution Scope
Our local toxicity study service is comprehensive, covering a wide array of AOT modalities:
- Injectable Formulations
Assessment of site reactions for GLP-1 Receptor Agonists and dual/triple co-agonists.
- Transdermal Systems
Testing of microneedle patches and topical gels for skin irritation and sensitization.
- Implantable Delivery Devices
Evaluation of the foreign body response and local tissue compatibility for long-term release pumps.
- Adipose-Targeting Agents
Specific toxicity screens for drugs designed to induce browning of white adipose tissue (WAT) or localized lipolysis.
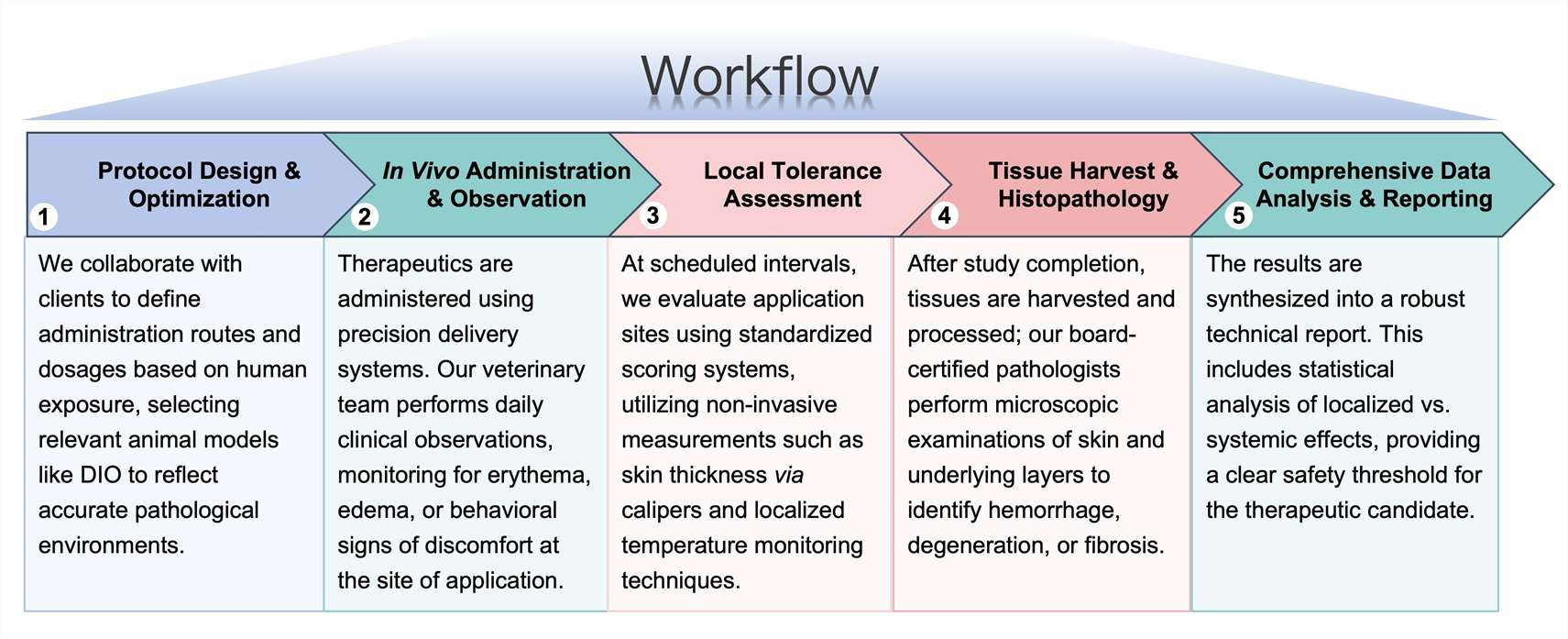
Workflow
To ensure the most precise and data-driven evaluation of your lead anti-obesity candidate, our workflow integrates a high-resolution scientific framework designed to map the exact biological impact of localized drug delivery.
Click Here to Submit an Inquiry
Fields of Application
The data generated through our services is vital for:
- Lead Optimization: Comparing multiple formulations to select the one with the highest local tolerance.
