Anti-Obesity Therapeutic Toxicogenomics Study Service
InquiryAs the global obesity epidemic continues to drive the demand for more sophisticated metabolic interventions, the pharmaceutical industry is shifting from single-target ligands to complex, multimodal pharmacotherapies. However, with increased complexity comes a heightened risk of off-target effects and metabolic disruptions.
Transcriptomics-Driven Safety Profiling for Anti-Obesity Therapeutics
Protheragen provides a specialized anti-obesity therapeutic toxicogenomics study service, designed to de-risk drug development at the preclinical stage. By integrating high-resolution transcriptomics with advanced cell-based models, we help researchers identify the molecular "genetic fingerprints" of anti-obesity candidates, ensuring that potential safety liabilities—such as endocrine disruption or maladaptive adipogenesis—are identified long before clinical trials.
Core Technologies
Our service leverages a multi-dimensional technology stack to provide a comprehensive view of how your therapy affects metabolic pathways:
- Transcriptomic Fingerprinting (RNA-Seq)
We utilize high-depth RNA sequencing to monitor the expression of thousands of genes simultaneously, identifying differentially expressed genes (DEGs) that serve as biomarkers for both efficacy and toxicity.
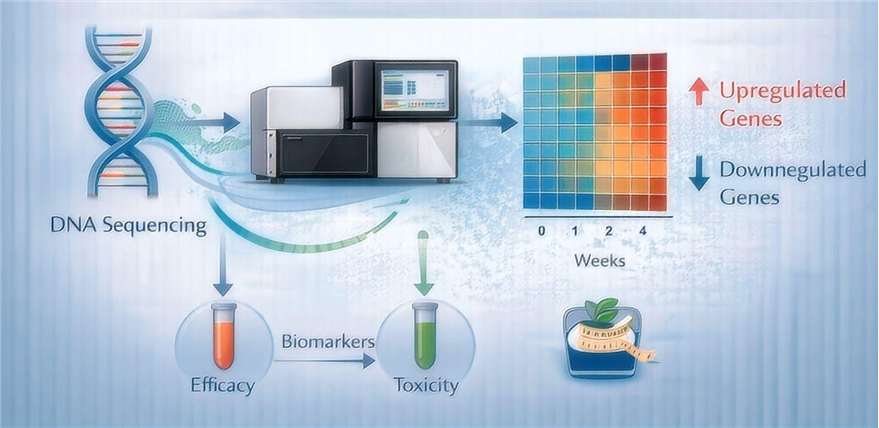
- 3T3-L1 Adipogenesis Platform
A standardized, highly reproducible in vitro model for screening obesogenic and anti-obesity compounds. This platform allows for the precise measurement of lipid accumulation and the expression of key adipogenic proteins like PPARγ, C/EBPα, and FABP4.
- Toxicogenomic Predictive Algorithms
Protheragen employs proprietary bioinformatics pipelines to translate gene expression data into predictive safety profiles, identifying risks related to hepatotoxicity, neurotoxicity, and metabolic reprogramming.
- Multimodal Metabolic Profiling
For next-generation agonists (e.g., GLP-1/GIP/GCG analogs), we integrate transcriptomics with biochemical assays to map the cross-talk between gut-brain and adipose-muscle signaling axes.
Solution Scope
Our specialized solutions encompass the full spectrum of preclinical molecular assessment, providing granular insights into how your candidates influence lipid metabolism and cellular homeostasis at the genomic level.
- Early-Stage Lead Prioritization
Rank multiple candidates based on their toxicogenomic safety profiles.
- Mechanistic De-risking
Elucidate the underlying molecular pathways of observed adverse effects in adipocytes or myocytes.
- Biomarker Discovery
Identify specific genetic markers that can be used in later-stage preclinical animal studies to monitor response.
- Environmental Obesogen Screening
Evaluate whether your compound or its metabolites possess unintended endocrine-disrupting properties that could promote adiposity.
