Anti-Obesity Therapeutic Developmental and Reproductive Toxicology Study Service
InquiryThe global rise in obesity has catalyzed the development of next-generation anti-obesity medications (AOMs), including GLP-1 receptor agonists and multi-receptor peptides. However, the physiological intersection of obesity and reproductive health presents a complex challenge for drug developers. Maternal obesity is scientifically linked to chronic low-grade inflammation, altered placental nutrient transport, and developmental programming that predisposes offspring to metabolic disorders later in life. Consequently, evaluating how anti-obesity therapeutics affect fertility, embryo-fetal development, and postnatal health is a regulatory and ethical imperative.
Developmental and Reproductive Toxicology Study Service for Anti-Obesity Therapeutics
Protheragen provides specialized anti-obesity therapeutic, developmental, and reproductive toxicology (DART) study services. Our preclinical programs focus on addressing the unique biological challenges of metabolic drug development by providing high-resolution data on how new weight-management therapies interact with reproductive systems. We help sponsors identify potential reproductive risks—ranging from altered hypothalamic-pituitary-gonadal (HPG) axis function to teratogenicity—before clinical trials begin. By focusing on the metabolic context of the target indication, Protheragen ensures that your drug's safety profile is as robust as its efficacy.
Core Technologies
Protheragen utilizes a suite of high-precision analytical and biological platforms to assess the reproductive safety of anti-obesity candidates.
- Precision Biological Models
We utilize a range of specialized animal models, including both rodent and non-rodent systems, to gain a comprehensive understanding of how weight-management therapies distribute through the body and impact reproductive health. These models are selected for their biological relevance to human metabolic pathways, allowing us to track therapeutic exposure and observe physical development with high accuracy.
- High-Resolution Histopathology
Detailed examination of reproductive tissues, including follicular dynamics in ovaries and spermatogenesis stages in testes, often replaces or supplements initial fertility assessments.
- Endocrine & Inflammatory Profiling
Multiplex assays to measure hormones (LH, FSH, testosterone) and pro-inflammatory cytokines (TNF-α, IL-6) that are critical in the context of obesity-related inflammation.
- Placental Transfer & Function Analysis
Specialized techniques to determine if therapeutics cross the placental barrier and how they influence the "Barker hypothesis" of fetal programming.
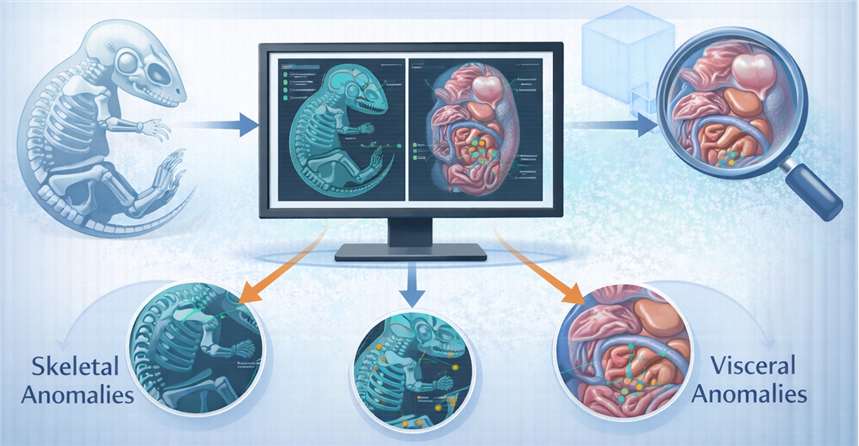
- Digital Morphometric Imaging
Advanced software for fetal skeletal and visceral examinations to identify subtle developmental anomalies or maturation delays.
