Anti-Obesity Therapeutic Immunotoxicity Study Service
InquiryIn the rapidly evolving landscape of metabolic health, anti-obesity therapeutics—ranging from GLP-1 receptor agonists to novel peptide multi-agonists—have emerged as transformative interventions. However, the intersection of metabolic dysfunction and the immune system presents a unique toxicological challenge. Obesity is characterized by chronic, low-grade systemic inflammation and a significant "rewiring" of immune responses, often shifting from Th1-driven to Th2-like inflammatory profiles. This altered baseline means that conventional immunotoxicity assessments in lean animal models may fail to predict adverse immune reactions in the intended metabolic environment.
Immunotoxicity Evaluation for Anti-Obesity Therapies
Protheragen offers specialized preclinical anti-obesity therapeutic immunotoxicity study services designed to interrogate the complex "immunometabolic axis." We evaluate how new drug candidates interact with an already primed immune system, identifying potential risks such as exacerbated cytokine release, hypersensitivity, or unintended immunosuppression. Our services ensure that your therapeutic profile is robustly characterized before transitioning to the next phase of development.
Core Technologies
To provide high-resolution insights into drug-immune interactions, Protheragen utilizes a suite of cutting-edge technologies tailored for metabolic research:
- Multiplex Cytokine & Adipokine Profiling
Utilizing bead-based assays to simultaneously quantify systemic pro-inflammatory markers (TNF-α, IL-6, IL-1β) and metabolic regulators (leptin, adiponectin) to detect subtle shifts in the inflammatory milieu.
- High-Dimensional Flow Cytometry
Specialized panels for deep immunophenotyping of T-cell subsets, M1/M2 macrophage polarization in adipose tissue, and B-cell activation states.
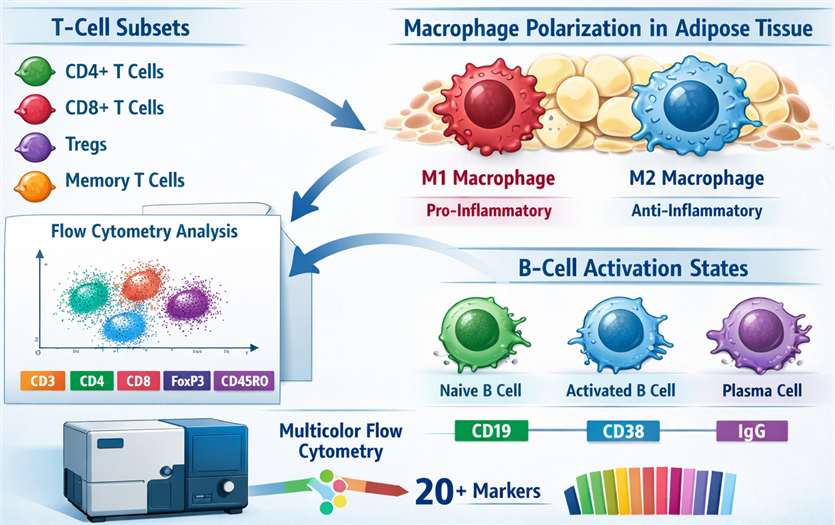
- Diet-Induced Obesity (DIO) & Genetic Models
Validated animal models that replicate human metabolic syndrome, including insulin resistance and chronic inflammation, provide a biologically relevant background for toxicity testing.
- Adipose Tissue-Specific Histopathology
Advanced imaging and immunohistochemistry (IHC) to evaluate "crown-like structures" (CLS) and local immune cell infiltration within white and brown adipose depots.
- Ex Vivo Immune Function Assays
Assessments of T-cell proliferation and NK cell cytotoxicity within the context of metabolic exhaustion to determine if therapeutics inadvertently impair host defense.
Solution Scope
Our preclinical immunotoxicity services cover a broad spectrum of anti-obesity modalities:
- Incretin Mimetics & Multi-agonists
Safety profiling for GLP-1, GIP, and Glucagon Receptor Agonists, focusing on potential injection site hypersensitivity and systemic cytokine modulation.
- Small Molecule Metabolic Modulators
Evaluation of mitochondrial uncouplers, kinase inhibitors, and GPCR ligands for off-target immunosuppression.
- Peptide & Protein Therapeutics
Assessment of immunogenicity and the development of anti-drug antibodies (ADAs) that could alter pharmacokinetics or trigger immune-mediated adverse events.
