Anti-Obesity Therapeutic Nephrotoxicity Study Service
InquiryThe global rise in metabolic disorders has catalyzed the development of next-generation anti-obesity therapeutics, including GLP-1 receptor agonists, GIP/GLP-1 co-agonists, and various novel small molecules. However, the metabolic milieu of obesity—characterized by glomerular hyperfiltration, chronic low-grade inflammation, and lipotoxicity—significantly alters renal physiology. This heightened baseline vulnerability means that even minor drug-induced stress can precipitate severe nephrotoxicity.
Preclinical Nephrotoxicity Assessment in Metabolic Disease Models
At Protheragen, we provide a specialized anti-obesity therapeutic nephrotoxicity study service designed to evaluate the renal safety profile of candidates within the specific context of the "obese kidney." Our preclinical services move beyond standard toxicological screening by integrating metabolic stressors that mimic the clinical reality of obese and diabetic populations. We focus on identifying early markers of structural and functional damage, ensuring that your drug candidate is not only efficacious for weight loss but also safe for the millions of patients with obesity-associated renal susceptibility.
Core Technologies
Our platform leverages advanced bioanalytical and physiological modeling technologies to detect subtle renal insults that traditional assays often miss.
- Translational Metabolic Models
We utilize diet-induced obesity (DIO) and high-fat diet (HFD) models that exhibit the hallmark hemodynamic changes of obesity, such as increased renal plasma flow and intraglomerular hypertension.
- High-Resolution 3D Kidney-on-a-Chip
Our microphysiological systems (MPS) replicate the human proximal tubule environment, allowing for the assessment of drug-induced injury under fluid shear stress, which is critical for evaluating tubular handling of large-molecule anti-obesity agents.
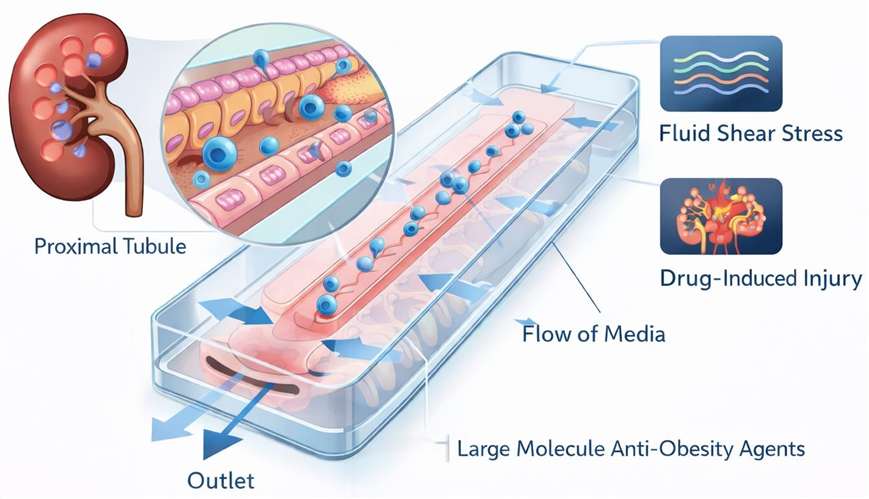
- Multiparametric 7T MRI Imaging
Non-invasive longitudinal monitoring of renal architecture, vascular dynamics, and tissue water content (T1/T2 mapping) to detect early-stage edema and fibrosis.
- Novel Biomarker Panel
Detection of FDA-recognized urinary markers, including KIM-1, NGAL, clusterin, and cystatin C, which provide higher sensitivity than traditional serum creatinine for early drug-induced kidney injury (DIKI).
- Oxidative Stress & Lipotoxicity Profiling
Quantitative assessment of ROS generation, lipid peroxidation (MDA levels), and mitochondrial dysfunction specifically in renal cortical tissues.
Solution Scope
Our service is tailored to handle a diverse range of anti-obesity modalities and metabolic contexts:
- Peptide & Large Molecule Evaluation
Assessing the renal accumulation and potential toxicity of GLP-1, GIP, and Glucagon Receptor Agonists.
