Adipose Tissue Organoid Modeling Service for Obesity Research
InquiryObesity remains one of the most significant global health challenges, yet drug development is often hindered by the poor translational predictability of Animal Models. At Protheragen, we provide a sophisticated adipose tissue organoid modeling service that utilizes human-derived cells to create physiologically relevant 3D microenvironments.
Automated Adipose Organoid Systems for High-Throughput Metabolic Analysis
Our organoids accurately recapitulate the architectural and functional complexity of human white, brown, and beige adipose tissues. By integrating stromal vascular fraction (SVF) cells, adipocytes, and essential extracellular matrix (ECM) components, Protheragen offers a high-fidelity platform for studying adipocyte hypertrophy, insulin resistance, and inflammatory signaling in a controlled in vitro setting.
Core Technologies
Our service leverages a suite of proprietary and advanced technologies to ensure the highest level of physiological relevance:
- Xeno-Free Hydrogel Scaffolding
We utilize advanced, animal-free hydrogel matrices that support apical-out polarity and mimic the mechanical properties of human fat depots.
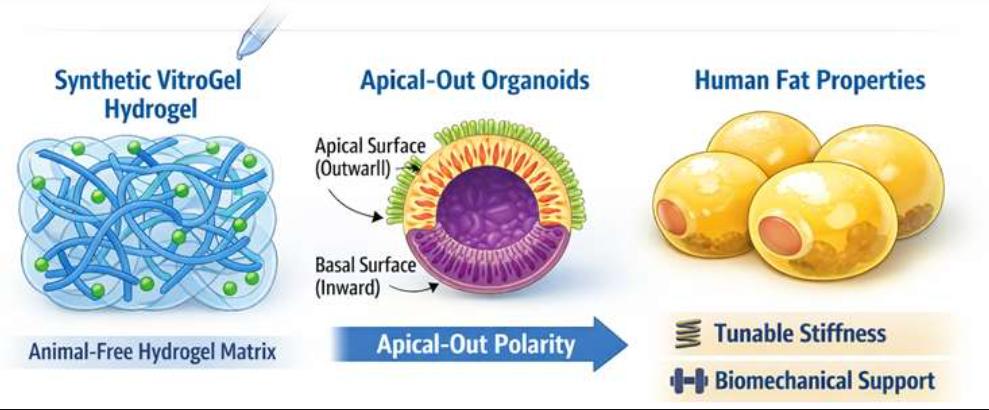
- Magnetic Bioprinting & Stabilization
To overcome the natural buoyancy of mature, lipid-laden adipocytes, we employ magnetic bioprinting. This technology allows for the precise positioning and retention of organoids during long-term culture and high-throughput imaging.
- Multi-Cellular Co-Culture Systems
Our models incorporate the stromal vascular fraction (SVF), including preadipocytes, endothelial cells, and immune cells (macrophages), to simulate the chronic low-grade inflammation characteristic of obesity.
- Automated High-Content Imaging
We utilize deep-learning-based 3D imaging platforms to quantify lipid droplet size, organoid volume, and metabolic activity markers without disrupting the 3D architecture.
Solution Scope
Protheragen offers a modular and highly customizable suite of modeling solutions designed to capture the functional diversity of human adipose depots, from standard metabolic profiling to complex disease-state simulations.
- White Adipose Organoid (WAO) Modeling
Focused on energy storage, hypertrophy, and the development of insulin resistance.
- Thermogenic Brown/Beige Modeling
Screening for "browning" agents that increase UCP1 expression and enhance energy expenditure.
- Inflammation & Fibrosis Assays
Modeling the recruitment of M1 macrophages and the deposition of collagen within the adipose niche.
- Lipid Metabolism Profiling
Detailed quantification of lipogenesis, lipolysis, and fatty acid oxidation.
- Custom Disease Models
Tailored development of organoids representing specific comorbidities like NASH/MASH-related adipose dysfunction.
Partner with Protheragen to Gain the Metabolic Insights You Need.
