New Discovery on the Culprit Behind Obesity: How Fat Cells Ignite Inflammation and Metabolic Crisis Through the B2M Protein?
December 3, 2025
December 8, 2025
The global prevalence of type 2 diabetes and obesity is primarily driven by chronic low-grade inflammation. This pathological state is typically triggered by "metabolic endotoxemia": dangerous signals originating from the gut, such as lipopolysaccharides (LPS), penetrate the bloodstream, thereby activating the body's innate immune system. While we know the microbiome plays a starring role in this process, the specific microbial compounds that act as the "off switches" for this inflammation have remained elusive.
In the complex dialogue between our gut microbiome and our own cells, the "words" are often small molecules called metabolites. For years, one such molecule, trimethylamine (TMA), was largely overshadowed by its liver-converted cousin, TMAO, which is notorious for its links to heart disease. However, a groundbreaking study recently published in Nature Metabolism (2025) titled "Inhibition of IRAK4 by microbial trimethylamine blunts metabolic inflammation and ameliorates glycemic control" has turned the tide. Led by an international team of researchers, this study reveals that TMA is not just a metabolic byproduct but a potent signaling molecule that can actually shield the body from the devastating effects of a high-fat diet (HFD).
The findings of this study are both extensive and remarkably consistent across different biological scales, ranging from purified molecular interactions and cellular pathways to complex whole-organism physiology. This multi-layered alignment underscores the robustness of the microbial-host crosstalk: while kinome screenings identified the high-affinity binding at the protein level, follow-up experiments in human cell lines and diverse mouse models confirmed that this molecular "handshake" effectively translates into a systemic metabolic shield against the inflammatory ravages of an HFD.
Initially, the researchers observed a surprising phenomenon in mice fed an HFD. While the mice became obese, those supplemented with high levels of choline (HC-HFD) showed significantly better glucose tolerance and lower levels of systemic inflammatory cytokines like IL-6 and TNF compared to those on a low-choline HFD. Using 1H-NMR spectroscopy, they confirmed that this benefit was strongly correlated with a massive 20-fold increase in circulating TMA levels.
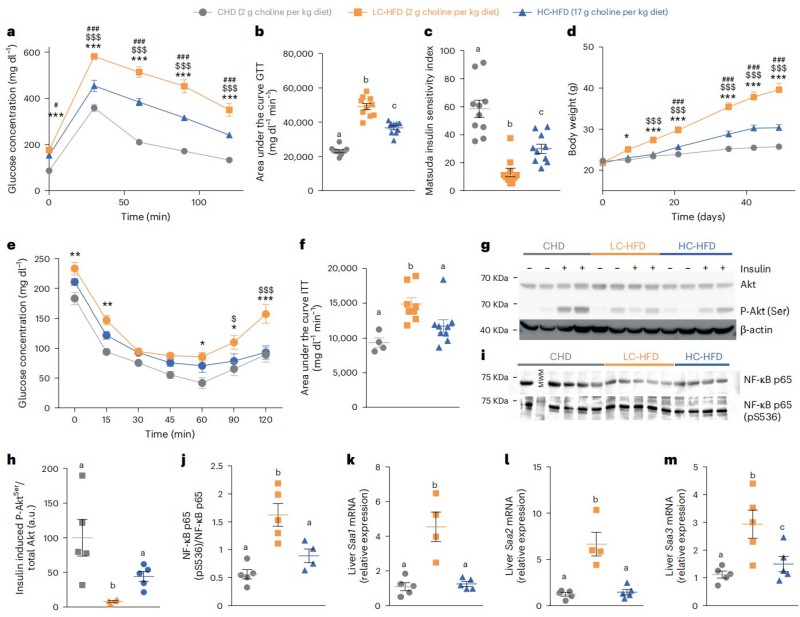 Fig.1 Choline supplementation improves glucose control, insulin sensitivity, and inflammation in HFD mice. (Chilloux, et al., 2025)
Fig.1 Choline supplementation improves glucose control, insulin sensitivity, and inflammation in HFD mice. (Chilloux, et al., 2025)
To prove TMA was the hero, the team used 3,3-dimethyl-1-butanol (DMB) to block the bacterial enzymes that produce TMA. When TMA production was shut down, the beneficial effects of choline supplementation vanished as glucose levels spiked and insulin resistance returned. Conversely, when they bypassed the gut and delivered TMA directly to HFD-fed mice via osmotic pumps, it mimicked the benefits of choline, normalizing "Matsuda Index" (a key insulin sensitivity marker) and reducing hepatic inflammatory markers like Saa1, Saa2, and Saa3.