- Lipid-Targeting Agents
Investigating the impact of altered lipid metabolism on macrophage function and chronic inflammatory signaling.
Explore Our Full Preclinical Metabolic Service Catalog
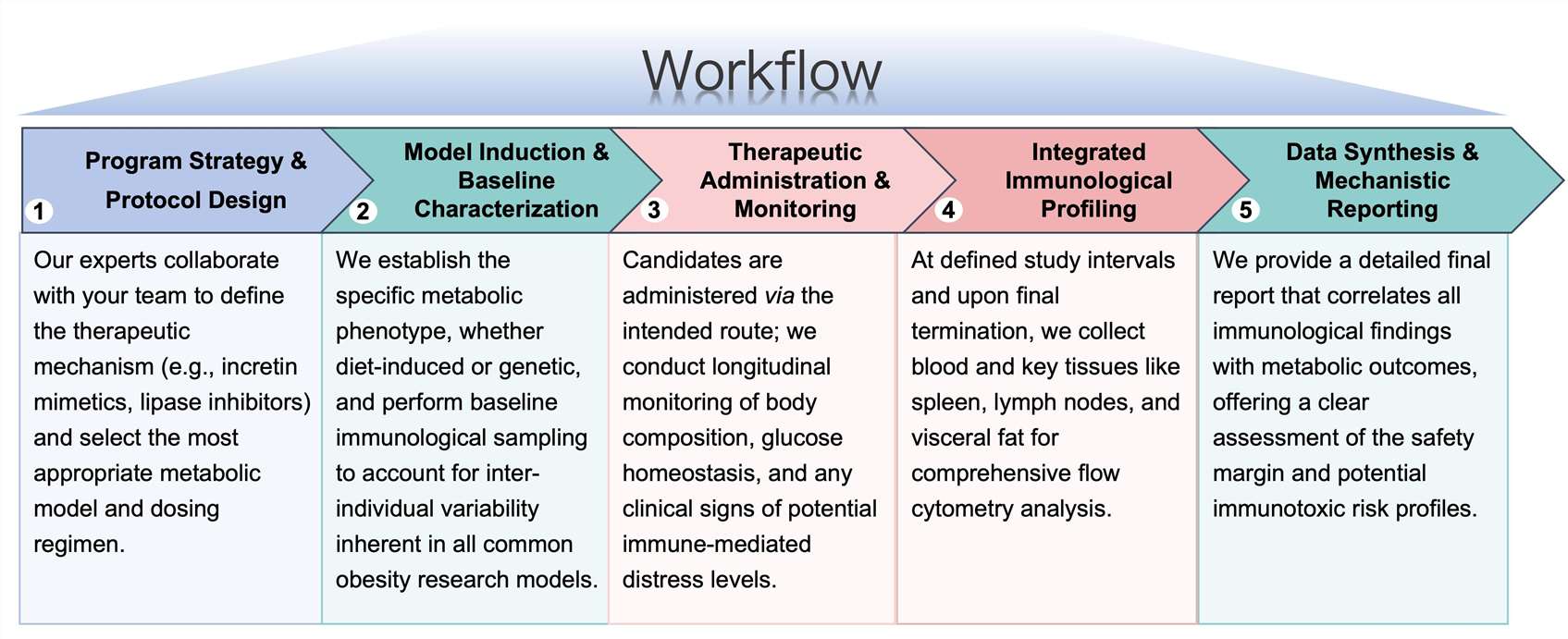
Workflow
Protheragen follows a rigorous, multi-stage workflow to ensure every immunotoxicity study is scientifically sound and data-rich.
Fields of Application
The clinical and commercial success of next-generation anti-obesity agents hinges on a profound understanding of their immunological impact within a metabolically "primed" environment, making our specialized immunotoxicity services a critical asset across several high-stakes therapeutic landscapes.
- Drug Safety & Toxicology: Identifying safe starting doses and potential immune-related adverse effects (irAEs).
- Metabolic Syndrome Research: Understanding how weight-loss agents influence systemic chronic inflammation (meta-inflammation).
- Onco-Metabolism: Evaluating how anti-obesity drugs might interact with cancer immunotherapies, given the known "obesity paradox" in checkpoint inhibitor efficacy.
- Cardiovascular Safety: Assessing if immune-mediated vascular inflammation is mitigated or exacerbated by novel anti-obesity candidates.
Consult with Our Specialists to Align Your Safety Program with Realities.
