- Home
- Advances in Novel Anti-Obesity Therapies
- Probiotic Synergy: How Clostridium butyricum Alleviates Obesity by Boosting Akkermansia muciniphila
Probiotic Synergy: How Clostridium butyricum Alleviates Obesity by Boosting Akkermansia muciniphila
December 9, 2025
The global escalation of obesity represents one of the most pressing public health challenges of the 21st century, with its etiology deeply rooted in the complex interplay between high-calorie diets and the trillions of microbes residing in our gastrointestinal tract. Recent scientific consensus identifies gut dysbiosis, specifically a reduction in beneficial taxa and a rise in inflammatory markers, as a primary driver of metabolic dysfunction. Consequently, finding targeted probiotic interventions remains an active area of exploration. This significant study, titled "Oral Administration of Clostridium butyricum alleviates High-Fat Diet-Induced Obesity in Mice by Modulating Gut Akkermansia muciniphila Abundance via Direct Growth Promotion," was published in the Journal of Microbiology and Biotechnology by researchers from prestigious institutions, including China Agricultural University. Despite prior evidence suggesting that C. butyricum (CLB) can improve lipid metabolism through butyrate production, a critical research gap persisted regarding its precise impact on other key beneficial bacteria. Specifically, the potential "cross-feeding" or stimulatory relationship between CLB and the mucin-degrading bacterium A. muciniphila remained underexplored until this investigation. This particular species is consistently negatively associated with obesity, making its modulation a key focus of the study.
Highlights
- Novel Mechanistic Insight: For the first time, researchers demonstrated that CLB does not just work in isolation but acts as a growth catalyst for A. muciniphila, a "next-generation" probiotic candidate.
- Dual-Pronged Experimental Design: The study seamlessly integrates in vivo mouse models of high-fat diet (HFD) induced obesity with in vitro co-culture experiments to prove direct growth promotion.
- Focus on Metabolite Cross-Feeding: By analyzing CLB lysates and culture supernatants, the team identified that the metabolic byproducts of CLB are the primary drivers of A. muciniphila proliferation.
- Selective Microbiota Modulation: Unlike many interventions that attempt to overhaul the entire gut ecosystem, this study highlights how CLB selectively restores "depleted" beneficial genera to achieve metabolic balance.
Key Findings
The experimental results provide a comprehensive and detailed account of how CLB intervention effectively counteracts the damage caused by a chronic high-fat diet.
- Phenotypic Alleviation of Obesity
The administration of CLB (1 × 10¹⁰ CFU/kg/day) for 8 weeks resulted in striking physical improvements. As illustrated, mice in the HFD + CLB group showed a 13.20% reduction in body weight gain compared to the HFD-only group (16.77 g vs. 19.32 g). Crucially, this weight loss was not due to reduced food intake but rather metabolic shifts. Fig. 1 quantifies the fat mass reduction, where CLB-treated mice exhibited significantly lower fat percentages (21.10%) than the HFD model group (23.39%). Histological analysis further confirmed these findings, showing that the hypertrophic (enlarged) adipocytes typically seen in obese mice were noticeably smaller and more organized in the CLB group.
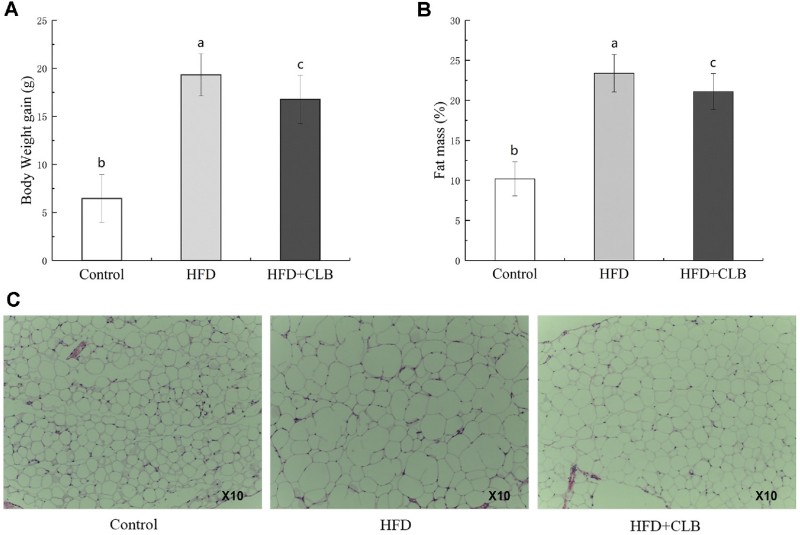 Fig.1 Comparison of body weight, fat content, and adipose tissue size in HFD and control mice. (Dong, et al., 2025)
Fig.1 Comparison of body weight, fat content, and adipose tissue size in HFD and control mice. (Dong, et al., 2025)
- Restoration of Short-Chain Fatty Acids
A hallmark of CLB's metabolic activity is the production of butyrate. Data in Table 1 reveal that a high-fat diet severely depletes fecal short-chain fatty acids (SCFAs). However, CLB supplementation partially restored butyrate levels to 11.73 μg/g, a significant increase from the 7.27 μg/g observed in the HFD group (P<0.05). This restoration of butyrate is vital for maintaining the integrity of the intestinal barrier and suppressing low-grade systemic inflammation.