Inquire About Our DART Study Capabilities for Metabolic Programs
Solution Scope
Our DART services are meticulously tailored to the specific pharmacological profiles of anti-obesity agents:
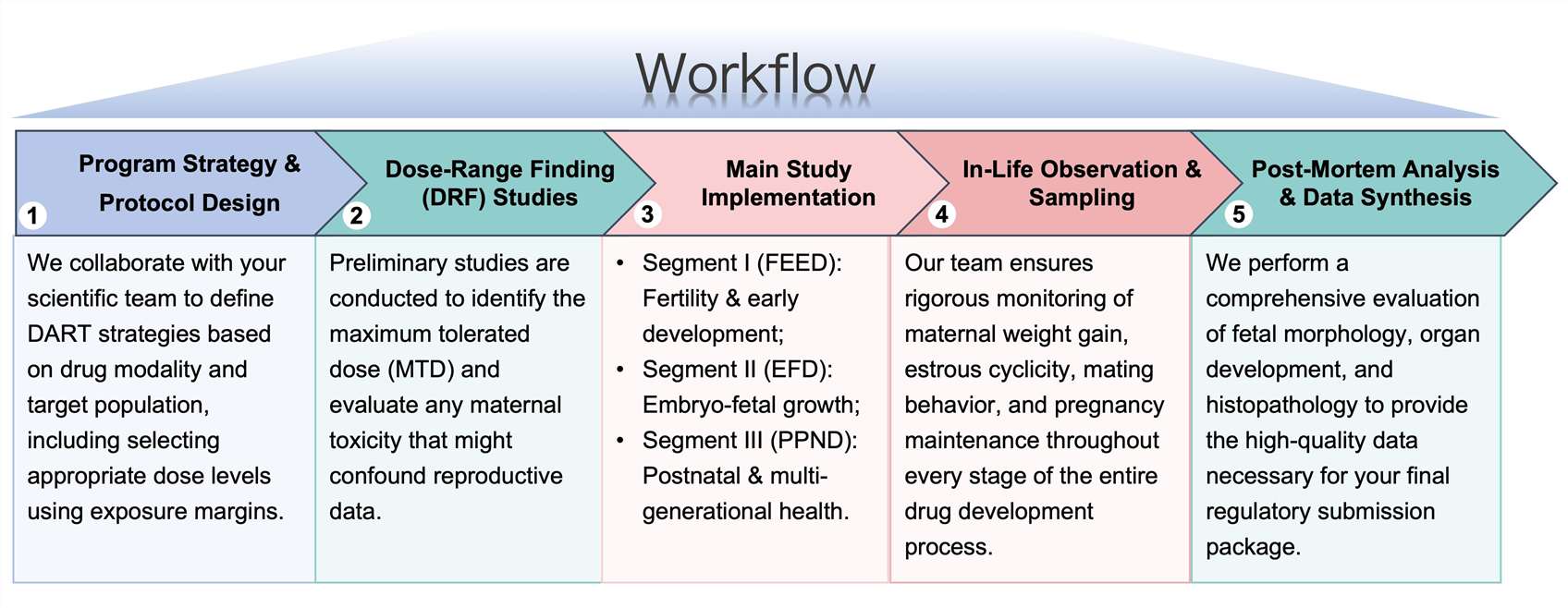
- Fertility and Early Embryonic Development (FEED)
Evaluation of mating performance, sperm quality, and early pregnancy maintenance in the context of rapid weight loss.
- Embryo-Fetal Development (EFD)
Assessing the risk of malformations (teratology) and growth restriction during the critical window of organogenesis.
- Pre- and Postnatal Development (PPND)
Studying the impact on the offspring's survival, growth, neurological development, and reproductive capacity in adulthood.
- Specialized Juvenile Toxicity
Targeted studies for pediatric indications, evaluating the drug's impact during neonatal to adolescent maturation.
- Alternative Assays
In vitro screening and Non-Mammalian Models for early-stage risk assessment and the 3Rs (reduction, refinement, replacement) compliance.
Workflow
Protheragen follows a rigorous, multi-stage workflow to ensure data integrity for every DART program.
Fields of Application
Our specialized DART study services provide essential safety insights across a diverse range of therapeutic classes and metabolic conditions, supporting the advancement of innovative treatments for weight management and its associated health complications.