- Small Molecule Nephrotoxicity
Screening novel metabolic modulators for off-target effects on organic anion/cation transporters (OAT/OCT) in the kidney.
- Combination Therapy Screening
Evaluating the synergistic renal risks of anti-obesity drugs when co-administered with standard-of-care treatments like SGLT2 inhibitors or RAAS modulators.
- Drug-Induced AKI to CKD Transition
Assessing the potential for acute insults to progress into chronic interstitial fibrosis and glomerulosclerosis.
- Dose Extrapolation Studies
Helping determine safe starting doses for clinical trials by identifying the "no observed adverse effect level" (NOAEL) in metabolically compromised models.
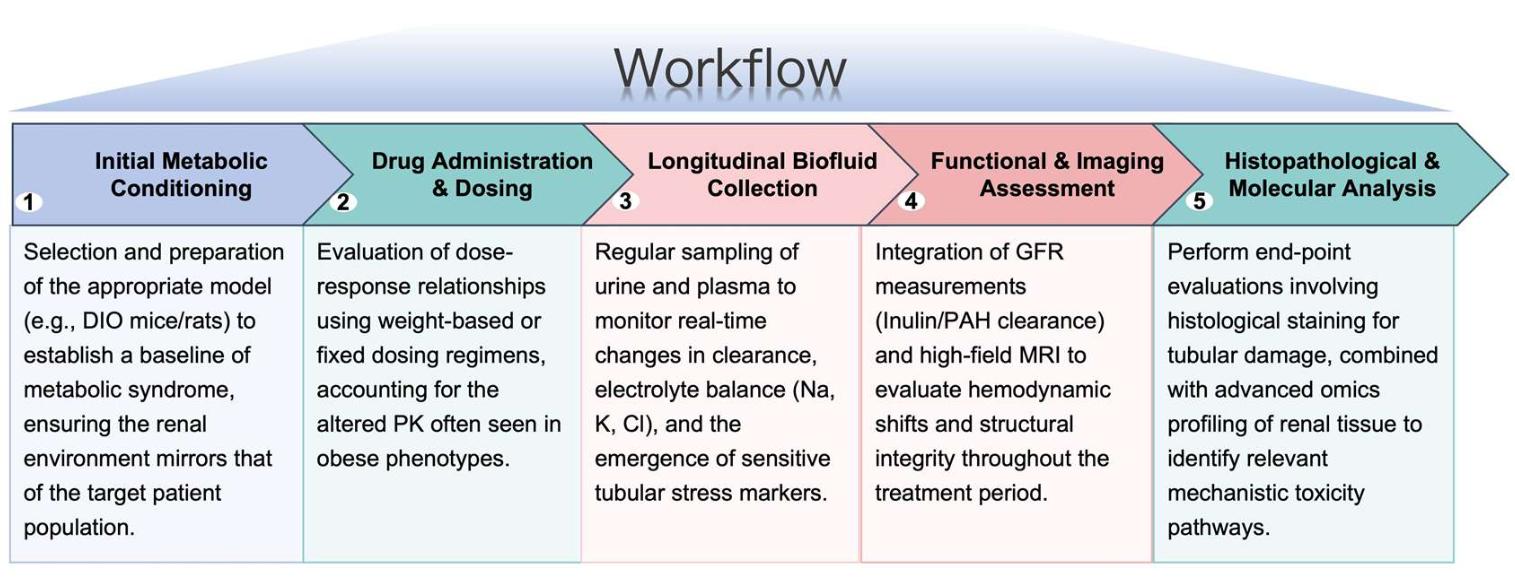
Workflow
Protheragen employs a rigorous, multi-stage workflow to ensure comprehensive data generation for your therapeutic pipeline.
Contact Our Team for a Detailed Project Consultation
Fields of Application
Our anti-obesity therapeutic nephrotoxicity study service serves as a vital bridge between metabolic discovery and candidate selection, providing the precise data required across various stages of the development pipeline.
- Metabolic Drug Discovery: Early-stage lead optimization to de-risk candidates with potential renal liability.