Workflow
Protheragen follows a rigorous, standardized workflow to ensure reproducible and high-quality data for every project:
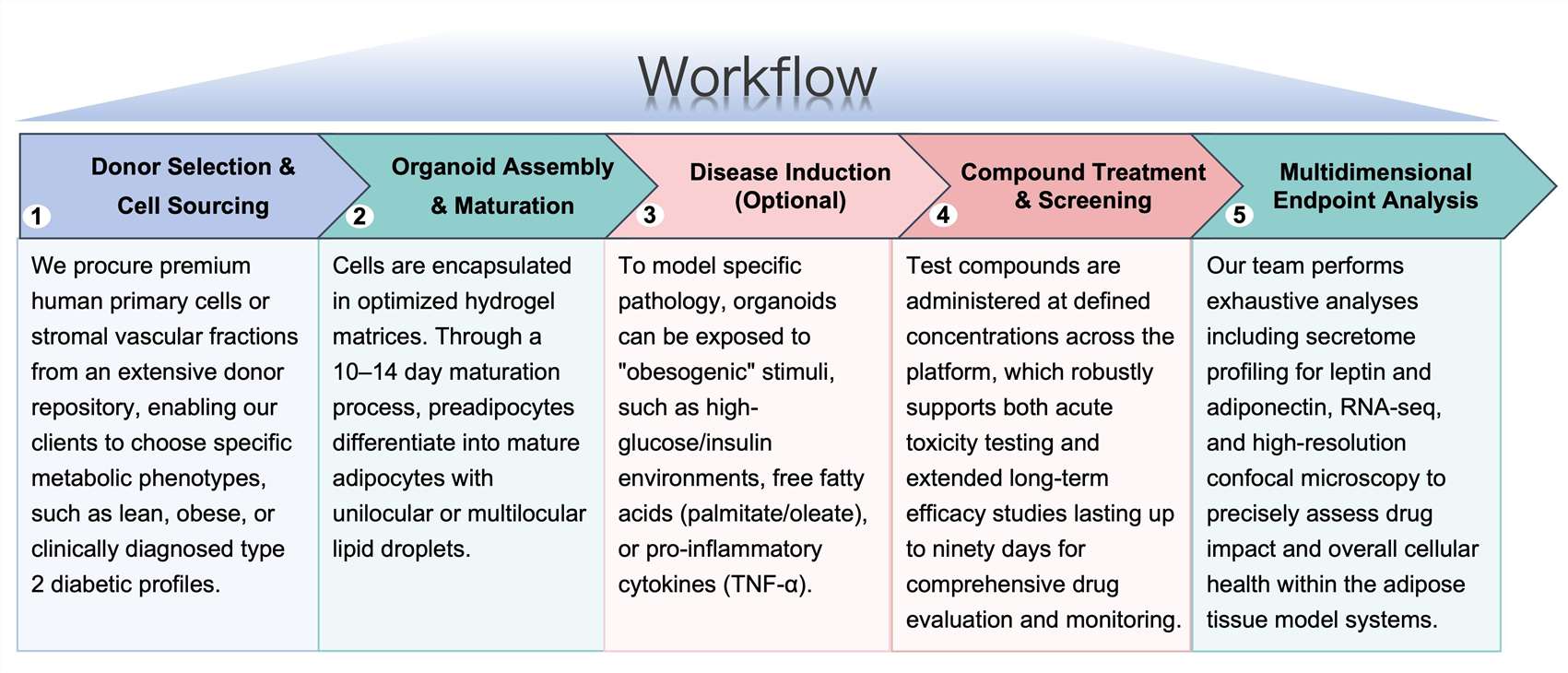
Fields of Application
By effectively bridging the gap between traditional in vitro cultures and complex human physiology, the Protheragen adipose organoid platform serves as a versatile engine for innovation across a spectrum of metabolic research domains.
- Anti-Obesity Drug Discovery: Identifying small molecules or biologics that reduce lipid accumulation or enhance lipolysis.
- Metabolic Syndrome Research: Investigating the crosstalk between adipose tissue and systemic insulin sensitivity.
- Nutraceutical Testing: Validating the efficacy of natural compounds in modulating fat cell metabolism.
- Endocrine Disruptor Screening: Assessing the impact of environmental chemicals on adipogenesis and hormone secretion.
Advantages
Protheragen's adipose organoid platform offers distinct benefits over traditional preclinical methods:
Human-Centric Translation and Published Data Support
Eliminate species-specific metabolic differences inherent in rodent models by using 100% human-derived cellular components. Our methodologies are backed by internal validation studies and published data demonstrating a 0.85 correlation with clinical adipokine profiles.
Long-Term Viability and Scaffold-Free Options
Our optimized culture conditions maintain functional adipocytes for over 3 months, enabling the study of chronic drug effects and metabolic aging. For specialized mechanics, we offer scaffold-free magnetic assembly that prevents the "floating" issues common in standard fat cell cultures.
High-Throughput Compatibility
Our system is scaled for 96-well and 384-well formats, making it ideal for primary library screening. This allows researchers to move directly from large-scale screening to deep-dive analysis, significantly reducing the time-to-lead for large therapeutic portfolios.
