Anti-Obesity Therapeutic Skin Toxicology Study Service
InquiryAs the global prevalence of obesity rises, the development of anti-obesity therapeutics has shifted from simple weight-loss agents to complex multi-mechanistic treatments, including GLP-1 receptor agonists, dual/triple agonists, and novel botanical formulations. However, these systemic interventions often carry significant risks of secondary dermatological complications. Preclinical evidence suggests that obesity itself alters skin physiology—increasing transepidermal water loss, impairing wound healing, and exacerbating radiation-induced or drug-induced skin toxicity.
Advanced Preclinical Safety: Toxicological Evaluation of Anti-Obesity Therapeutics on Dermal Integrity and Skin Health
Protheragen provides a specialized anti-obesity therapeutic skin toxicology study service designed to evaluate the safety profile of weight-management candidates specifically within the context of the obese phenotype. Our service focuses exclusively on preclinical evaluation, utilizing advanced animal models of diet-induced obesity (DIO) and genetic obesity to identify potential dermal irritation, sensitization, and systemic-to-skin toxicological manifestations before clinical entry. By integrating histopathological analysis with metabolic profiling, we help developers de-risk their pipelines and ensure that novel therapies do not compromise skin integrity.
Core Technologies
To ensure high translational accuracy, Protheragen employs a suite of advanced technologies tailored for metabolic and dermatological cross-analysis:
- Phenotype-Specific Animal Models
Utilization of high-fat diet (HFD) induced obesity models in Sprague-Dawley rats and New Zealand white rabbits, ensuring the "obese skin" environment—characterized by altered subcutaneous fat and inflammatory markers—is accurately represented.
- Multiplex Biomarker Profiling
Integration of adipokine profiling (e.g., adiponectin, leptin, and resistin) with skin-specific inflammatory markers (IL-6, TNF-α) to monitor the cross-talk between metabolic correction and dermal health.
- Precision Histopathology & Digital Imaging
Advanced staining techniques (including Oil Red O and Masson's trichrome) and AI-driven digital pathology to quantify changes in dermal thickness, lipid accumulation, and collagen density.
- Toxicokinetic (TK) Modeling
Specialized assessment of how systemically administered anti-obesity agents distribute to the skin tissue, providing critical data on local exposure versus systemic toxicity.
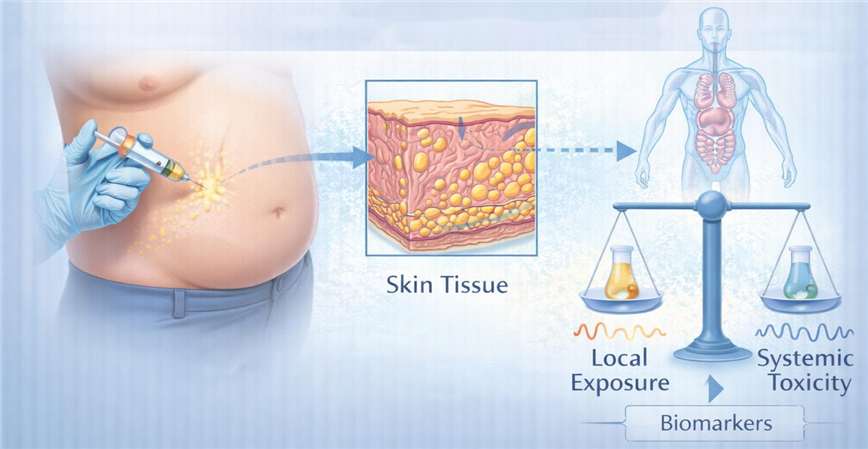
Solution Scope
Our expertise covers a wide range of anti-obesity modalities and their potential dermatological impacts:
- Systemic Safety Evaluation
Assessing skin-side effects of GLP-1, GIP, and Glucagon Receptor Agonists.
- Botanical & Synergistic Formulations
Toxicology of herbal extracts (e.g., Moringa, Curcuma) focusing on dermal sensitization and primary irritation.