Tab. 1 Levels of short-chain fatty acids in mouse fecal samples. (Dong, et al., 2025)
| Groups | Acetate | Propionate | Butyrate |
|---|---|---|---|
| Control | 288.62 ± 122.32 | 129.26 ± 57.39 | 63.25 ± 22.80 |
| HFD | 23.61 ± 5.03 | 14.00 ± 5.71 | 7.27 ± 3.40 |
| HFD + CLB | 17.96 ± 2.44 | 7.25 ± 4.05 | 11.73 ± 4.99 |
- Selective Modulation of Gut Microbiota
The 16S rRNA gene sequencing provided deep insights into the microbial shifts. While CLB did not completely return the overall microbial community structure to that of the control group, it significantly shifted the HFD-shaped microbiota. Akkermansia abundance, which was nearly eradicated by the HFD, was significantly restored by CLB intervention. The researchers noted that among 11 obesity-related genera, Akkermansia was the primary target stimulated by CLB, suggesting a synergistic relationship where CLB creates a hospitable environment for A. muciniphila to thrive.
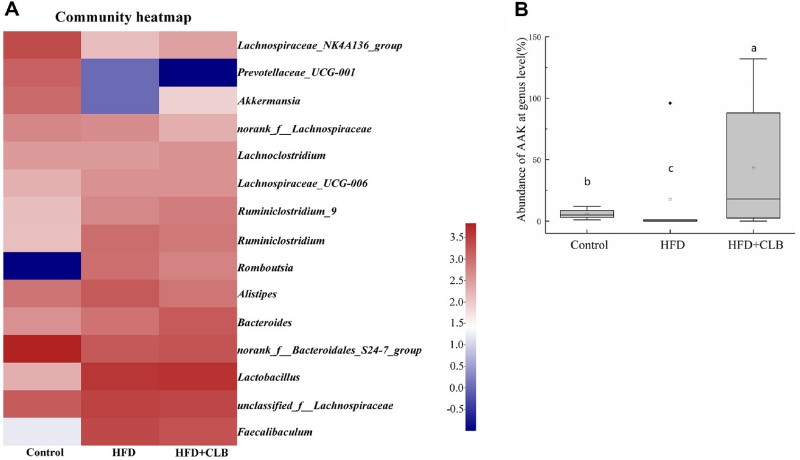 Fig.2 CLB supplementation alters gut microbiota and increases A. muciniphila abundance. (Dong, et al., 2025)
Fig.2 CLB supplementation alters gut microbiota and increases A. muciniphila abundance. (Dong, et al., 2025)
- Direct Growth Promotion In Vitro
To verify if CLB directly promotes A. muciniphila, the team conducted in vitro cultures. Fig. 5 shows that sodium butyrate alone increases A. muciniphila growth in a dose-dependent manner. More significantly, it demonstrates that adding CLB cell lysate to the growth medium results in a markedly higher optical density (OD600) for A. muciniphila. CLB culture supernatant (at 25% and 50% concentrations) significantly enhances A. muciniphila proliferation compared to controls (P<0.05). This confirms that CLB secretes specific metabolites or bioactive components that A. muciniphila utilizes to grow more efficiently.
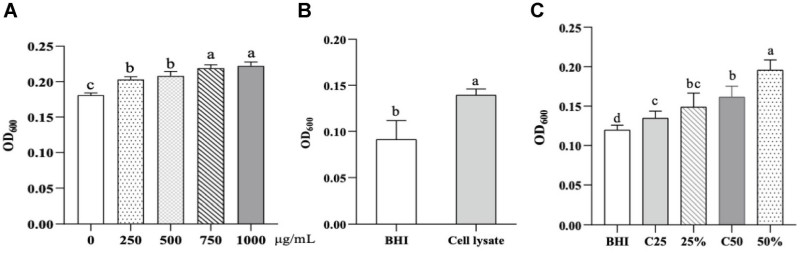 Fig.3 Butyrate and CLB components promote A. muciniphila growth in a dose-dependent manner. (Dong, et al., 2025)
Fig.3 Butyrate and CLB components promote A. muciniphila growth in a dose-dependent manner. (Dong, et al., 2025)
Interpretation & Translational Value
The significance of these findings extends far beyond the laboratory, offering a profound reinterpretation of how probiotics like CLB function within the complex gut "social network." By demonstrating that CLB acts as a growth promoter for A. muciniphila, this research highlights a "microbial cross-feeding" mechanism that could revolutionize the development of metabolic therapies. Instead of merely supplementing a single strain, future treatments could focus on "consortia" or specific metabolic precursors that leverage these internal synergies. From a translational perspective, this suggests that CLB can be used as a potent strategy to bolster the levels of A. muciniphila, which is notoriously difficult to culture and deliver directly to humans due to its strict anaerobic nature. Furthermore, the ability of CLB to increase butyrate while simultaneously reducing systemic fat mass provides a solid scientific foundation for using butyrate-producing spores as a low-cost, stable, and effective dietary intervention for managing obesity and its associated metabolic syndromes, such as type 2 diabetes and nonalcoholic fatty liver disease.
Research Support
For researchers dedicated to exploring gut microbiome interventions or metabolic studies, Protheragen offers comprehensive Preclinical and Translational Research services encompassing experimental design, microbiome profiling, and data analysis. Our platform features advanced 16S/metagenomic sequencing technology, short-chain fatty acid quantification, and specialized in vitro co-culture models to help you uncover the next breakthrough in microbial cross-feeding and metabolic health. Additionally, we provide Novel Therapeutic Development Services that have already supported extensive anti-obesity research.
Reference
- Dong, X.; et al. Oral administration of Clostridium butyricum alleviates high-fat diet-induced obesity in mice by modulating gut Akkermansia muciniphila abundance via direct growth promotion. J Microbiol Biotechnol. 2025, 35: e2509005. (CC BY 4.0)