Publication Data
Title: Breaking barriers in obesity research: 3D models of dysfunctional adipose tissue
Journal: Trends in Biotechnology, 2025
DOI: https://doi.org/10.1016/j.tibtech.2024.09.017
Summary: This paper focuses on the pivotal role of adipose tissue (AT) dysfunction in obesity-related comorbidities and the transformative potential of 3D scaffold-based in vitro models in obesity research. It highlights the limitations of traditional 2D in vitro models (lack of tissue complexity) and in vivo animal models (species-specific differences, ethical issues) and details how 3D scaffold-based models overcome these by mimicking AT's extracellular matrix (ECM), mechanical properties, cellular composition (e.g., adipocytes, immune cells), and vascularization. The review covers engineering strategies for these models (biomaterial selection, tuning stiffness/deformation, inducing adipocyte hypertrophy, co-culturing immune cells, and integrating vasculature) and their applications in studying AT pathophysiology, drug screening, and personalized medicine. It also discusses future prospects, including multi-organ chip integration and contributions to tissue engineering and cultivated meat.
Key Findings
- AT Dysfunction Mechanisms: Obesity drives AT expansion (hypertrophy/hyperplasia), leading to hypoxia, immune cell infiltration (macrophage "crown-like" structures), inflammation (via adipokines like TNF-α, IL-6), and fibrosis, which collectively cause insulin resistance and metabolic complications.
- Limitations of Traditional Models: 2D models fail to replicate 3D tissue architecture and cell–ECM interactions; animal models have poor human relevance and ethical concerns, hindering translational research.
- Advantages of 3D Scaffold-Based Models: They offer superior biomimicry (replicating AT's ECM, stiffness, and cellular heterogeneity), support long-term adipocyte phenotype maintenance, enable high-throughput drug screening, and align with the 3Rs (reduction, refinement, replacement) principles.
- Critical Engineering Criteria: Successful models require suitable biomaterials (e.g., collagen, alginate-gelatin composites, hydrogels), tunable mechanical properties (mimicking healthy/dysfunctional AT stiffness), adipocyte hypertrophy induction, immune cell co-culture (to replicate inflammation), and vascularization (for nutrient/oxygen delivery).
- Diverse Applications: These models facilitate studying AT pathophysiology, screening anti-obesity therapeutics, developing patient-specific replicas (via iPSCs), and integrating with microfluidics to create multi-organ chips for comorbidity research; they also inform tissue engineering and cultivated meat technologies.
- Future Challenges: Key unresolved questions include enhancing model complexity while maintaining reproducibility, mimicking depot-specific AT properties, optimizing high-throughput scalability, and addressing ethical/regulatory barriers for clinical translation.
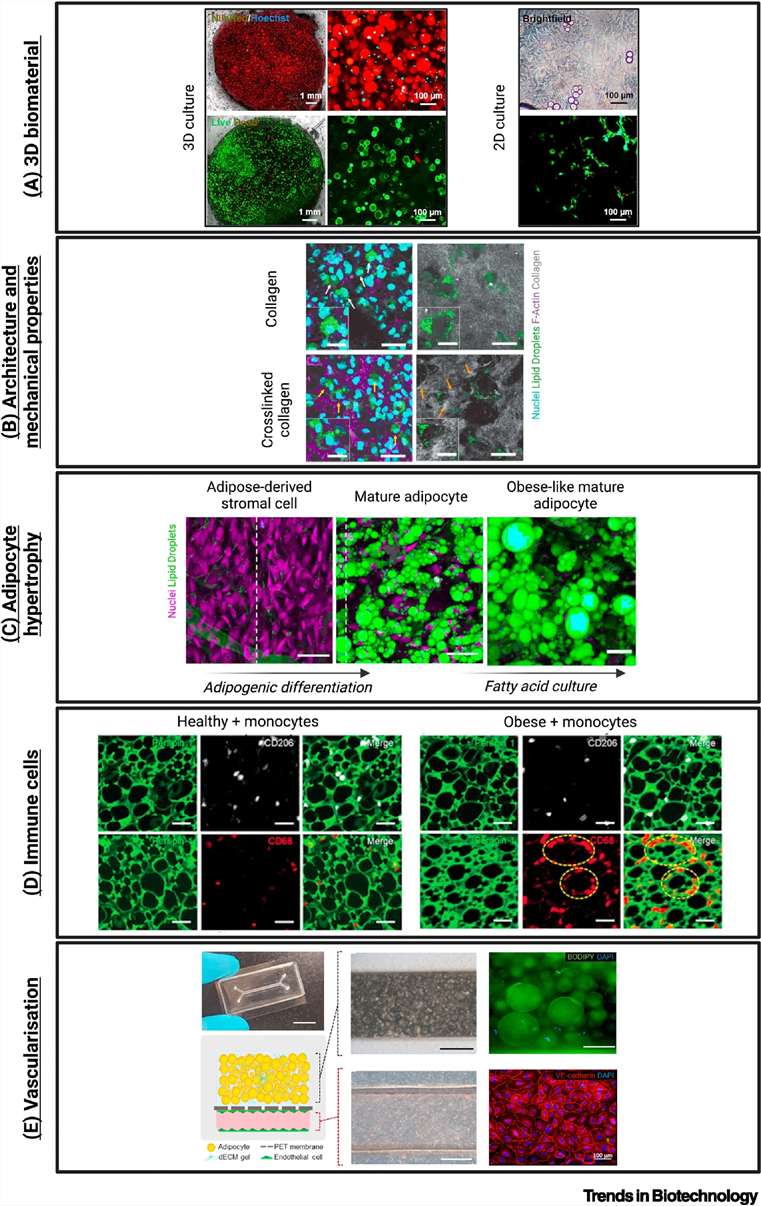 Fig.1 Applications of 3D scaffold-based in vitro platforms to mimic dysfunctional adipose tissue in obesity and metabolic disease. (Contessi, et al., 2025)
Fig.1 Applications of 3D scaffold-based in vitro platforms to mimic dysfunctional adipose tissue in obesity and metabolic disease. (Contessi, et al., 2025)