- Peptide Therapeutics: Safety assessment for long-acting GLP-1 and multi-agonist peptides.
- Small Molecule AOMs: Evaluating the reproductive impact of oral anti-obesity candidates.
- PCOS & Infertility Treatments: Preclinical support for drugs targeting the metabolic drivers of polycystic ovary syndrome.
- Nutraceuticals & Bioactives: Specialized DART screening for metabolic health supplements.
- Pediatric Anti-Obesity Solutions: Juvenile toxicology to support labels for younger populations.
Advantages
Choosing Protheragen means partnering with a leader in metabolic safety assessment.
Expertise in Metabolic Modalities
Unlike generic CROs, we understand the specific nuances of GLP-1, GIP, and glucagon receptor agonists, including class-specific fetal toxicity signals noted in "published data".
Innovative Data Integration, Speed & Efficiency
We combine traditional toxicology with molecular biomarkers to provide a holistic view of the drug's impact on the reproductive axis. Our streamlined workflows and optimized study designs allow for faster transitions from preclinical results to clinical trial enablement.
Scientific Standardization and Global Quality
Our studies adhere to international frameworks, ensuring robust and reproducible data. We provide you with a quality-assured data package capable of standing up to the most demanding scientific scrutiny.
Empower Your Preclinical Program – Contact Our Experts for a Tailored DART Strategy
Publication Data
Title: What is known about the use of weight loss medication in women with overweight/obesity on fertility and reproductive health outcomes? A scoping review
Journal: Clinical Obesity, 2024
DOI: https://doi.org/10.1111/cob.12690
Summary: The recent scoping review highlights a critical knowledge gap in the reproductive safety of modern anti-obesity medications. While obesity is a known driver of subfertility, the rapid adoption of GLP-1 receptor agonists (GLP-1 RAs) like semaglutide and liraglutide has outpaced the available clinical data regarding their impact on pregnancy and fetal development. The review underscores that these medications are currently contraindicated during pregnancy due to potential teratogenicity—a risk largely identified through preclinical animal studies. As weight loss from these therapies can inadvertently trigger the resumption of ovulation, the risk of early-pregnancy exposure is significant, necessitating robust preclinical safety data.
Key Findings
- Critical Evidence Deficit: There is an absolute "absence of primary data" regarding the reproductive health effects of GLP-1 RAs in women of childbearing age who do not have PCOS or diabetes.
- Preclinical Toxicity Signals: Animal models for GLP-1 RAs have already demonstrated clear signals of reproductive toxicity, which serves as the primary basis for current clinical contraindications.
- Washout Requirements: Due to long half-lives and potential developmental risks, a 2-month washout period is currently recommended for semaglutide before planned conception.
- Ovulatory Rebound: Pharmacological weight loss can restore fertility in previously anovulatory women, increasing the likelihood of conception during treatment and highlighting the need for rigorous developmental and reproductive toxicology (DART) assessments.
- Orlistat Limitations: Existing trials for orlistat show that while weight and reproductive hormones improve, these changes do not consistently translate into increased live birth rates, suggesting that "weight loss alone" is not the only factor in reproductive success.