- Formulation Bridging: Assessing if changes in excipients or delivery concentrations impact tissue compatibility.
- Mechanism of Action (MoA) Safety: Confirming that locally acting lipolytic agents do not cause collateral damage to the surrounding vascular or nervous systems.
- Clinical Readiness Support: Providing the definitive local safety data packages necessary to validate a lead candidate's transition from the laboratory into human-facing development phases.
Advantages
Protheragen provides a distinct competitive edge in the preclinical landscape by combining specialized metabolic expertise with high-resolution analytical tools. Our platform is engineered to transform raw safety data into actionable insights for your development pipeline.
Metabolic Context Precision and Validated Predictability
Unlike generic safety screens, we utilize specialized diet-induced obesity (DIO) models to ensure that local toxicity is assessed within the specific physiological environment of the target patient population. According to published data, our integrated local tolerance metrics demonstrate a high correlation with successful transitions into human-facing development phases, significantly de-risking your lead candidate.
High-Resolution Tissue Mapping and Bio-Molecular Insight
We employ advanced micro-dissection and digital pathology to evaluate the integrity of the epidermis, dermis, and underlying adipose layers at a cellular level, identifying sub-clinical irritation that standard assessments might overlook. We go beyond visual scoring by profiling local inflammatory biomarkers and cytokines, providing a molecular-level confirmation of tissue compatibility.
Longitudinal Residence Tracking and Accelerated Decision Timelines
Utilizing our IVIS, we provide quantitative data on the residence time of sustained-release formulations, ensuring that efficacy does not lead to cumulative local tissue stress. Our streamlined laboratory operations are optimized for rapid data generation, allowing your team to make critical "go/no-go" decisions without compromising on scientific rigor.
Inquire Now to Discuss Your Study Design with Our Experts.
Customer Review
Identifying Hidden Risks in Next-Gen Therapeutics
"Working with Protheragen was a turning point for our peptide program. Their expertise in local tissue response allowed us to identify a formulation issue that systemic screening had missed. The histopathology reports were incredibly detailed, providing us with the confidence to move forward into the next phase of development. Their team feels like an extension of our own R&D department."
Senior Director of Pharmacology, Global Pharmaceutical Lead
From Microphysiological Data to Strategic Confidence
"The precision Protheragen brought to our microneedle patch study was exceptional. They provided not only the safety data we needed for our filing but also insights into the skin's recovery kinetics that were vital for our delivery device design. We plan to utilize their local toxicity services for all our future transdermal candidates."
Lead Formulation Scientist, Mid-Biotech
Frequently Asked Questions
-
Why is local toxicity specifically important for anti-obesity drugs?
Many modern AOTs are administered subcutaneously. Since these treatments are often chronic, even mild local irritation can lead to poor patient compliance or long-term tissue scarring.
-
Can you perform studies on obese animal models?
Yes, we frequently use DIO mice or rats, as the skin and adipose physiology in these models more accurately reflects the target patient population.
-
What species are available for these studies?
We offer studies in rodents (mice, rats) and non-rodent models (rabbits, mini-pigs), depending on the delivery system and regulatory requirements.
-
How do you quantify "minor" irritation?
We use a combination of standardized visual scoring and quantitative molecular markers of inflammation to provide an objective assessment.
-
Is Protheragen capable of testing microneedle patches?
Absolutely. We have specialized protocols to assess skin recovery and potential micro-lesions caused by transdermal delivery systems.
-
Do you provide systemic toxicity services as well?
While this service focuses on local effects, we can integrate systemic blood chemistry and hematology to provide a holistic safety overview.
-
What is the typical duration of a local toxicity study?
Studies range from single-dose acute irritation (24–72 hours) to repeat-dose local tolerance studies lasting 28 days or longer.
-
How is the data presented?
Clients receive high-resolution digital pathology images, tabulated scoring data, and a professional summary of findings.
-
Can you help with dosage selection for these studies?
Our senior scientists provide consultancy to determine the optimal safe dose (OSD) based on your drug's potency and intended clinical use.
How to Contact Us
Protheragen is committed to accelerating the development of safe and effective anti-obesity therapies. Our specialized preclinical services provide the rigorous data necessary to navigate the complexities of local tissue safety.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

