Anti-Obesity Therapeutic Toxicokinetics Study Service
InquiryAs the global prevalence of metabolic disorders reaches unprecedented levels, the development of next-generation anti-obesity medications—including GLP-1 receptor agonists, GIP/GCG dual agonists, and novel small molecules—has become a cornerstone of the pharmaceutical industry.
Toxicokinetics Study Service for Anti-Obesity Therapeutics
At Protheragen, we provide specialized toxicokinetics (TK) study services tailored specifically for anti-obesity drug candidates. Our preclinical TK programs bridge the gap between initial toxicology results and systemic exposure, defining the relationship between dose, plasma concentration, and toxicity. By integrating high-resolution bioanalytical platforms with rigorous metabolic profiling, we help developers determine safe starting doses for human trials and identify potential off-target effects related to weight loss mechanisms, such as gastrointestinal distress or metabolic imbalances.
Core Technologies
Protheragen leverages a suite of high-sensitivity analytical and computational tools designed to handle the unique pharmacokinetic profiles of anti-obesity therapeutics.
- High-Resolution Mass Spectrometry (Liquid Chromatography-tandem Mass Spectrometry (LC-MS/MS))
Utilized for the ultra-precise quantification of peptide mimetics and small molecules in complex biological matrices, ensuring detection even at picomolar concentrations.
- Multiplex Immunoassays
Optimized for monitoring biomarkers of adiposity and endocrine response, such as leptin, insulin, and ghrelin, alongside the therapeutic compound.
- Advanced TK Modeling Software
Implementation of non-compartmental analysis (NCA) and physiologically based pharmacokinetic (PBPK) modeling to predict tissue distribution and accumulation.
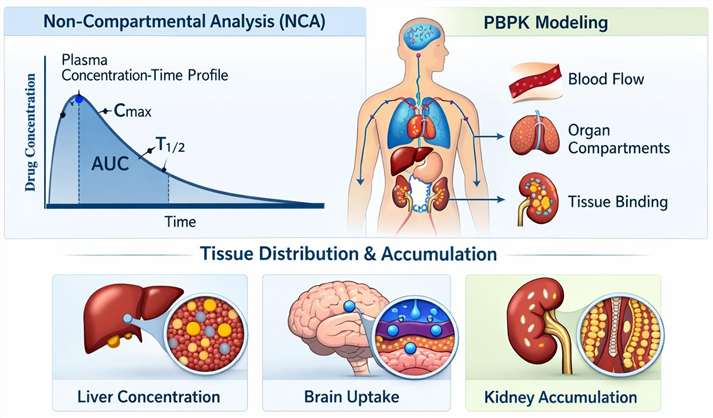
- In Vivo Metabolic Monitoring
Real-time tracking of metabolic rates and oxygen consumption during TK sampling to correlate drug exposure with physiological energy expenditure.
Solution Scope
Our expertise spans a wide range of anti-obesity modalities and study types:
- Single and Multiple Dose TK
Evaluation of accumulation risks and steady-state kinetics over chronic administration periods.
Determining if exposure increases linearly with dose to establish a reliable therapeutic window.
Identification of active or reactive metabolites that may contribute to the safety profile of the anti-obesity agent.
TK studies performed in diet-induced obesity (DIO) models to better reflect the disease state's impact on drug distribution and elimination.
Assessing the penetration of drugs into target tissues like white adipose tissue (WAT) or the central nervous system (CNS).
Contact Our Experts to Tailor a Study Scope that Fits Your Specific Therapeutic Modality.
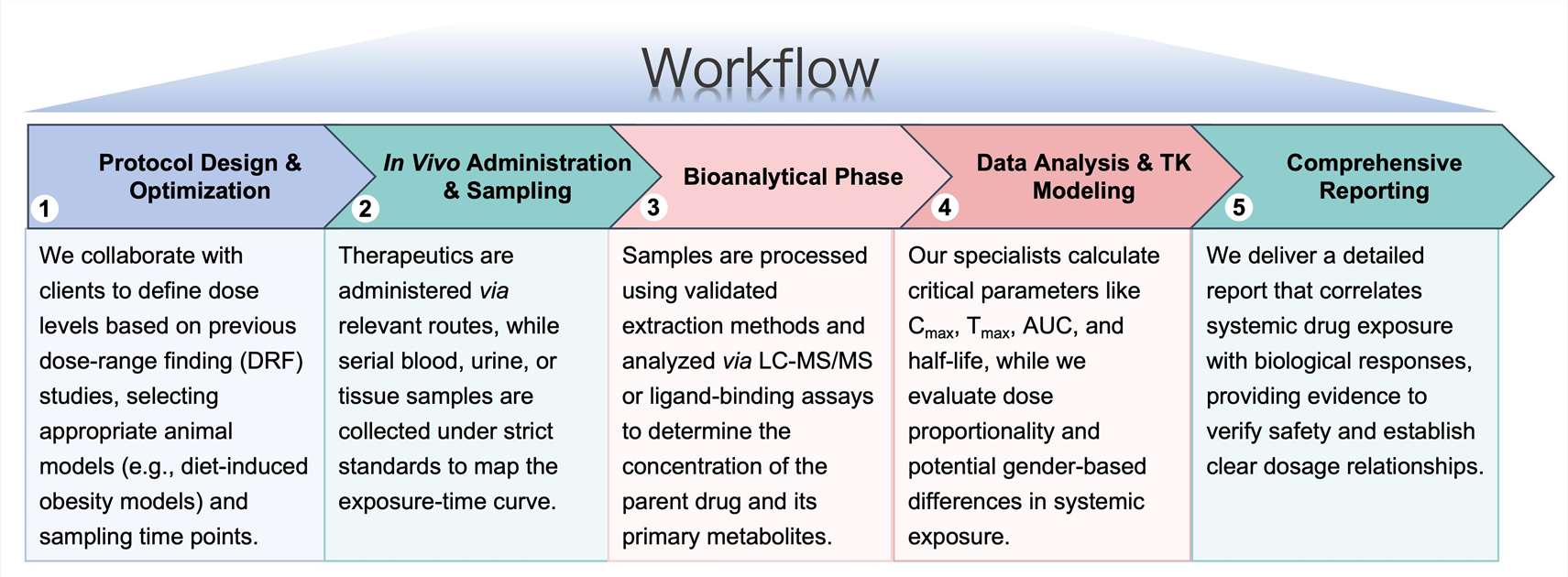
Workflow
Our optimized preclinical TK workflow is engineered to maximize data precision and accelerate the transition from initial testing to advanced development phases.