- Specialized Radiodermatitis Interaction
Evaluating how anti-obesity agents influence skin sensitivity in models of radiation therapy (critical for oncological obesity research).
- Dermal Irritation & Structural Analysis
Rigorous preclinical evaluation and specialized testing to determine the irritation potential and tissue impact of novel compounds on skin integrity.
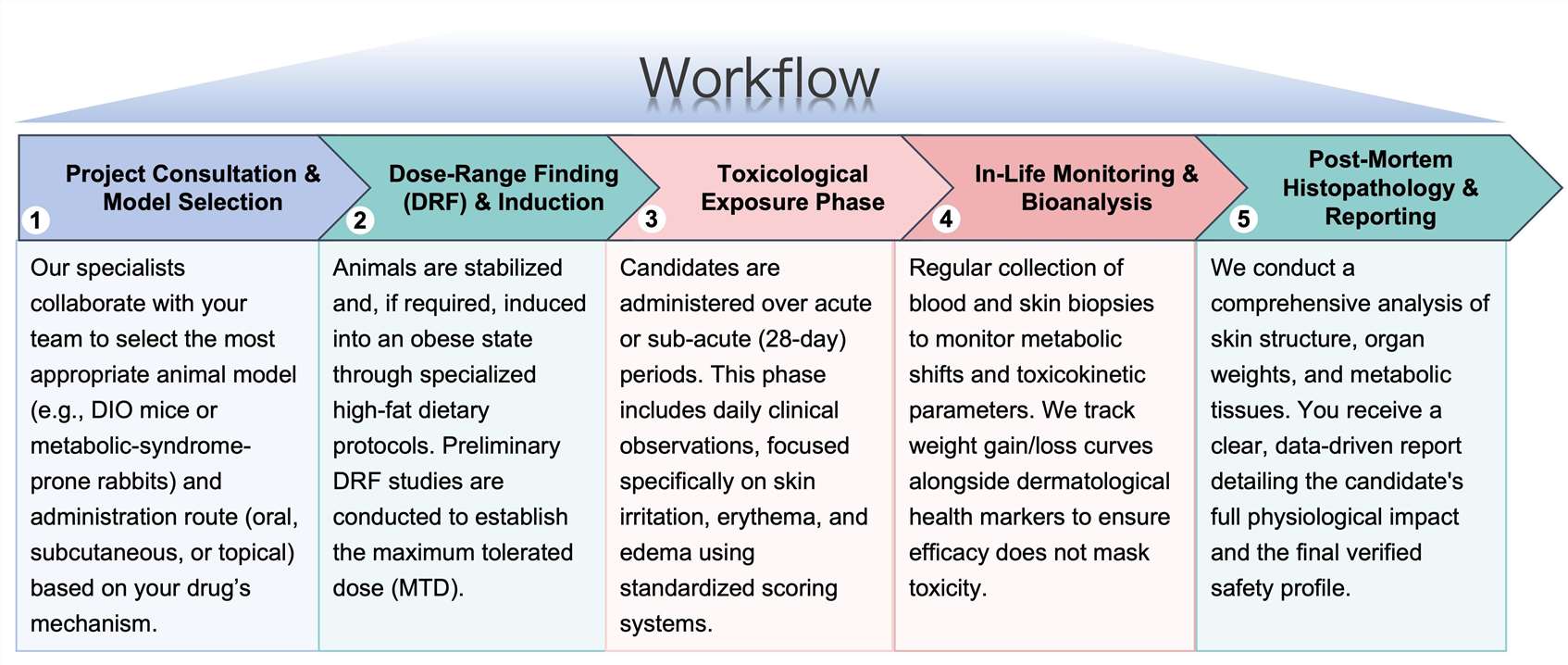
Workflow
Protheragen follows a rigorous, multi-stage preclinical workflow to provide comprehensive toxicological insights:
Contact Our Team for More Information and to Discuss Your Project.
