Anti-Obesity Therapeutic Genotoxicity Study Service
InquiryAs the global prevalence of metabolic disorders rises, the development of anti-obesity medications (AOMs) has seen a paradigm shift from simple lipase inhibitors to complex GLP-1 receptor agonists and dual/triple incretin mimetics. However, the long-term safety profile of these chronic therapies is paramount. Protheragen provides specialized preclinical anti-obesity therapeutic genotoxicity study services designed to identify potential DNA damage, mutagenic risks, and chromosomal aberrations at the earliest stages of drug development.
Genotoxicity Study Service for Anti-Obesity Therapeutics
Since many anti-obesity agents interact with endocrine pathways or involve chronic systemic exposure, assessing their genotoxic potential—beyond standard regulatory batteries—is critical. Our services focus on the molecular mechanisms by which these compounds might induce oxidative stress or disrupt DNA repair mechanisms, ensuring that only the safest candidates progress to clinical phases.
Core Technologies
Protheragen leverages a suite of high-throughput and high-sensitivity assays tailored for metabolic research:
- High-Resolution Comet Assay (SCGE)
Utilized to detect single and double-stranded DNA breaks in metabolically active tissues such as the liver and pancreas, which are often targets for anti-obesity drugs.
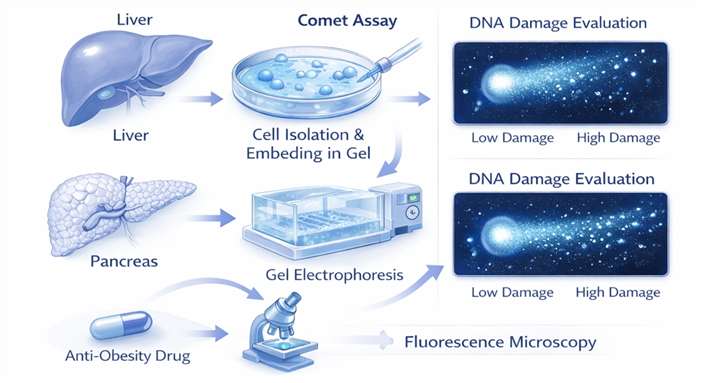
- Automated Flow Cytometric Micronucleus Scoring
Enhances the precision of identifying clastogenic or aneugenic agents by analyzing tens of thousands of cells for micronuclei formation, minimizing human bias.
- Ames Mutagenicity Suite
Optimized with specific S9 metabolic activation fractions to simulate the hepatic metabolism of lipophilic anti-obesity compounds.
- In Vitro Chromosomal Aberration Testing
High-content imaging platforms to visualize structural changes in chromosomes following exposure to therapeutic concentrations.
Solution Scope
Our expertise covers a broad range of anti-obesity modalities:
Genotoxic screening for lipase inhibitors and metabolic rate enhancers.
- Peptide & Protein Therapeutics
Specialized assays for GLP-1, GIP, and Glucagon Receptor Agonists, focusing on potential secondary genotoxicity driven by hormonal signaling.
- Combination Therapies
Assessing the synergistic genotoxic risks of multi-drug regimens.
Identifying whether the parent compound or its metabolic byproducts pose a greater risk to genomic integrity.
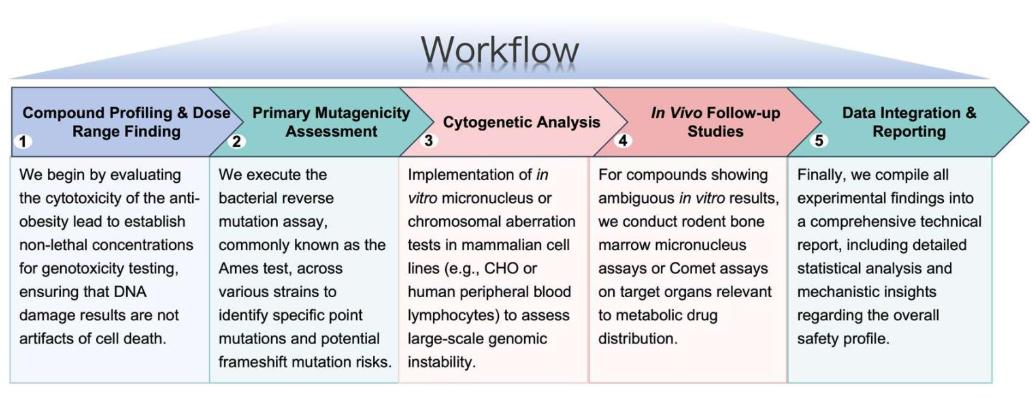
Workflow
Our standardized yet flexible workflow ensures the generation of rigorous, high-quality data to validate the genomic safety of your therapeutic candidates:
Fields of Application
Our specialized genotoxicity assessments are integral throughout the drug development lifecycle, providing critical safety insights that guide the evolution of next-generation metabolic therapies from initial discovery to final validation.
