Cerebral Organoid Modeling Service for Obesity Research
InquiryObesity is no longer viewed merely as a metabolic storage disorder but as a complex neurological condition driven by the central nervous system (CNS). At the heart of this pathology lies the hypothalamus, the brain's "control center" for energy homeostasis, appetite regulation, and metabolic rate. Traditional 2D cell cultures and animal models often fail to capture the unique cytoarchitecture and genetic nuances of the human brain, leading to high failure rates in preclinical drug development.
Precision Cerebral Organoid Modeling: Pioneering Preclinical Solutions for Obesity Research
Protheragen provides a revolutionary cerebral organoid modeling service specifically optimized for obesity research. By utilizing human induced pluripotent stem cells (iPSCs), we generate 3D hypothalamic and cerebral organoids that faithfully recapitulate the human neural circuits involved in hunger and satiety. Our platform offers an unprecedented window into the neurobiological drivers of obesity, enabling researchers to study human-specific responses to metabolic hormones and potential therapeutics in a physiologically relevant, preclinical environment.
Core Technologies
Our service leverages a suite of cutting-edge technologies to ensure the highest fidelity in disease modeling:
- Hypothalamic-Specific Patterning
We employ guided differentiation protocols using precise concentrations of morphogens (such as SHH and BMP inhibitors) to specify progenitors into arcuate nucleus (ARC) and paraventricular nucleus (PVN) identities.
- iPSC-Derived Patient Modeling
We generate organoids from donor-specific iPSC lines, including those from super-obese individuals (BMI ≥ 50), to study polygenic risk factors and personalized metabolic responses.
- Neuro-Metabolic Sensing Assays
Integration of bioluminescence-based metabolic profiling and calcium imaging to monitor real-time neuronal firing in response to appetite-regulating hormones like Leptin and Ghrelin.
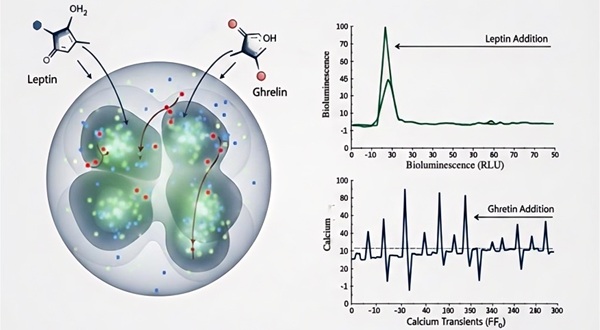
- High-Throughput 3D Architecture
Our scaffold-free, hanging-droplet, and microfluidic systems ensure uniform organoid size and enhanced nutrient exchange, crucial for maintaining long-term functional viability.
Solution Scope
Our modeling services are comprehensive, covering various aspects of metabolic neurobiology:
- Hypothalamic Circuit Modeling
Specifically targeting the leptin-melanocortin signaling pathway.
- Blood-Brain Barrier (BBB) Integration
Optional co-culture models to study the transport of metabolic drugs into the CNS.
- Inflammation & ER Stress Profiling
Assessment of neuroinflammation and endoplasmic reticulum stress induced by high-fat environments or obesogenic signatures.
- High-Content Drug Screening
Scalable platforms for evaluating the efficacy and toxicity of anti-obesity candidates before they reach clinical stages.
Workflow
Protheragen follows a rigorous, standardized workflow to ensure reproducible and high-quality data for every preclinical project:
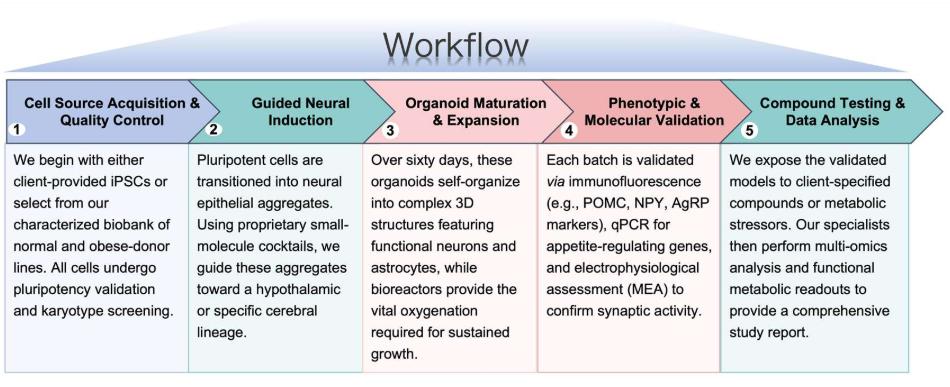
Fields of Application
The utility of Protheragen's cerebral and hypothalamic organoids extends across the entire spectrum of metabolic neuroscience, providing a high-fidelity human platform where traditional animal models often reach their translational limits. By recapitulating the intricate "gut-brain axis" and the central regulation of energy homeostasis, our models serve as essential tools for the following specialized research areas:
- Drug Discovery & Target Validation
Identifying and validating novel molecular targets within the human melanocortin-4 receptor (MC4R) pathway and other appetite-suppressing circuits.