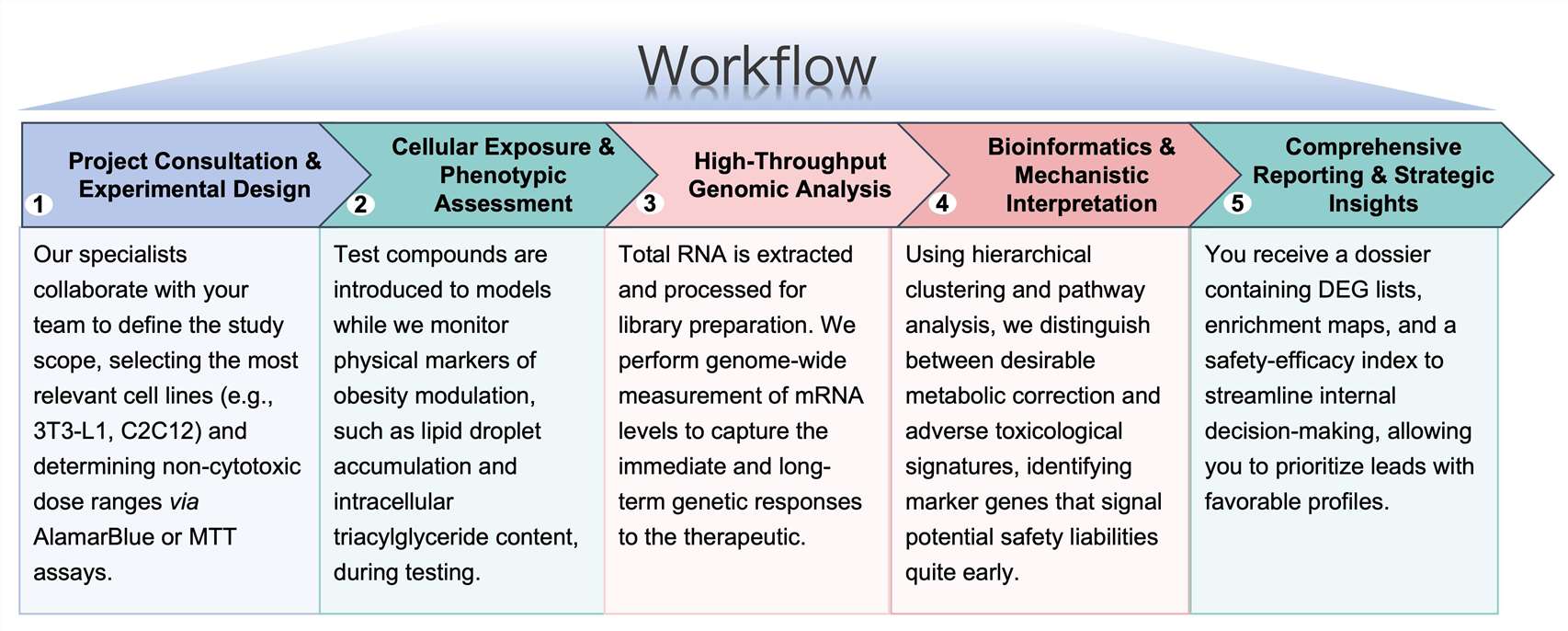
Workflow
Protheragen ensures a seamless transition from compound delivery to data-driven insights through a structured 5-stage process:
Click Here to Submit an Inquiry
Fields of Application
The versatility of the Protheragen toxicogenomics platform allows it to be seamlessly integrated across diverse therapeutic research areas, from metabolic disease modeling to the safety evaluation of novel endocrine modulators.
- Small Molecule Discovery: Assessing oral incretin mimetics and non-peptide agonists.
- Biologics & Monoclonal Antibodies: Evaluating muscle-preserving agents (e.g., myostatin/activin antagonists) for off-target genetic effects.
- RNA Therapeutics: Monitoring the specificity and systemic metabolic impact of ASOs or siRNA-based anti-obesity treatments.
- Nutraceuticals: Screening natural compounds for adipogenic inhibition and safety.
Advantages
Protheragen stands at the intersection of biology and data science. Our anti-obesity toxicogenomics service offers:
Precision Safety Profiling
Unlike traditional toxicity assays that only detect cell death, our toxicogenomic approach identifies "silent" stressors and metabolic reprogramming that could lead to long-term safety issues.
Standardized Reproducibility
We use strictly controlled assay conditions (standardized serum batches and passage numbers) to eliminate the variability common in 3T3-L1 models.
Accelerated Timelines and Published Data Correlation
Get high-resolution, decision-ready data in just 8–12 weeks, significantly faster than traditional chronic in vivo toxicity studies. Our internal databases correlate genomic signatures with published data on known reference obesogens (e.g., rosiglitazone, tributyltin) to provide a comparative benchmark for your candidate.
Contact Our Specialist Team Today to Request a Custom Study Design for Your Anti-Obesity Candidate.
Publication Data
Title: Pharmacogenomics of Anti-Obesity Drugs: A Bioinformatics Approach
Journal: Research Square, 2025
DOI: https://doi.org/10.21203/rs.3.rs-6370544/v1
Summary: This article explores the pharmacogenomics of three major anti-obesity drugs (liraglutide, semaglutide, tirzepatide) using a bioinformatics approach, aiming to address interindividual variability in treatment response. By integrating data from public databases (PharmGKB, GWAS Catalog, GTEx, STRING), the study analyzes genetic variants, gene expression patterns, protein-protein interactions, and metabolic pathways. It employs machine learning models to predict BMI reduction efficacy and maps drug-specific signaling pathways via KEGG. The research identifies clinically actionable genetic biomarkers in receptor genes (GLP1R, GIPR) and metabolic enzymes (CYP3A4, CYP2C8), providing a scientific basis for personalized obesity treatment strategies that optimize drug efficacy and safety.
