- Home
- Genetics and Epigenetics
- Decoding the Gastric Genetic Signature: Understanding FTO and MC4R Expression in Sleeve Gastrectomy Patients
Decoding the Gastric Genetic Signature: Understanding FTO and MC4R Expression in Sleeve Gastrectomy Patients
December 9, 2025
The global escalation of morbid obesity has positioned bariatric surgery as the most effective long-term intervention for significant weight loss and the resolution of metabolic comorbidities, yet the biological underpinnings of why individual outcomes vary so drastically remain partially obscured by the complexities of genetic predisposition. Published in Obesity Surgery (2025/2026), the study titled "In Vivo gastric expression of FTO and MC4R in Sleeve Gastrectomy Patients: Diagnostic Utility Without Predictive Value for Weight Loss" by S. A. G. S. M. de Oliveira and a team from the Alexandria Medical Research Institute addresses a critical gap in clinical knowledge. The FTO and MC4R genes have been extensively studied through peripheral blood samples and genome-wide association studies for their roles in hypothalamic energy regulation. However, their local behavior within the human gastric wall as a primary site of nutrient sensing and hormonal output remains poorly understood.
Past research has frequently struggled with the "missing heritability" problem, where genetic variants in blood do not always reflect the phenotypic behavior of the organs directly involved in digestion. This prospective case-control study investigated whether mRNA expression levels of FTO and MC4R in the gastric mucosa can serve as diagnostic biomarkers for obesity or as predictive tools to forecast weight loss success after laparoscopic sleeve gastrectomy (LSG).
Highlights
Innovative Local Profiling: This study transitions from systemic blood analysis to in vivo gastric mucosal analysis, providing a direct look at how the gastric tissue reacts to chronic obesity at a molecular level.
Precise Diagnostic Thresholds: Using receiver operating characteristic (ROC) curve analysis, the research establishes absolute molecular cutoffs that distinguish individuals with obesity from non-obese controls with 100% sensitivity.
