Anti-Obesity Therapeutic Ocular Toxicity Study Service
InquiryIn the rapidly evolving landscape of metabolic health, anti-obesity medications—particularly GLP-1 and dual GLP-1/GIP receptor agonists—have revolutionized patient care. However, recent clinical signals, including concerns regarding non-arteritic anterior ischemic optic neuropathy (NAION) and the progression of diabetic retinopathy (DR), have placed ocular safety at the forefront of regulatory scrutiny.
Advanced Ocular Safety Assessment for Metabolic & Anti-Obesity Therapeutics
Protheragen provides specialized preclinical anti-obesity therapeutic ocular toxicity study services designed to mitigate these risks early in the development cycle. By integrating advanced ophthalmic imaging with robust metabolic disease models, we empower biopharmaceutical developers to secure their path to research success with definitive safety data.
Core Technologies
To deliver the high-fidelity insights necessary for precise therapeutic characterization, Protheragen utilizes a sophisticated suite of advanced diagnostic and analytical technologies:
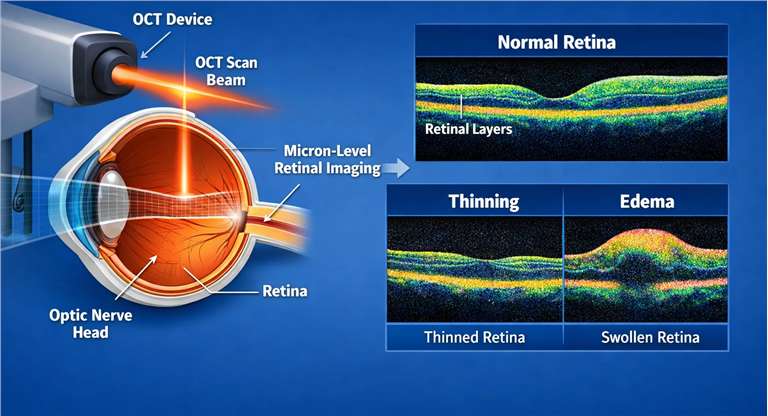
- High-Resolution Optical Coherence Tomography (OCT)
Provides non-invasive, micron-level cross-sections of the retina and optic nerve head to detect thinning or edema.
- Fluorescein & Indocyanine Green (ICG) Angiography
Essential for evaluating the blood-retinal barrier and choroidal perfusion, specifically targeting potential ischemic signals related to GLP-1 mechanisms.
- Full-Field & Pattern Electroretinography (ERG)
Measures the functional response of retinal cells to detect sub-clinical neurotoxicity or metabolic dysfunction.
- Slit-Lamp Biomicroscopy & Indirect Ophthalmoscopy
Utilizing the McDonald-Shadduck scoring system for standardized assessment of the anterior and posterior segments.
- Automated Tonometry
Precise monitoring of intraocular pressure (IOP) to assess the impact of systemic metabolic shifts on ocular fluid dynamics.
Solution Scope
Protheragen supports a wide range of anti-obesity modalities, including:
- Incretin Mimetics
Comprehensive safety profiling for GLP-1, GIP, and Glucagon Receptor Agonists.
- Small Molecule Metabolites
Evaluation of mitochondrial uncouplers and enzyme inhibitors.
- CNS-Acting Appetite Suppressants
Neuro-ophthalmic assessments to detect off-target effects on the visual pathway.
- Peptide & Antibody Therapeutics
Monitoring for immune-mediated ocular inflammation or vascular deposition.