Advantages
Protheragen stands at the forefront of immunometabolic research. Our specialized focus allows us to provide insights that generalist CROs often overlook.
Disease-Relevant Background
We do not test metabolic drugs on healthy, lean animals alone. We understand that obesity alters the PD-1/PD-L1 axis and T-cell exhaustion markers; therefore, our studies are conducted in a "metabolically stressed" environment.
Expert Interpretation
Our senior scientists possess over 20 years of experience in both immunology and endocrinology, allowing us to distinguish between the beneficial anti-inflammatory effects of weight loss and direct drug-induced immunotoxicity.
Advanced Adipose Analysis
We go beyond systemic blood draws, analyzing the immune landscape within the adipose tissue—the primary site of metabolic inflammation.
Reliable Data Standards
Our methodologies are supported by published data demonstrating the correlation between preclinical immunotoxic markers and metabolic outcomes in complex models.
Publication Data
Title: Antiinflammatory actions of glucagon-like peptide-1–based therapies beyond metabolic benefits
Journal: The Journal of Clinical Investigation, 2025
DOI: https://doi.org/10.1172/JCI194751.
Summary: This article reviews the weight loss–dependent and independent anti-inflammatory effects of GLP-1–based therapies, extending beyond their well-known metabolic benefits (e.g., glucose control, weight loss for type 2 diabetes and obesity). GLP-1 medicines (e.g., semaglutide, tirzepatide) exert anti-inflammatory actions through direct mechanisms (modulating immune cells like T cells, microglia) and indirect pathways (neuro-immune crosstalk, gut-brain circuits), with preclinical and clinical evidence supporting their efficacy in mitigating inflammation-related diseases—including cardiovascular, liver, kidney, neurological, joint, pulmonary, and intestinal disorders. The review also discusses emerging dual/triple receptor agonists (e.g., tirzepatide targeting GLP-1R/GIPR) and highlights unresolved questions, such as species differences in GLP-1 regulation and precise cellular targets of GLP-1R signaling, which require further investigation to optimize therapeutic applications.
Key Findings
- Dual Antiinflammatory Pathways: GLP-1 medicines reduce inflammation through both metabolic benefits (weight loss, glucose control) and intrinsic actions independent of metabolism (e.g., inhibiting immune cell cytokine release).
- Broad Disease Efficacy: They mitigate inflammation in multiple conditions, including cardiovascular, liver (MASH), kidney, neurological, joint, pulmonary, and intestinal disorders.
- Immune Cell Regulation: GLP-1R signaling suppresses proinflammatory cytokines (IFN-γ, TNF-α) in T cells, microglia, and other immune cells, dampening overactive immune responses.
- Neural and Gut-Brain Crosstalk: Central GLP-1R activation (in brain neurons) and intestinal L cells (as inflammation sensors) drive indirect antiinflammatory effects via neuro-immune and gut-brain circuits.
- Emerging Agonists: Dual GLP-1R/GIPR agonists like tirzepatide offer unique anti-inflammatory benefits compared to single agonists, enhancing efficacy in specific conditions.
- Strong Clinical Evidence: Trials show semaglutide resolves MASH (63% vs. 34% placebo) and reduces CKD progression (24% risk reduction), while tirzepatide improves obstructive sleep apnea.
- Unresolved Questions: Species differences in GLP-1 regulation, low peripheral GLP-1R expression, and long-term safety (e.g., cholecystitis risk) require further research.