- Cardiovascular-Renal-Metabolic (CRM) Research: Studying the interplay between weight loss, heart function, and kidney health.
- Pharmacokinetics & ADME: Understanding how obesity-induced changes in drug transporters affect renal clearance.
- Evidence-Based Safety Profiling: We generate high-fidelity, comprehensive safety datasets that substantiate the renal tolerability of your lead compounds.
Advantages
Protheragen offers a unique competitive edge in the preclinical safety space:
Context-Specific Sensitivity
Standard healthy-animal models often hide nephrotoxic potential. By using DIO models, we expose toxicities that only manifest under the "double hit" of metabolic stress and drug exposure.
Precision Phenotyping
Our use of AI-assisted histopathology scoring ensures unbiased assessment of glomerulosclerosis and tubular atrophy, providing "published data" quality results.
Human-Centric In Vitro Models
Our 3D human kidney-on-a-chip models reduce species-specific bias, providing a more accurate prediction of human renal handling and toxicity.
Rapid Turnaround
Optimized protocols for UUO (unilateral ureteral obstruction) and accelerated CKD models allow for faster screening of drug efficacy vs. safety.
Expert Interpretation
Our senior biologists provide not just data, but mechanistic insights into why a compound is causing injury, enabling early-stage lead optimization.
Customer Review
Identifying Hidden Risks in Next-Gen Therapeutics
"Working with Protheragen was a turning point for our GLP-1/GIP co-agonist program. Their DIO models revealed a subtle but significant increase in KIM-1 levels that standard toxicology had missed. This allowed us to adjust our dosing strategy before moving into larger studies, potentially saving us millions in development costs. The depth of their biological expertise is unmatched."
Senior VP of Discovery, Mid-size Biotech
From Microphysiological Data to Strategic Confidence
"The team at Protheragen provided not just data, but a true partnership. Their 3D kidney-on-a-chip results gave us the mechanistic evidence we needed to satisfy internal safety committees. We are already planning our next project with them to look at the transition from obesity-induced hyperfiltration to early-stage CKD."
Head of Safety Pharmacology, Global Pharmaceutical Lead
Frequently Asked Questions
-
Why do I need a specialized nephrotoxicity study for anti-obesity drugs?
Obesity induces renal hyperfiltration and low-grade inflammation. A drug that is safe in a healthy kidney might be toxic in an obese kidney. Our service identifies these specific risks early.
-
Can you model both acute kidney injury (AKI) and chronic kidney disease (CKD)?
Yes, we offer models for both rapid onset toxicity and long-term fibrotic progression to cover the full spectrum of renal safety.
-
What species do you typically use for these studies?
We primarily utilize DIO rats and mice, as well as human-derived in vitro 3D kidney models for cross-species validation.
-
How do you measure GFR in your preclinical models?
We use gold-standard techniques such as Inulin and p-aminohippuric acid (PAH) clearance to provide precise measurements of filtration and plasma flow.
-
Are your urinary biomarkers more sensitive than serum creatinine?
Absolutely. Biomarkers like KIM-1 and NGAL can detect injury days before serum creatinine shows a significant rise.
-
Do you provide histopathology services?
Yes, our service includes full histopathological evaluation, including H&E, PAS, and Masson's trichrome staining with semi-quantitative grading by expert pathologists.
-
How does your 3D kidney-on-a-chip improve upon 2D cultures?
It incorporates fluid shear stress and 3D architecture, which are essential for the proper expression of drug transporters and more accurate toxicity responses.
-
Can you help with weight-based dosing calculations?
Our team has extensive experience in PK/PD modeling for obese subjects, ensuring that dosing in animal models accurately reflects potential human exposure.
-
What is the typical duration of a nephrotoxicity study?
Duration varies by model, ranging from short-term AKI studies (1-2 weeks) to long-term metabolic studies (8-16 weeks).
How to Contact Us
Protheragen provides high-end, context-specific nephrotoxicity studies for anti-obesity therapeutics. By combining translational metabolic models, advanced imaging, and sensitive biomarkers, we ensure your candidate drugs are rigorously tested for renal safety in the most clinically relevant environments.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