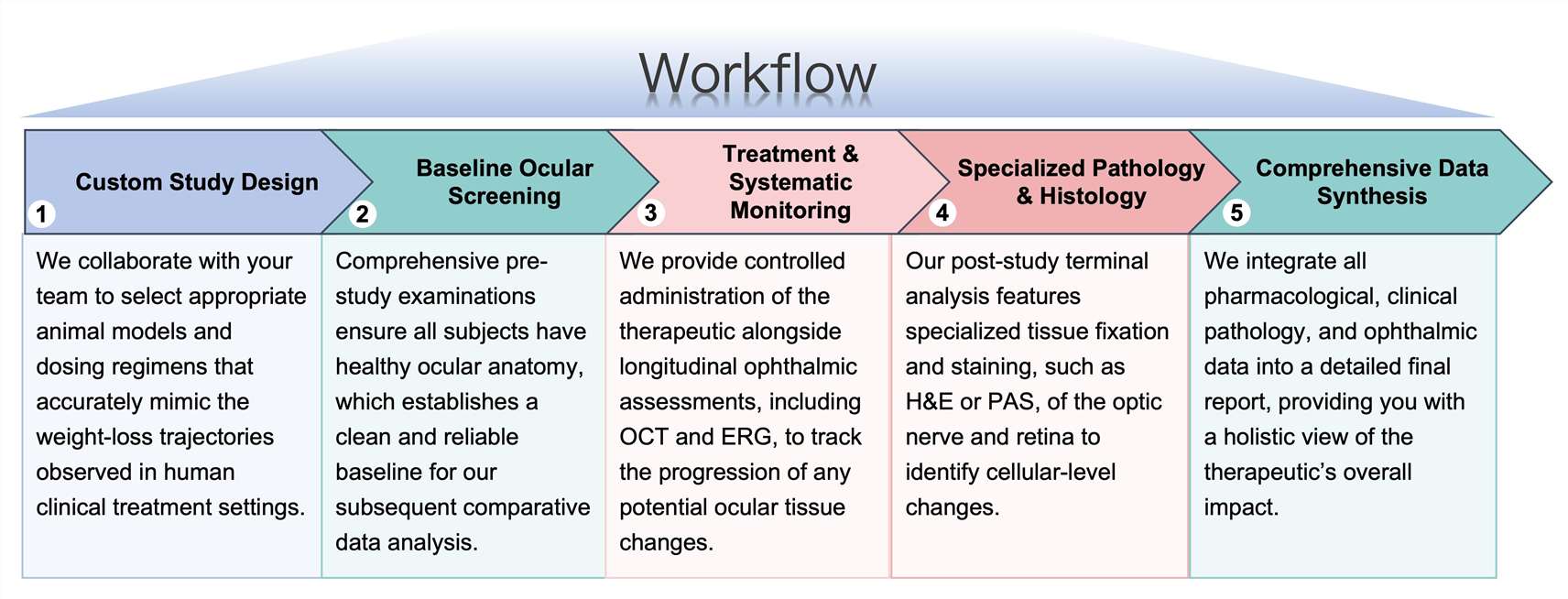
Workflow
Our workflow is engineered for rigorous precision at every stage, providing a seamless transition from initial study design to comprehensive final reporting.
Fields of Application
Our specialized assessment platform is critical for various stages of therapeutic discovery and mechanistic evaluation:
- Safety Profiling & Characterization: Establishing a definitive ocular safety profile for systemic drug candidates during early-stage development.
- Mechanistic Investigative Studies: Exploring the biological pathways behind observed ocular changes or metabolism-related vision shifts to understand the "why" behind the data.
- Comparative Candidate Selection: Differentiating between multiple lead compounds by identifying the molecule with the most favorable ocular safety margin.
- Metabolic Impact Assessment: Distinguishing between the ocular physiological changes caused by rapid systemic weight loss versus direct therapeutic interaction with ocular tissues.
- Off-Target Effect Screening: Evaluating the impact of CNS-acting agents or peptides on the visual pathway and retinal microenvironment to ensure narrow therapeutic targeting.
Contact Our Team for More Information and to Discuss Your Project
Advantages
Choosing Protheragen means partnering with a specialist who understands the unique intersection of metabolic disease and vision science.
We utilize specialized models of obesity and diabetes (e.g., high-fat diet models) to reflect the "disk-at-risk" anatomy often seen in the target patient population.
Sensitivity to Ischemic Signals
Our protocols are specifically tuned to detect early indicators of NAION and retinal vascular compromise, utilizing "published data" to benchmark normative vs. adverse ranges.
Seamless Integration
We provide integrated PK/PD and ocular distribution data, allowing you to correlate systemic drug levels with ocular safety profiles.