- Preclinical Drug Discovery: Screening diverse chemical libraries to eliminate genotoxic leads early.
- Safety Pharmacology: Investigating the mechanisms of DNA damage in specific organ systems like the liver, kidneys, or adipose tissue.
- Optimization of Drug Delivery: Assessing the safety of novel delivery vehicles, such as lipid nanoparticles used for metabolic gene therapies.
- Comprehensive Genomic Safety Assessment: Completing the full suite of DNA integrity tests required to validate your candidate's safety profile.
Contact Our Team for a Consultation on Your Anti-Obesity Pipeline.
Advantages
Protheragen distinguishes itself through deep specialization in metabolic pharmacology:
Metabolic Relevance
Unlike generic CROs, we optimize our assays to account for the unique pharmacokinetic profiles of anti-obesity drugs, particularly their interaction with lipid metabolism.
Advanced Detection Sensitivity
Our platforms can detect subtle DNA damage that might be overlooked by standard protocols, supported by published data demonstrating higher correlation with long-term carcinogenicity studies.
Expert Interpretative Consulting
We do not just provide raw data; our PhD-level biologists provide context on whether a positive result is a direct chemical effect or a secondary physiological response.
Customer Review
Overcoming Scaffolding Risks with Mechanistic Clarity
"Working with Protheragen was a turning point for our dual-agonist program. We were concerned about potential DNA reactivity in a novel scaffold. Their team not only performed the standard Ames and Micronucleus tests but also provided a mechanistic Comet assay that clarified the safety profile for our target tissues. Their expertise in metabolic drugs is unmatched."
Senior VP of Toxicology, Biotech Startup
Precision Safety Validation for Complex Lipophilic Candidates
"What impressed us most was their willingness to adapt the protocol for our highly lipophilic compound, which had caused solubility issues at other CROs. We plan to keep Protheragen as our primary partner for all upcoming preclinical safety assessments."
Principal Scientist, Global Pharmaceutical Firm
Frequently Asked Questions
-
Why is genotoxicity testing specifically important for anti-obesity drugs?
Since these drugs are often taken chronically—sometimes for a lifetime—any low-level mutagenic potential could significantly increase long-term cancer risks.
-
Can Protheragen handle lipophilic compounds often found in weight loss research?
Yes, we utilize specialized solvent systems and delivery methods to ensure these compounds remain bioavailable in our in vitro assay systems.
-
How do you differentiate between apoptosis and true genotoxicity?
We use concurrent cytotoxicity markers and "dye-exclusion" methods to ensure that DNA fragmentation is a primary event rather than a byproduct of cell death.
-
What is the typical sample requirement for a full battery?
Depending on the assays, we typically require 500mg to 1g of the test article, though micro-scale options are available for early screening.
-
Can you perform Comet assays on specific tissues like the hypothalamus?
Yes, our in vivo comet assay service allows for tissue-specific DNA damage assessment in any harvested organ.
-
Do you provide support if a "positive" result is found?
Yes, we offer mechanistic follow-up studies to determine if the result is a "false positive" or if it can be mitigated through structural modifications.
-
How can I start a project with Protheragen?
You can reach out through our website inquiry form to schedule a technical meeting with our lead toxicologist.
How to Contact Us
Protheragen provides a comprehensive, high-precision preclinical anti-obesity therapeutic genotoxicity study service. By combining advanced molecular assays with deep expertise in metabolic biology, we ensure that your drug candidates are evaluated with the highest level of scientific rigor. From initial Ames screening to complex in vivo comet assays, we provide the data you need to progress with confidence.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