Rigorous IPSW-Adjusted Methodology: To ensure data integrity, the researchers employed inverse propensity score weighting (IPSW) to balance age and sex variables, isolating the true metabolic differences between the 50 patients with obesity and 48 controls.
Long-term Outcome Correlation: Unlike short-term studies, this research tracked the percentage of total weight loss (%TWL) up to 12 months, providing a definitive answer regarding the prognostic value of these gastric biomarkers.
Key Findings
The results of this study reveal a stark contrast between the high diagnostic accuracy of gastric gene expression and its inability to predict therapeutic success, reinforcing a critical conceptual distinction between diagnostic and prognostic applications.
- Quantitative Expression Discrepancies
The research demonstrated a dramatic shift in the gastric molecular environment. FTO mRNA expression was significantly upregulated, showing a mean fold-change of 4.68 (p<0.001) compared to the control group. Conversely, MC4R expression was significantly downregulated, with a mean fold-change of -0.91 (p<0.001). As shown in the scatter plots, the study confirms that the obese state is associated with a specific "gastric genetic signature" characterized by high FTO and low MC4R activity, reflecting local adaptations to chronic nutrient exposure.
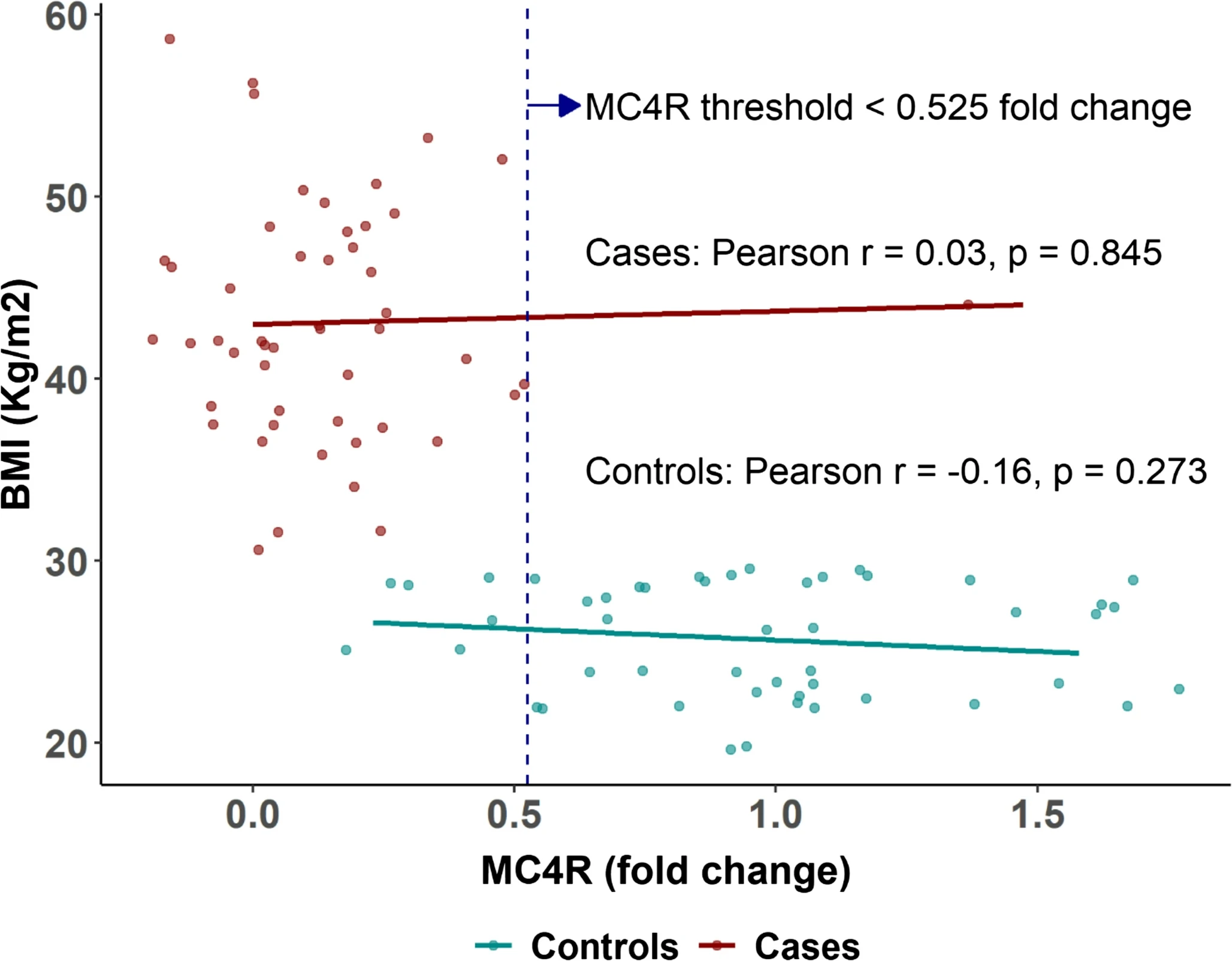 Fig.1 MC4R expression vs. BMI scatter plot: This scatter plot illustrates the distribution of MC4R expression and BMI across the obese and control groups, along with the threshold distinguishing obesity status. (Hany, et al., 2025)
Fig.1 MC4R expression vs. BMI scatter plot: This scatter plot illustrates the distribution of MC4R expression and BMI across the obese and control groups, along with the threshold distinguishing obesity status. (Hany, et al., 2025)
- Unprecedented Diagnostic Accuracy
The identification of specific thresholds for obesity classification is a major contribution. The ROC analysis identified an FTO fold-change threshold of > 1.515 and an MC4R threshold of < 0.525. Both markers achieved an area under the curve (AUC) of 1.00 (or 0.97 for MC4R), meaning these gastric markers can discriminate between obese and non-obese individuals with near-perfect accuracy. This establishes their role as effective tools for the molecular phenotyping of obesity.