- Precision Medicine & Genotype-Phenotype Correlation
Evaluating how specific genetic variants (e.g., FTO, POMC, or LEPR mutations) influence individual responses to anti-obesity medications like GLP-1 and GIP receptor agonists.
- Neurodevelopmental Origins of Obesity
Investigating the impact of maternal metabolic health and environmental stressors on the formation of fetal hypothalamic circuits.
- Neuroinflammation & Metabolic Stress Research
Mapping the cellular mechanisms by which chronic high-fat environments trigger hypothalamic inflammation and ER stress, leading to leptin resistance.
- Preclinical Safety & Toxicology
Assessing the potential neurotoxic effects of systemic metabolic regulators on human neural tissue before advancing to further stages of development.
Request a Project Consultation with Our Scientists Today.
Advantages
Partnering with Protheragen offers distinct advantages for your preclinical programs:
Superior Human Relevance
Unlike rodent models, our human organoids express human-specific receptors (e.g., GLP-1R and GIPR) in the correct anatomical context, ensuring better translatability of results.
Recapitulation of Obese Phenotypes
Published data indicate that organoids derived from super-obese donors retain disease-specific signatures, such as dysregulated ghrelin-leptin signaling and altered axonal guidance.
Consistent Scalability and Integrated Multi-Omics
We do not just provide cells; we provide insights. Our service includes detailed transcriptomic and metabolic profiling to identify novel mechanism-of-action (MoA) data. Our automated culture systems minimize batch-to-batch variability, providing the statistical power needed for robust drug screening.
Publication Data
Title: Modeling maternal cholesterol exposure reveals a reduction of neural progenitor proliferation using human cerebral organoids
Journal: Life Medicine, 2023
DOI: https://doi.org/10.1093/lifemedi/lnac034
Summary: This paper models maternal high cholesterol using human pluripotent stem cell–derived cerebral organoids. Elevating cholesterol in culture produced enlarged endosomes, strongest in ventricular zone–like regions. Cholesterol exposure suppressed neural progenitor proliferation and increased cell-cycle exit, helping explain reduced organoid expansion, while apoptosis or senescence was not the main driver. Transcriptomic and single-cell analyses supported disrupted cell-cycle programs and abnormal cholesterol metabolism, alongside a shift toward earlier neurogenesis. Comparisons with published datasets showed transcriptional similarity to autism spectrum disorder organoids, suggesting a plausible link between prenatal cholesterol environment and neurodevelopmental risk mechanisms.
Key Findings
- A human-relevant prenatal exposure model was established. The study built an hPSC-derived cerebral organoid system to mimic maternal high-cholesterol exposure during an early-gestation-like window, enabling mechanistic testing in a human cellular context.
- Cholesterol exposure triggers endosomal swelling in neural progenitor niches. Enlarged endosomes were detected, particularly in VZ-like regions and within RGCs, implicating altered endosomal maturation/trafficking as an early cellular phenotype.
- Neural progenitor proliferation is suppressed—without a major apoptosis/senescence signature. Multiple readouts pointed to lower proliferation and increased cell-cycle exit, while assays for apoptosis/senescence did not explain the reduced growth, strengthening the conclusion that proliferation impairment is central.
- Neurogenesis is pushed forward prematurely. Cholesterol-exposed organoids exhibited earlier differentiation toward neurons, reflected by increased neuronal markers and progenitor redistribution consistent with accelerated neurogenic output.
- Single-cell transcriptomics links cholesterol exposure to altered cell-cycle programs and sterol regulation. scRNA-seq revealed broad differential expression and enrichment of pathways tied to mitotic transition/cell-cycle regulation in progenitor populations, alongside shifts in genes involved in cholesterol handling.
- Molecular overlap points toward ASD-relevant transcriptional patterns. Differentially expressed genes overlapped most strongly with ASD-associated gene sets, and correlation analyses suggested similarity to published ASD organoid transcriptomes—supporting a biologically plausible exposure-to-risk connection.
- A practical translational angle: an assayable, early-warning phenotype. The combination of reduced progenitor proliferation + premature differentiation provides a measurable phenotype that could be useful for hypothesis-driven screening of maternal metabolic exposures in human models.