Customer Review
Overcoming Species-Specific Translational Barriers in Lead Optimization
"The transition from rodent models to Protheragen's human adipose organoids was a turning point for our metabolic pipeline. Their platform allowed us to identify a lead compound that showed no efficacy in mice but demonstrated potent lipolytic activity in the human 3D environment. We are currently planning our third project with them to explore inflammatory pathways."
Senior Director of Pharmacology, Global Biopharma
Longitudinal Metabolic Insights Through High-Content 3D Imaging Excellence
"Working with Protheragen has been a seamless experience. The data quality from their high-content imaging is exceptional, and the ability to maintain our organoids for over 60 days allowed us to see long-term metabolic shifts we previously couldn't capture. Their expertise in adipose biology is second to none."
Principal Scientist, Metabolic Research Institute
Frequently Asked Questions
-
How do your organoids compare to 2D preadipocyte cultures?
2D cultures fail to maintain mature, buoyant adipocytes and lack the complex ECM interactions found in our 3D models, which better represent in vivo morphology.
-
Can you model donor-specific metabolic variations?
Yes, we can utilize cells from donors with specific BMIs, ages, or diabetic statuses to match your target population.
-
What is the typical turnaround time for a screening project?
Most projects, from cell sourcing to the final data report, take approximately 6 to 8 weeks.
-
Are your models suitable for studying "browning" of fat?
Absolutely. We have validated protocols for inducing beige adipocyte differentiation and measuring UCP1-mediated thermogenesis.
-
How do you handle the buoyancy of mature adipocytes?
We use specialized hydrogel encapsulation or magnetic bioprinting to keep the organoids fixed in place for consistent imaging and media exchange.
-
Do you offer co-culture with immune cells?
Yes, we can integrate macrophages to study the inflammatory markers (e.g., IL-6, TNF-α) associated with obesity-related tissue dysfunction.
-
Can you perform RNA-sequencing on the organoids?
Yes, we provide comprehensive transcriptomic analysis as a downstream service to identify gene expression changes post-treatment.
-
Is the platform compatible with fluorescent lipid dyes?
Yes, our organoids are compatible with boron-dipyrromethene, Nile Red, and other live-cell imaging markers for real-time lipid tracking.
-
Can these organoids be used for toxicity testing?
Our platform is excellent for identifying adipocyte-specific toxicity that might be missed in standard hepatic or cardiac screens.
How to Contact Us
Protheragen provides a market-leading adipose tissue organoid modeling service that empowers researchers to explore obesity and metabolic diseases with unprecedented human relevance. By combining advanced 3D culture, magnetic stabilization, and automated analysis, we provide the insights needed to accelerate your drug discovery programs.
Contact Protheragen for More Information and to Discuss Your Project
Reference
- Contessi, N.N.; et al. Breaking barriers in obesity research: 3D models of dysfunctional adipose tissue. Trends in Biotechnology. 2025, 43(5): 1079–1093. (CC BY 4.0)
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.