Fields of Application
Our specialized toxicokinetics studies provide critical safety and exposure insights across a diverse range of therapeutic modalities targeting metabolic regulation and weight management.
- GLP-1/GIP Agonist Development: Understanding the extended-release profiles of peptide-based weight loss drugs.
- Small Molecule Lipase Inhibitors: Assessing systemic absorption and potential gastrointestinal toxicity.
- Thermogenic Agents: Monitoring the safety of compounds that increase energy expenditure.
- Appetite Suppressants: Evaluating CNS penetration and potential neurotoxicity through exposure-response modeling.
Advantages
Protheragen stands at the forefront of metabolic drug development by offering unparalleled precision in preclinical safety assessment.
Expertise in Peptide Kinetics and Integrated Pathology
We possess deep experience handling the long half-lives and stability challenges associated with modern incretin mimetics. We correlate TK exposure data with histopathological findings in metabolic organs (liver, pancreas, kidneys) to provide a holistic view of drug safety.
Speed and Flexibility and Proven Success
Our agile laboratory structure allows for rapid turnaround times without compromising the depth of scientific analysis. We have supported numerous successful preclinical programs where our TK data directly informed the selection of the "no observed adverse effect level" (NOAEL).
Standardized Quality Assurance
Every TK study adheres to rigorous global safety protocols and quality benchmarks, ensuring all datasets meet the highest scientific standards for international evaluation and peer review.
Maximize Your Preclinical Success—Inquire About Our TK Services Today
Customer Review
Driving Peptide Innovation Through Deep Tissue Insights
"Working with Protheragen was a turning point for our peptide program. Their ability to quantify our lead candidate in adipose tissue while simultaneously providing a clear TK profile allowed us to justify our dosing strategy to our investors. The scientific depth of their team is remarkable."
Principal Scientist, Biotech Startup
Critical Metabolite Discovery and Safety Mapping
"Protheragen identified a specific metabolite we hadn't previously tracked, which proved crucial for our safety narrative. We plan to utilize their services for our entire metabolic pipeline moving forward."
Director of Toxicology, Mid-sized Pharma
Frequently Asked Questions
-
Why is TK specifically important for anti-obesity drugs?
Anti-obesity drugs often require chronic dosing. TK studies identify if the drug accumulates over time, which is critical for preventing long-term toxicity.
-
Can you perform TK studies in obese animal models?
Yes, we frequently use DIO mice and rats, as the physiological changes in obesity can significantly alter drug metabolism and distribution.
-
What matrices can you analyze?
While plasma is standard, we also analyze bile, urine, and specific tissues like adipose or muscle to understand the drug's full journey.
-
How do you handle high-potency compounds with very low systemic concentrations?
We use state-of-the-art LC-MS/MS with specialized sample enrichment techniques to achieve ultra-low limits of quantification.
-
How is dose linearity assessed?
We test multiple dose levels and use statistical modeling to determine if the increase in exposure is proportional to the increase in dose.
-
What is the typical turnaround time?
Depending on the complexity, a standard TK study from dosing to final report typically takes 6 to 10 weeks.
-
Can you help with metabolite identification?
Absolutely. Our high-resolution mass spectrometry can identify and quantify major metabolites to ensure they do not pose independent safety risks.
-
How do you ensure data integrity?
All processes are governed by a rigorous quality management system, including independent QA audits of raw data and reports.
How to Contact Us
Protheragen is committed to providing the high-quality toxicokinetic data required to advance your anti-obesity therapeutics. Our team of experts is ready to assist you in designing a study that meets your specific research goals and regulatory requirements.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