Key Findings
- Core Genetic Variants Modulating Drug Response: Variants in GLP1R (rs6923761, rs1030542) reduce receptor binding affinity and adipose tissue expression, impairing GLP-1RA efficacy; GIPR rs10423928 modulates tirzepatide's incretin effect via cAMP signaling; CYP3A422 (rs35599367) slows drug metabolism, increasing exposure and adverse effects.
- Tissue-Specific Gene Expression Patterns: GTEx data shows GLP1R is primarily expressed in subcutaneous adipose tissue (TPM ~1.2), GIPR is moderately expressed in both subcutaneous and visceral adipose tissue, and DPP4 has high adipose tissue expression (TPM ~15.3), correlating with drug mechanism and metabolism.
- Drug-Specific Pathway Activation: Liraglutide and semaglutide act through GLP1R, engaging insulin signaling, cAMP signaling, and PI3K-Akt pathways; semaglutide additionally modulates hypothalamic appetite circuits; tirzepatide's dual GIPR/GLP1R agonism activates incretin signaling, CNS appetite circuits, and adipocyte remodeling networks.
- Machine Learning-Predicted Efficacy: The models forecast genotype-dependent BMI reductions: 8.5% for liraglutide, 14.2% for semaglutide, and 16.8% for tirzepatide, with efficacy linked to specific variants and tissue expression levels of target genes (e.g., GLP1R, MC4R, ADIPOQ).
- Clinically Actionable Biomarkers: 38% of identified variants (primarily in CYP450 enzymes and GLP1R/GIPR) are classified as PharmGKB Level 1/2 (clinically actionable), supporting personalized dose adjustment and drug selection to minimize adverse effects and maximize therapeutic outcomes.
Customer Review
Early Risk Mitigation: Uncovering Hidden Molecular Stressors
"The team at Protheragen provided us with a level of molecular insight we couldn't find elsewhere. Their toxicogenomics report identified a subtle but significant upregulation of pro-inflammatory pathways in our lead anti-obesity candidate that standard assays missed. This allowed us to pivot our chemical optimization early, saving us months of wasted effort."
Senior Director of Discovery, Mid-sized Biotech
Data-Driven Confidence: Accelerating Multimodal Agonist Pipelines
"Protheragen's standardized 3T3-L1 platform was exactly what we needed for our screening program. The reproducibility of their data gave our stakeholders the confidence to advance our multimodal agonist into the next phase. We look forward to continuing our partnership as our pipeline expands."
Head of Metabolic Research, Pharmaceutical Client
Frequently Asked Questions
-
Why use toxicogenomics instead of standard toxicity assays?
Standard assays often only detect terminal events like cell death. Toxicogenomics detects subtle changes in gene expression that precede physical damage, allowing for the identification of safety risks like endocrine disruption or liver stress much earlier.
-
Which cell models are most effective for anti-obesity studies?
The 3T3-L1 pre-adipocyte line is the gold standard for studying adipogenesis. We also use C2C12 myoblasts to evaluate effects on muscle mass preservation, which is critical for new multimodal therapies.
-
Can this service predict human response?
While these are preclinical studies, toxicogenomics identifies conserved metabolic pathways. By comparing your candidate's signature to human-relevant biomarkers, we provide a high level of translational confidence.
-
How do you handle the high sensitivity of 3T3-L1 cells?
We maintain strict protocols regarding plasticware brands, serum batches, and cell density to ensure that any observed changes are due to your compound, not environmental noise.
-
What is the turnaround time for a full study?
Typically, from the compound receipt to the final report, the process takes 8 to 12 weeks.
-
Can you help identify the mechanism of action (MOA)?
Yes. By analyzing which pathways (e.g., PPAR signaling, lipid metabolism, inflammation) are upregulated or downregulated, we can confirm the intended MOA or discover unexpected secondary effects.
-
Is your platform suitable for combination therapy studies?
Absolutely. We can evaluate synergistic effects and potential cumulative toxicity when two or more compounds are administered simultaneously.
-
What data format will I receive?
You will receive raw sequencing data, processed DEG lists, and a comprehensive narrative report with visualization tools like heatmaps and volcano plots.
How to Contact Us
Protheragen is your partner in navigating the complexities of anti-obesity drug development. Our expertise in toxicogenomics ensures that your path to the clinic is paved with robust, high-resolution scientific data.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Andrade, L. J.; et al. Pharmacogenomics of Anti-Obesity Drugs: A Bioinformatics Approach. Research Square. 2025, PREPRINT (Version 1). (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