Publication Data
Title: The Effect of Semaglutide and GLP-1 RAs on Risk of Nonarteritic Anterior Ischemic Optic Neuropathy
Journal: American Journal of Ophthalmology, 2025
DOI: https://doi.org/10.1016/j.ajo.2025.02.025
Summary: This retrospective matched cohort study, conducted using the TriNetX US collaborative network (116 million patients), investigates whether glucagon-like peptide-1 receptor agonists (GLP-1 RAs) — including semaglutide — increase the risk of nonarteritic anterior ischemic optic neuropathy (NAION) or ischemic optic neuropathy (ION) in patients with type 2 diabetes (T2DM) or high BMI (overweight/obese). Propensity score matching balanced demographics and risk factors, with outcomes evaluated at multiple time points. The study concludes that semaglutide and other GLP-1 RAs do not significantly raise NAION/ION risk compared to non-GLP-1 RA medications, addressing prior uncertainties about the drug class's ocular safety.
Key Findings
- No elevated NAION/ION risk with semaglutide: In T2DM patients, semaglutide showed no significant increase in NAION (5-year RR=0.7, 95% CI: 0.523-0.937) or ION (5-year RR=0.788, 95% CI: 0.609-1.102); high BMI patients also had no increased risk at 1-2 years.
- Consistent results across GLP-1 RA class: T2DM and high BMI patients taking any GLP-1 RA (e.g., liraglutide, tirzepatide) had similar NAION/ION risk to controls (T2DM 5-year NAION RR=0.887, 95% CI: 0.735-1.071).
- Obesity sub-analysis confirms safety: Patients with BMI ≥30 kg/m² prescribed semaglutide or any GLP-1 RA showed no higher NAION risk at 1-2 years.
- Negative control validates results: No difference in allergic contact dermatitis risk (unrelated to GLP-1 RAs) between study and control groups, confirming no confounding biases.
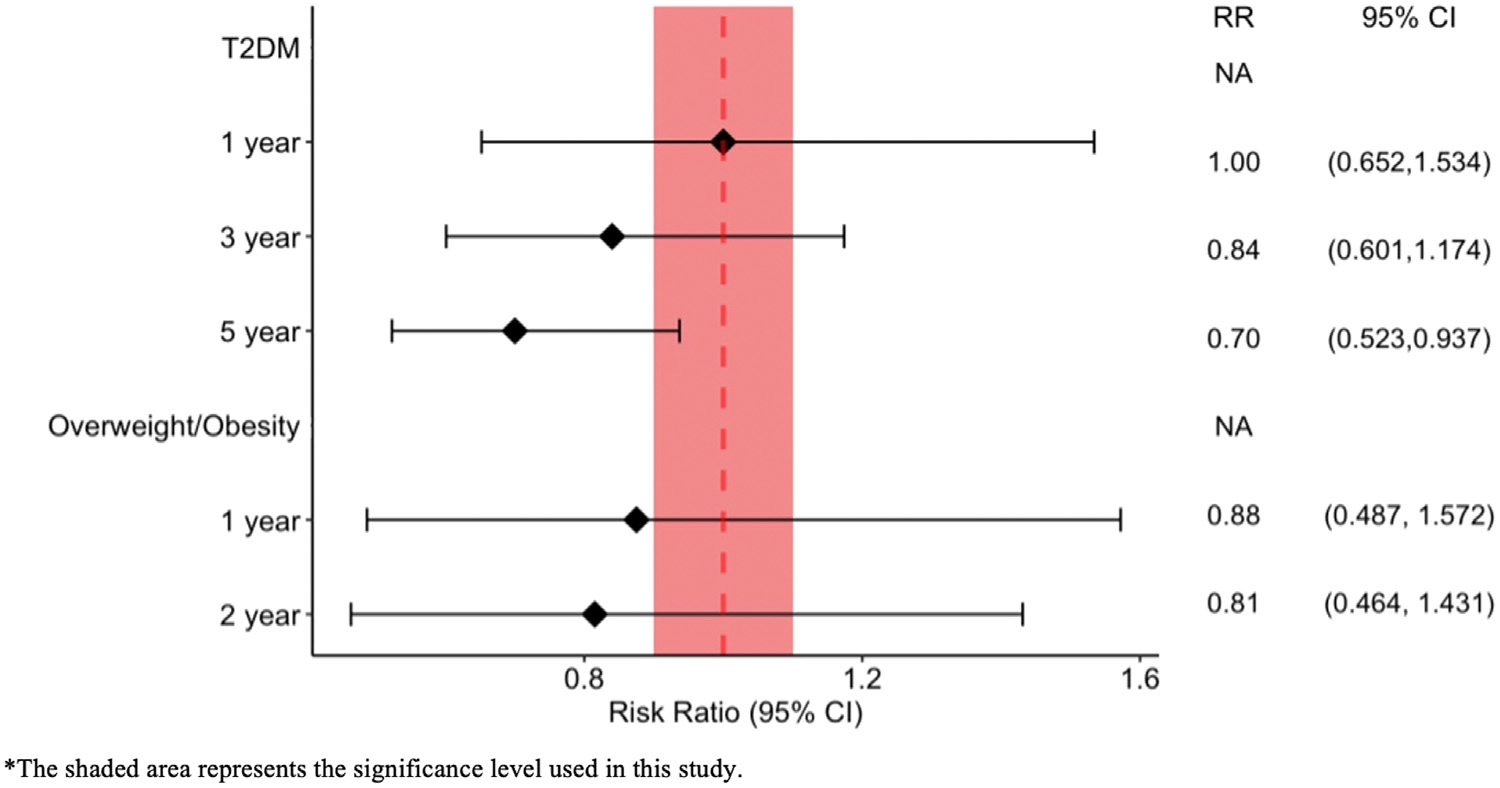 Fig.1 Risk ratio of NAION in patients prescribed semaglutide vs matched controls. (Abbass, et al., 2025)
Fig.1 Risk ratio of NAION in patients prescribed semaglutide vs matched controls. (Abbass, et al., 2025)
Customer Review
Unlocking Scientific Clarity in Metabolic Research
"The team at Protheragen provided indispensable insights for our latest dual-agonist program. Their ability to correlate rapid weight loss metrics with high-resolution OCT imaging allowed us to confirm that the minor retinal changes we observed were related to systemic metabolic shifts rather than direct drug toxicity. This level of scientific clarity was pivotal for our internal data confidence and program direction."
Principal Scientist, Metabolic Research