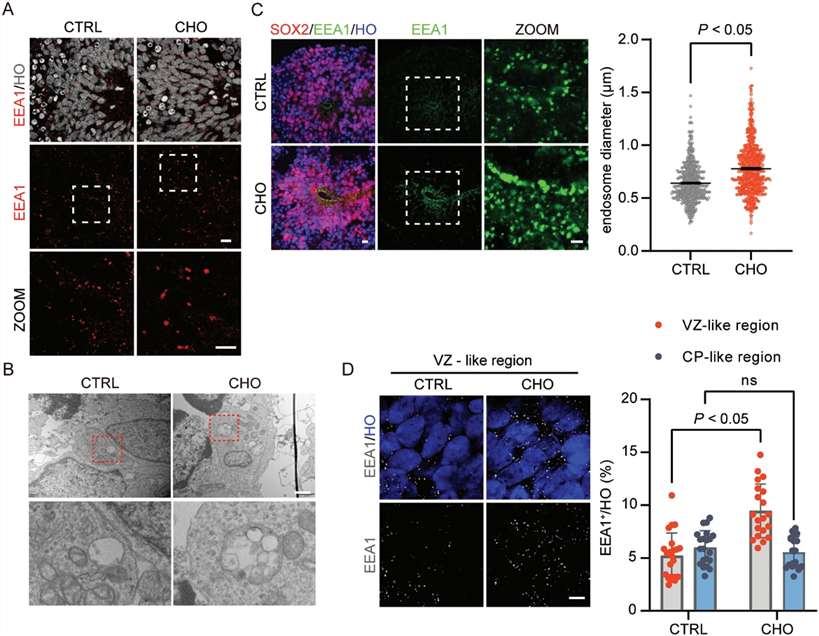 Fig. 1 High Cholesterol drives endosomal swelling in ventricular zone–like regions of human brain organoids. (Fan, et al., 2023)
Fig. 1 High Cholesterol drives endosomal swelling in ventricular zone–like regions of human brain organoids. (Fan, et al., 2023)
Customer Review
Bridging the Translational Gap: Identifying Human-Specific Responses in Target Validation
"Working with Protheragen transformed our approach to target validation. Their hypothalamic organoids allowed us to observe a human-specific response to our lead compound that was completely absent in our previous mouse studies. The team's expertise in neuro-metabolic signaling is unmatched."
Director of Pharmacology, Mid-sized Biotech
Scalability Meets Precision: De-risking the Metabolic Pipeline via High-Throughput 3D Screening
"The quality and reproducibility of the organoids provided by Protheragen were exceptional. We were able to screen 50+ compounds in a human 3D environment, which significantly de-risked our pipeline before moving toward the next phase of development. Their detailed validation reports are a gold standard."
Principal Scientist, Metabolic Disease Research Institute
Frequently Asked Questions
-
Why use cerebral organoids instead of mice for obesity research?
Human hypothalamic circuitry differs significantly from that of mice, particularly in receptor distribution and neuropeptide expression. Organoids provide a human genetic background.
-
Can your organoids respond to satiety hormones like leptin?
Yes, our models are validated to show increased p-STAT3 and p-AKT signaling in response to insulin and leptin.
-
Do the organoids contain specific appetite-regulating neurons?
Yes, our hypothalamic organoids contain POMC (anorexigenic) and AgRP/NPY (orexigenic) neuron populations.
-
What is the typical duration of a modeling project?
Standard differentiation and validation usually take 10–12 weeks, depending on the complexity of the readout.
-
Can we provide our own patient-derived iPSCs?
Absolutely. Protheragen specializes in custom differentiation of client-provided lines.
-
Are these models suitable for high-throughput screening?
Yes, we utilize 96-well format systems optimized for automated imaging and supernatant analysis.
-
Do you provide clinical services?
No, Protheragen is strictly a preclinical contract research provider.
-
How do you ensure the maturity of the neurons?
We use MAP2/NeuN staining and multi-electrode array (MEA) recording to confirm mature synaptic function.
-
Can you model neuroinflammation associated with obesity?
Yes, we can monitor cytokine release (IL-6, CCL2) and glial activation in response to metabolic stressors.
-
What deliverables are provided at the end of a study?
Clients receive a comprehensive report including raw data, high-resolution microscopy images, and statistical analysis of all functional assays.
How to Contact Us
Protheragen is dedicated to empowering the next generation of obesity therapeutics through precision modeling. Our expertise in iPSC technology and 3D biology provides the clarity you need to move your preclinical projects forward with confidence.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Fan, P.; et al. Modeling Maternal Cholesterol Exposure Reveals a Reduction of Neural Progenitor Proliferation Using Human Cerebral Organoids. Life Medicine. 2023, 2(2): lnac034. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