Fields of Application
Our specialized toxicology assessment serves as a critical bridge between metabolic research and dermatological safety across multiple high-growth sectors. By understanding how anti-obesity candidates interact with skin physiology in a metabolic-compromised environment, we empower researchers to refine their formulations for specific therapeutic uses.
- Pharmaceutical Drug Discovery: For small molecules and peptides targeting metabolic pathways.
- Nutraceutical Development: Safety validation for "adipromin" type botanical weight-loss supplements.
- Oncology Support: Studying skin toxicity mitigation for obese breast cancer patients undergoing radiation.
- Dermacosmetic Research: Evaluating the skin-firming or lipid-reduction effects of topically applied anti-obesity agents.
Advantages
Choosing Protheragen provides your development program with unmatched specialized expertise:
Translational Relevance
Unlike standard toxicology services that use lean animals, we study your drug in the "target environment"—the obese animal. Published data indicate that BMI is a significant predictor of skin toxicity, making our obese-model approach essential for accurate risk assessment.
High Sensitivity Detection and Integrated Metabolic Insights
Our platforms are designed to detect "minimal irritation" and subtle histopathological changes that could lead to clinical-stage failures. We do not just look at the skin; we correlate skin health with adiponectin levels and lipid profiles, providing a holistic view of the drug's impact.
High-Standard Scientific Documentation
All studies are executed with rigorous precision to ensure clear, reproducible, and high-quality data that meet the most demanding internal research and development standards.
Customer Review
Validation of Safety Profiles for Novel Peptide Candidates
"Protheragen provided us with critical data on our lead GLP-1 analog. We were concerned about potential skin-site reactions seen in early iterations. Their DIO rat model showed that our formulation was non-irritating even in a pro-inflammatory obese state. The histopathology reports were of publication quality and gave our investors great confidence. We look forward to our next project together."
Senior Director of Toxicology, Biotech Startup
Strategic Insights into Metabolic and Dermal Interactivity
"The team at Protheragen understands the complexity of 'obese skin' better than any other CRO we've worked with. Their ability to correlate serum adiponectin levels with dermal collagen density provided a level of insight we didn't think was possible in a preclinical study. They are our go-to partner for metabolic safety."
Principal Scientist, Global Pharmaceutical Co
Frequently Asked Questions
-
Why is it necessary to use obese models for skin toxicology?
Obesity alters skin barrier function and inflammation. A drug that appears safe on lean skin may cause significant irritation or delayed healing in an obese subject.
-
Can you handle both oral and injectable anti-obesity agents?
Yes, our facility is equipped for various administration routes, including oral gavage, subcutaneous injection (common for GLP-1s), and topical application.
-
What species are available for these studies?
We primarily utilize Sprague-Dawley rats, New Zealand white rabbits, and C57BL/6 mice (including DIO and db/db models).
-
How do you measure skin irritation?
We use standardized scoring (e.g., Draize scale) for erythema and edema, supplemented by histopathological measurement of epidermal thickness.
-
What is the typical duration of a sub-acute study?
Our standard sub-acute skin toxicology protocol lasts 28 days, though this can be customized based on your needs.
-
Can you evaluate the impact of the drug on wound healing in obese models?
Yes, we offer specialized assays to monitor how anti-obesity treatments affect the repair of dermal layers.
-
Do you provide data on systemic absorption?
Yes, our toxicokinetics service measures the plasma concentration of the drug to correlate systemic exposure with local skin reactions.
-
Are botanical extracts tested differently?
Botanical formulations often require sensitization testing (e.g., LLNA) to ensure complex mixtures don't cause allergic contact dermatitis.
How to Contact Us
Protheragen offers a niche, high-expertise anti-obesity therapeutic skin toxicology study service that bridges the gap between metabolic research and dermatological safety. By utilizing disease-relevant animal models and sophisticated bioanalytical tools, we provide the evidence-based assurance needed to move your anti-obesity candidates forward.
Contact Protheragen for More Information and to Discuss Your Project
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