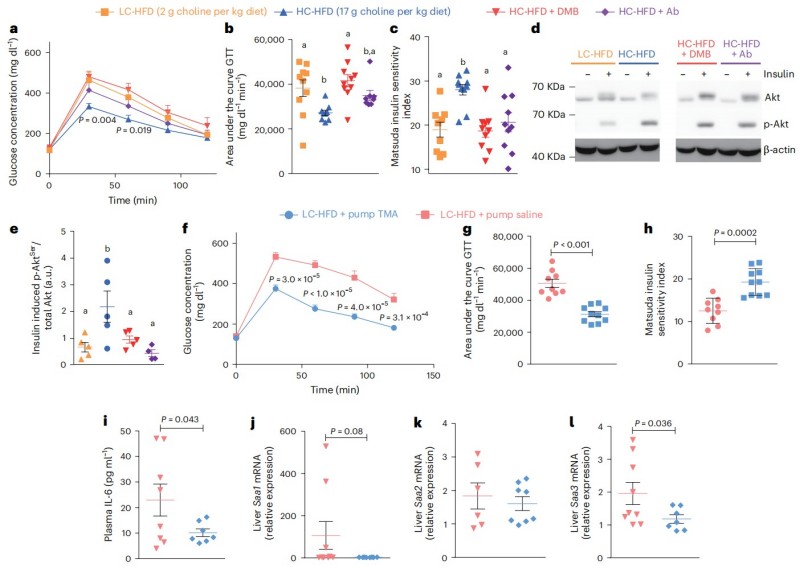 Fig.2 Inhibiting TMA production abolishes choline benefits, while TMA treatment reproduces metabolic effects. (Chilloux, et al., 2025)
Fig.2 Inhibiting TMA production abolishes choline benefits, while TMA treatment reproduces metabolic effects. (Chilloux, et al., 2025)
Through a high-throughput screen of 456 human kinases, the authors identified IRAK4 as the primary molecular target. TMA physically binds to IRAK4 with a dissociation constant (Kd) of 14 nM and functions as a potent inhibitor with an IC50 of 3.4 µM. This inhibitory effect was rigorously validated across multiple human models: in PBMCs, TMA blunted the pro-inflammatory response to LPS by reducing the phosphorylation of IRAK1 and NF-κB; in primary human hepatocytes, it rescued insulin signaling from palmitate-induced damage. Demonstrating its profound systemic impact, a single dose of TMA even significantly improved survival in a mouse model of lethal LPS-induced septic shock.
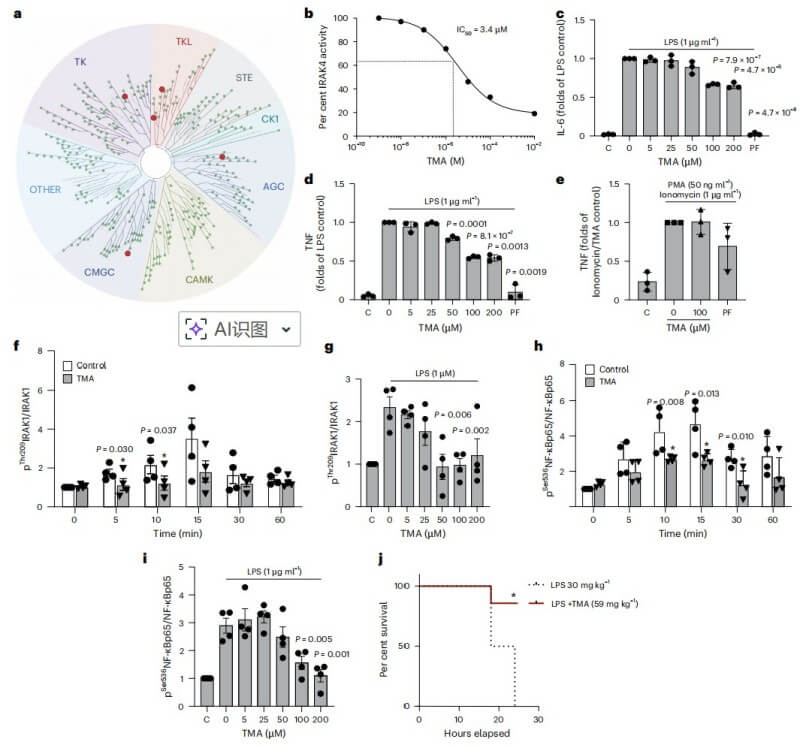 Fig.3 TMA suppresses IRAK4 activity and reduces LPS-induced inflammatory signaling. (Chilloux, et al., 2025)
Fig.3 TMA suppresses IRAK4 activity and reduces LPS-induced inflammatory signaling. (Chilloux, et al., 2025)
Finally, the team showed that mice genetically lacking IRAK4 (Irak4-/-) were naturally protected against the metabolic damage of an HFD. Using a known pharmaceutical IRAK4 inhibitor (PF06650833) produced the same therapeutic results as TMA treatment, solidifying IRAK4 as the primary target through which TMA exerts its metabolic magic.