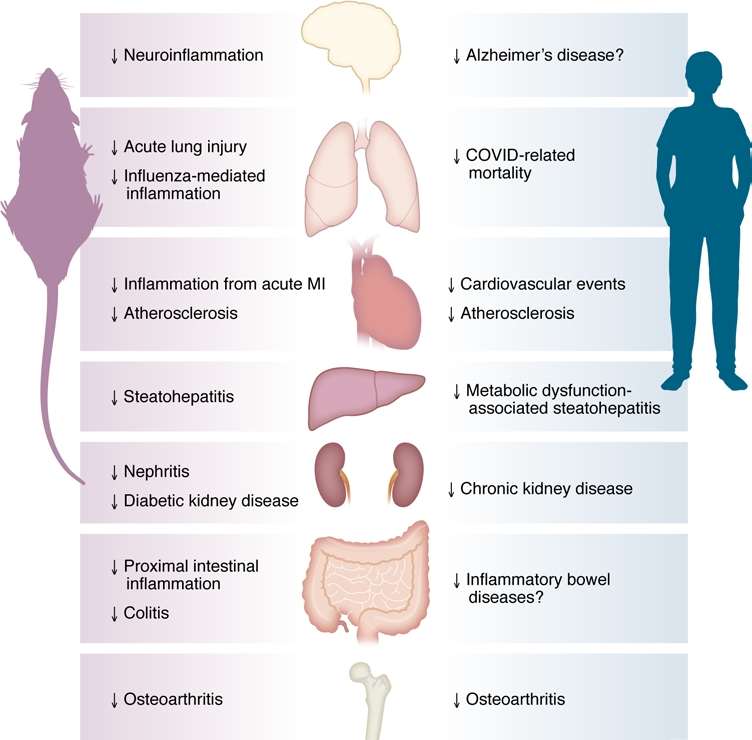 Fig.1 GLP-1 medicines exert broad anti-inflammatory effects across organs, targeting conditions like cardiovascular disease, MASH, chronic kidney disease, and osteoarthritis—extending beyond their metabolic benefits. (Wong & Drucker, 2025)
Fig.1 GLP-1 medicines exert broad anti-inflammatory effects across organs, targeting conditions like cardiovascular disease, MASH, chronic kidney disease, and osteoarthritis—extending beyond their metabolic benefits. (Wong & Drucker, 2025)
Customer Review
Strategic Validation of Lead Peptide Safety in High-Risk Models
"Working with Protheragen was a turning point for our lead peptide program. We were concerned about potential hypersensitivity in metabolic patients. Protheragen designed a DIO-specific immunotoxicity study that provided clear evidence of our candidate's safety profile, specifically showing no exacerbation of systemic cytokines. Their expertise in the immunometabolic axis is unparalleled."
Dr. E. V., Director of Early Development
Deep Immunophenotyping Insights for Stakeholder Confidence
"The depth of the flow cytometry data we received from Protheragen was impressive. They didn't just look at blood; they showed us exactly what was happening inside the visceral fat. This level of detail gave our stakeholders the confidence to move forward. We view Protheragen as a long-term strategic partner for our metabolic pipeline."
Mr. M. J., Senior Toxicologist
Frequently Asked Questions
-
Why is immunotoxicity testing different for anti-obesity drugs?
Obesity induces a baseline state of chronic inflammation. A drug that is safe in a lean model might trigger an exaggerated cytokine response or unexpected hypersensitivity in a pro-inflammatory metabolic environment.
-
Can you detect the shift from Th1 to Th2 immune responses?
Yes, our flow cytometry panels and cytokine assays are specifically designed to monitor shifts in helper T-cell polarization, which is a key feature of obesity-related immune rewiring.
-
How do you account for weight loss as a confounding factor?
We utilize "pair-fed" control groups and sophisticated statistical modeling to separate the direct immunotoxic effects of the drug from the indirect immunological changes caused by rapid weight loss.
-
What species do you typically use for these studies?
While we primarily utilize validated rodent DIO models (C57BL/6J), we can adapt protocols for other species based on the cross-reactivity of your therapeutic agent.
-
Can you assess "crown-like structures" (CLS) in adipose tissue?
Absolutely. Our histopathology team specializes in identifying and quantifying these indicators of macrophage-mediated inflammation in fat depots.
-
Do you measure anti-drug antibodies (ADAs)?
Yes, we provide robust assays to detect and characterize ADAs, which are critical for biologicals and peptide-based anti-obesity therapies.
-
How long does a typical DIO immunotoxicity study take?
Induction of the obesity phenotype typically takes 10-12 weeks, followed by a 4-8 week treatment and assessment period, depending on your specific requirements.
-
What is the "obesity paradox", and do you test for it?
The obesity paradox suggests that while obesity increases cancer risk, it may enhance response to certain immunotherapies. We can design studies to see if your drug interferes with these specific immune checkpoints.
-
How do I start a project with Protheragen?
Simply reach out via our inquiry form. We will schedule a technical consultation to discuss your molecule's mechanism and your specific safety concerns.
How to Contact Us
Protheragen provides the specialized expertise and advanced technological framework necessary to navigate the complexities of anti-obesity therapeutic development. By integrating metabolic relevance into every immunotoxicity study, we help our clients mitigate risk and accelerate the delivery of safe and effective treatments to patients.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Wong, C.K.; Drucker, D.J. Antiinflammatory actions of glucagon-like peptide-1–based therapies beyond metabolic benefits. J Clin Invest. 2025, 135(21):e194751. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