Exceptional Technical Depth and Longitudinal Support
"Partnering with Protheragen has been a seamless experience. Their veterinary ophthalmologists offer a deep understanding of how metabolic therapeutics interact with ocular anatomy, and their technical reporting is exceptionally thorough. We are already planning our next longitudinal study with them to further explore the long-term health of the optic nerve head."
Lead Pharmacologist, Global Biopharma
Frequently Asked Questions
-
Why is ocular toxicity specifically important for anti-obesity drugs?
Clinical data for certain GLP-1s have shown a potential link to NAION and retinopathy progression; preclinical studies help determine if this is a drug effect or a metabolic shift.
-
Which animal species do you recommend for these studies?
We typically use rodents (ZDF rats) for metabolic disease modeling and NHPs for their human-like ocular anatomy.
-
Can you detect early signs of NAION preclinically?
Yes, using OCT and angiography, we monitor optic nerve head perfusion and structural integrity for ischemic signals.
-
How long does a typical ocular toxicity study take?
Timelines vary by dosing duration (e.g., 28-day vs. 6-month), but most projects take 4–6 months from initiation to final report.
-
Do you assess the impact of rapid blood glucose drops on the retina?
Yes, we correlate ocular findings with systemic glucose monitoring to differentiate between drug toxicity and glycemic-related "re-entry" retinopathy.
-
What is the disk-at-risk, and do you model it?
It refers to a small cup-to-disk ratio; we select models and individuals that mimic this vulnerability to increase the study's predictive value.
-
Can you perform specialized IHC on the optic nerve?
Yes, we offer immunohistochemistry for markers of neuroinflammation, oxidative stress, and vascular integrity.
-
Do you provide bioanalytical support for ocular tissues?
We offer full PK support, measuring drug concentrations in the vitreous, retina, and aqueous humor.
How to Contact Us
Protheragen is your dedicated partner in navigating the complexities of anti-obesity drug development. Our specialized ocular toxicity studies provide the clarity and confidence needed to advance your therapeutic from the bench to the clinic.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Abbass, N. J.; et al. The Effect of Semaglutide and GLP-1 RAs on Risk of Nonarteritic Anterior Ischemic Optic Neuropathy. American Journal of Ophthalmology. 2025, 274, 24–31. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