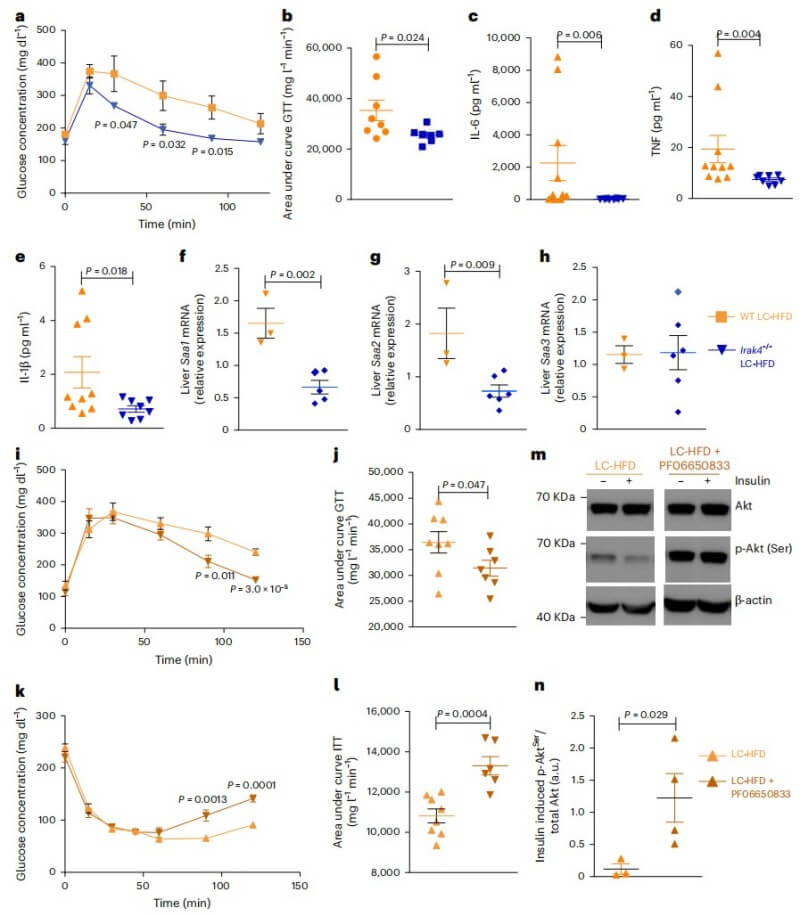 Fig.4 Loss or inhibition of IRAK4 improves glucose tolerance and inflammation in HFD mice. (Chilloux, et al., 2025)
Fig.4 Loss or inhibition of IRAK4 improves glucose tolerance and inflammation in HFD mice. (Chilloux, et al., 2025)
The significance of this study cannot be overstated for the fields of metabolic disease and drug development. It redefines TMA, once thought of as a mere waste product, as a "microbial signaling metabolite" that maintains a low immunological footprint. By identifying the kinome as a target for microbial metabolites, this research opens up a whole new category of therapeutic targets.
One of the most profound insights is the context-dependence of the TMA/TMAO ratio. While TMAO is often linked to cardiovascular risk via PERK activation and calcium-mediated platelet aggregation, the research presents a different perspective on its precursor. The findings suggest that increasing TMA levels, perhaps by inhibiting the FMO3 enzyme that converts TMA to TMAO, actually attenuates metabolic inflammation and atherosclerosis. This discovery explains why higher choline consumption is associated with improved insulin sensitivity in human populations.
While the study acknowledges limitations, such as the 250-fold difference between binding affinity (Kd) and inhibitory concentration (IC50) in purified assays, the consistent results across human hepatocytes, PBMCs, and four different in vivo mouse models make the findings highly robust. This work uncovers a strategic "hijacking" of the TLR signal transduction machinery by gut microbes for the host's benefit, offering a path toward new treatments that alleviate obesity-associated cardiovascular risks and metabolic disorders.
For researchers dedicated to exploring the complexity of gut-host kinase interactions or developing interventions for metabolic endotoxemia, Protheragen provides the essential tools to translate these insights into reality. We deliver comprehensive Preclinical and Translational Services spanning high-resolution microbial metabolomics, advanced liver and immune cell modeling, and specialized kinase activity assays. Whether you're investigating the effects of specific dietary supplements or screening novel microbial inhibitors, our expert team will fully support your experimental design, multi-omics data integration, and mechanism validation. This comprehensive approach empowers your next breakthrough in metabolic health and advances the discovery of novel anti-obesity therapeutic targets.
Reference
Copyright © Protheragen. All rights reserves.