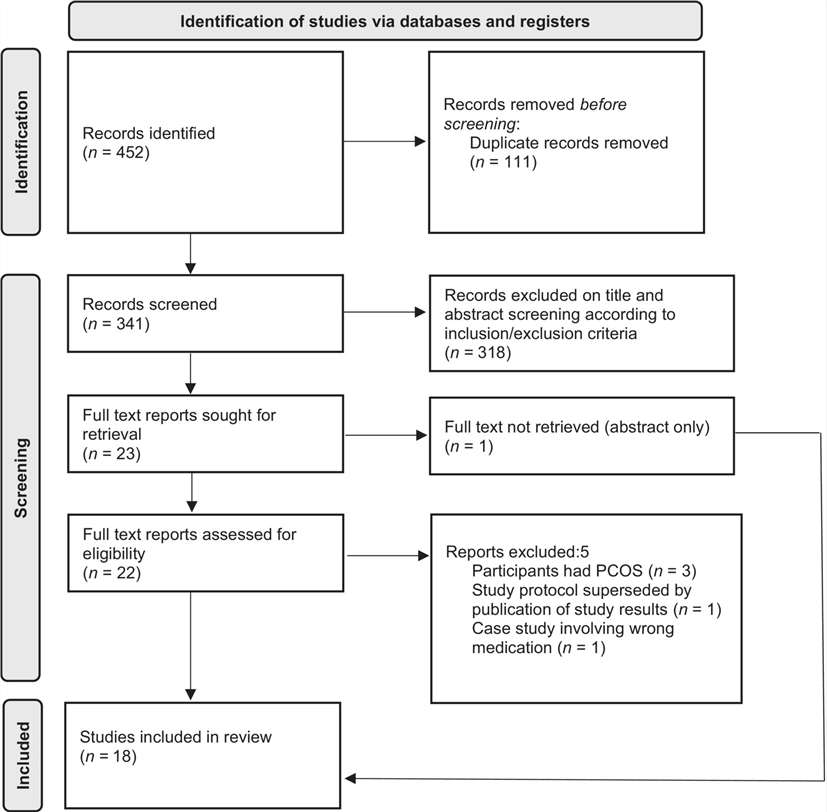 Fig.1 PRISMA flowchart. (Maslin, et al., 2024)
Fig.1 PRISMA flowchart. (Maslin, et al., 2024)
Customer Review
Resolving Complex Biological Signals in GLP-1 Development
"The team at Protheragen provided exceptional clarity on a complex fetal toxicity signal we encountered during early development. Their deep understanding of GLP-1 biology allowed our program to proceed to the next phase."
Senior VP of Toxicology, Biotech Firm
Enhancing Safety Profiles for Pediatric Metabolic Therapeutics
"Partnering with Protheragen was a game-changer for our pediatric anti-obesity candidate. Their expertise in juvenile toxicity and their ability to integrate hormonal profiling into standard DART protocols gave us the confidence we needed to validate our candidate's profile. We look forward to our next collaboration."
Chief Scientific Officer, Pharmaceutical Group
Frequently Asked Questions
-
Why is a DART study specifically important for anti-obesity drugs?
Obesity itself affects reproductive function; therefore, it is vital to distinguish whether reproductive changes are caused by the drug's chemical properties or the physiological effects of rapid weight loss.
-
How do you handle the confounder of weight loss during pregnancy?
We use carefully designed control groups and "published data" on maternal weight restriction to isolate the pharmacological effects of your candidate from the effects of reduced caloric intake.
-
Can you assess the impact of my drug on the offspring's metabolism?
Yes, our PPND studies can be extended to include metabolic profiling of the F1 generation to evaluate potential "developmental programming" effects.
-
Can histopathology replace a full fertility study?
In some early-stage programs, high-quality histopathology of the reproductive organs in repeat-dose studies can support initial clinical trials, as suggested by ICH guidelines.
-
What is the typical duration of a PPND study?
A full PPND study is the longest DART segment, as it follows the offspring through weaning and often into their own reproductive maturity, typically taking several months to complete.
-
How do you select dose levels for these studies?
We utilize PK/PD data to ensure dose levels provide a sufficient exposure margin over the intended human therapeutic dose while avoiding excessive maternal toxicity.
-
How can I get started with Protheragen?
Simply reach out through our inquiry portal to schedule a technical consultation with our DART specialists.
How to Contact Us
Protheragen is a premier provider of anti-obesity therapeutic, developmental, and reproductive toxicology study services. By combining advanced core technologies and a deep understanding of metabolic physiology, we offer the most comprehensive preclinical safety assessment available for the next generation of anti-obesity medications. Our focus remains on providing high-quality data that mitigates risk and accelerates your drug's path to the clinic.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Maslin, K.; et al. What is known about the use of weight loss medication in women with overweight/obesity on fertility and reproductive health outcomes? A scoping review. Clinical Obesity. 2024, 14(6): e12690. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