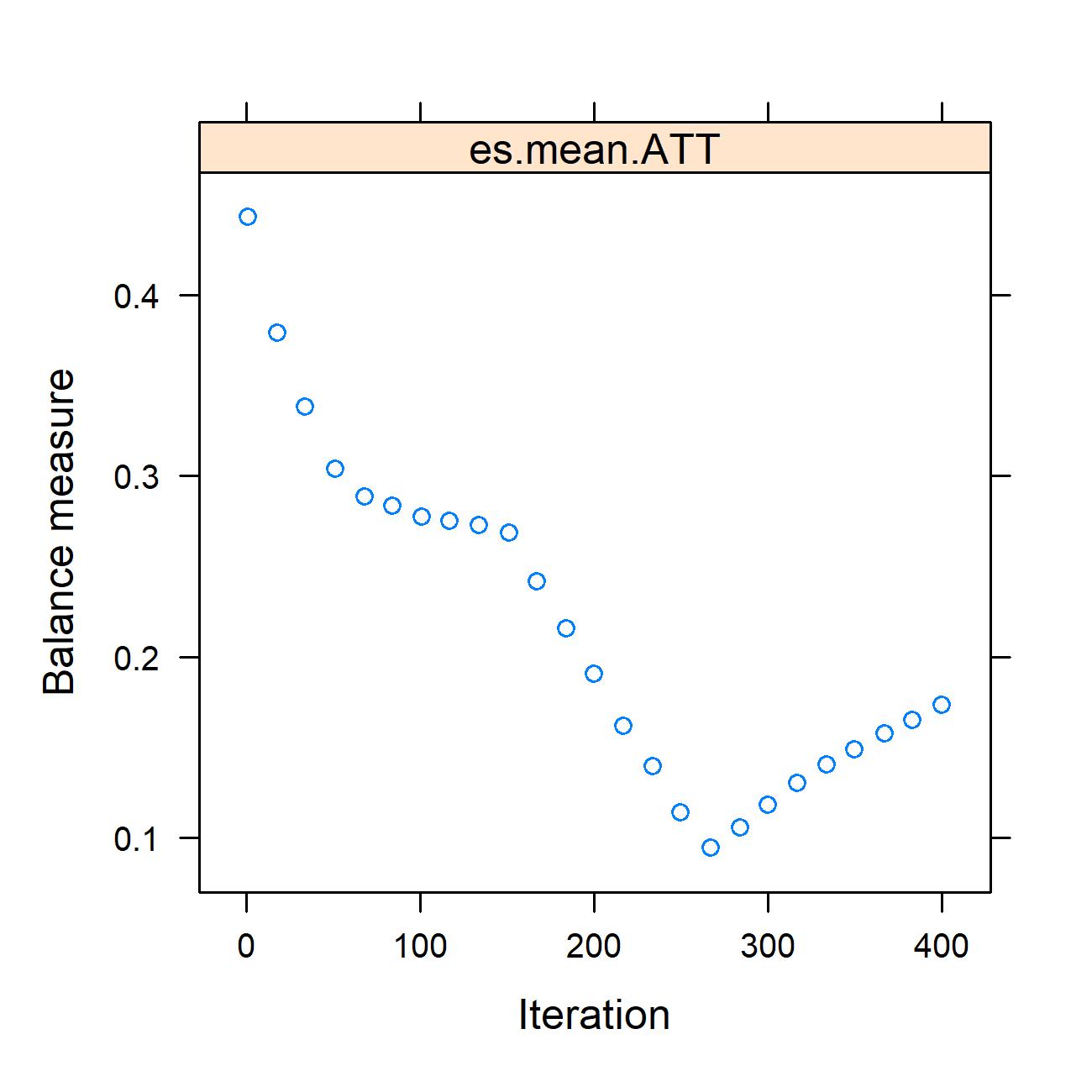 Fig.2 Weight balance optimization curve: This chart reflects the trend of optimal balance achieved for each covariate during iterative processing after inverse probability weighting. (Hany, et al., 2025)
Fig.2 Weight balance optimization curve: This chart reflects the trend of optimal balance achieved for each covariate during iterative processing after inverse probability weighting. (Hany, et al., 2025)
- The Predictive Paradox
Despite the clear diagnostic link, the study found a total absence of correlation between baseline expression levels and postoperative success. At 12-month follow-up, the Pearson correlation coefficients for %TWL were remarkably low (FTO: r=-0.07, p=0.607; MC4R: r=0.02, p=0.863). Whether a patient had extremely high FTO expression or low MC4R expression preoperatively did not determine their weight-loss response. This indicates that while surgery removes the tissue exhibiting these markers, the weight-loss trajectory is governed by broader physiological resets (like hormonal changes and gut-brain signaling) rather than baseline gastric transcriptomics.
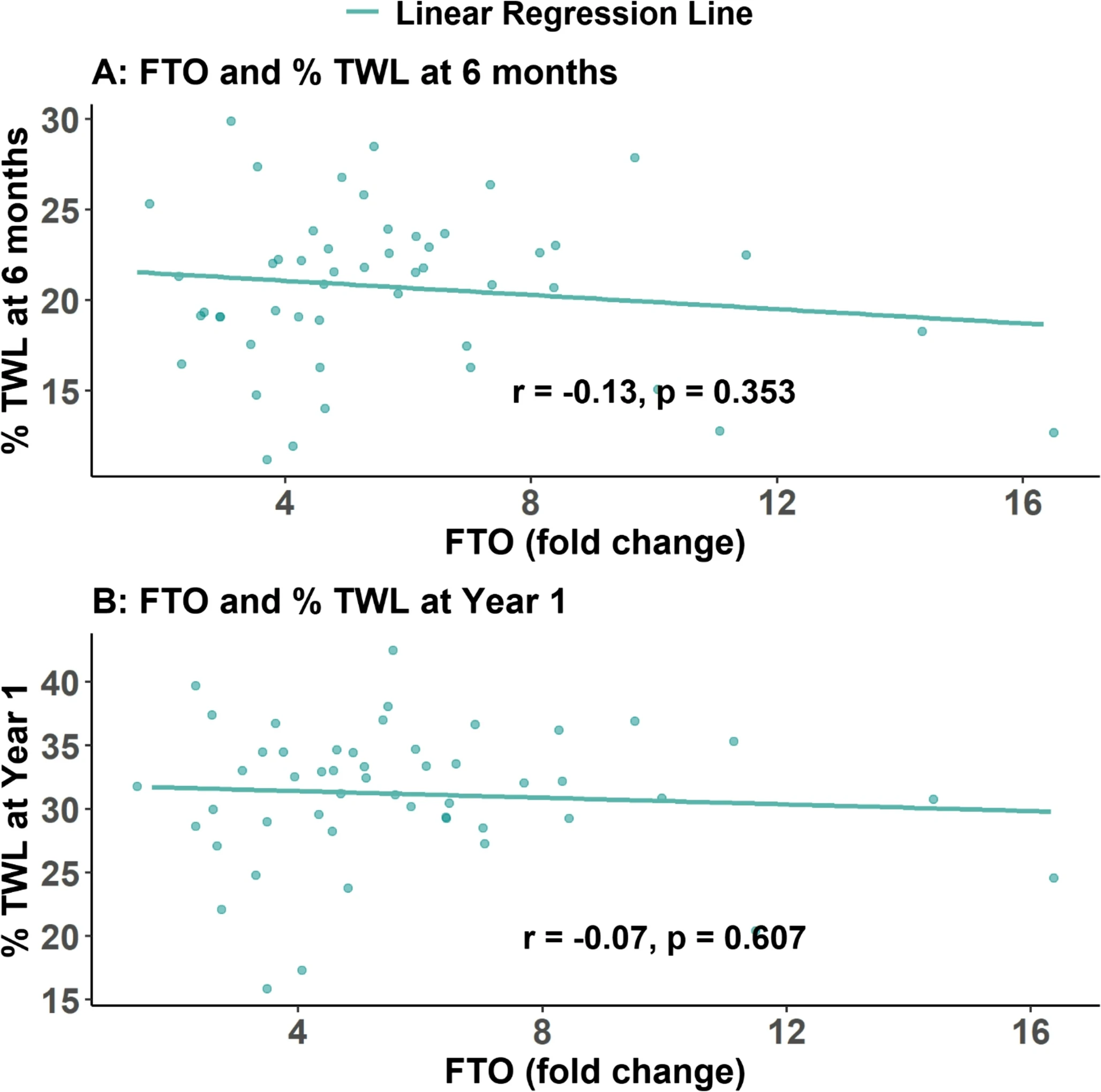 Fig.3 FTO expression and weight loss effect correlation diagram: These scatter plots confirm no significant correlation between preoperative gastric FTO gene expression levels and total weight loss percentage at six months and one year post-surgery. (Hany, et al., 2025)
Fig.3 FTO expression and weight loss effect correlation diagram: These scatter plots confirm no significant correlation between preoperative gastric FTO gene expression levels and total weight loss percentage at six months and one year post-surgery. (Hany, et al., 2025)
- Methodological Rigor and Robustness
The study utilized validated TaqMan qPCR assays with blinded triplicate analysis to ensure analytical precision. The researchers noted certain limitations, such as the lack of protein-level validation through western blot and the single-center study design. Nevertheless, the use of IPSW and double-adjusted models effectively minimized confounding risks from age and sex.
Interpretation & Translational Value: Rethinking Personalised Medicine in Bariatrics
The findings provide a profound shift in our understanding of "obesity genes" in peripheral tissues. Gastric FTO and MC4R expression levels represent a biological marker of the obese state itself, serving as a form of local metabolic adaptation. These markers should be viewed as indicators of obesity rather than determinants of surgical success. From a translational standpoint, this underscores that the "obese stomach" undergoes molecular reprogramming that reflects systemic metabolic stress. Notably, the study points to a broader concern: the link between FTO overexpression and gastrointestinal oncogenesis, suggesting that obesity-related gastric changes might increase tissue vulnerability to cancer. For the future of precision medicine, while these markers may not forecast surgery outcomes, they remain high-priority targets for pharmacological modulation. FTO demethylase inhibitors and selective MC4R agonists are emerging as precision-medicine hubs that could potentially regulate energy homeostasis from the "periphery" to the "center."
Research Support: Empowering Your Metabolic Discoveries
For researchers aiming to explore the local molecular drivers of obesity or validate new biomarkers in metabolic tissues, our organization offers specialized translational research services. Protheragen provides high-throughput qPCR assays, advanced IPSW-based statistical modeling, and comprehensive RNA/protein extraction services. Whether you are investigating nutrient sensing in the gastrointestinal tract, exploring epigenetic mechanisms like DNA methylation, etc., our platform is designed to support the development of high-impact metabolic research.
Reference
- Hany, M.; et al. In vivo gastric expression of FTO and MC4R in sleeve gastrectomy patients: Diagnostic utility without predictive value for weight loss. Obesity Surgery. 2025: 1-14. (CC BY 4.0)