- Home
- Inflammation and Immune Responses
- The Hidden Architect of Fat Health: How AQP7 Deficiency Drives Adipose Remodeling
The Hidden Architect of Fat Health: How AQP7 Deficiency Drives Adipose Remodeling
November 7, 2025
Maintaining metabolic balance is a complex biological feat, and recent research has identified a surprising key player in this process: a protein channel traditionally known for transporting water and glycerol. What exactly happens to our fat tissue when it stops functioning correctly? This is the central question behind the recent study titled "AQP7 deficiency drives adipose tissue remodeling and disrupts homeostasis," published in 2025 by a collaborative team in Nature Communications. Adipose tissue (AT) is no longer viewed as a simple storage depot; it is a vital endocrine organ that manages energy through the release of signaling molecules.
The aquaporin-7 (AQP7) channel is a specialized protein that facilitates the exit of glycerol from adipocytes, a process essential for energy balance. While past research established a link between AQP7 and adult-onset obesity, the precise mechanisms—especially how its deficiency triggers tissue remodeling and systemic dysfunction—remained a significant gap in metabolic science. This study explores how the loss of this specific channel leads to profound structural changes in fat, driving obesity and inflammation. By investigating how AQP7 influences the very maturation and structural integrity of AT, particularly under the stress of modern high-glucose diets, the researchers aim to bridge a critical gap in our understanding of metabolic health.
Highlights
- Integrated Evidence: It uniquely combines human clinical data (epigenetic profiling), transgenic mouse models, and advanced in vitro cellular secretome analysis.
- Beyond Glycerol Transport: It reveals that AQP7 is not just a passive "gate" for glycerol but a master regulator of the adipose tissue secretome and extracellular matrix (ECM) remodeling.
- Environmental Interaction: The study demonstrates a critical synergy between genetic AQP7 deficiency and high-glucose (HG) dietary stress, worsening metabolic outcomes.
- Peritoneal Connection: For the first time, it links AQP7-driven AT dysfunction to impaired transport across the peritoneal membrane, providing insights for patients undergoing peritoneal dialysis (PD).
Key Findings
The researchers provided an exhaustive look at how AQP7 deficiency disrupts metabolic health across species and experimental models, revealing a multifaceted involvement in adipocyte hypertrophy and tissue stiffening.
- Epigenetic Links in Humans
Analysis of a cohort of 65 women revealed that those in the obese group had significantly lower AQP7 expression in their visceral adipose tissue (VAT) compared to lean individuals. Critically, the team found a strong inverse correlation between AQP7 mRNA levels and Body Mass Index (BMI). This was tied to DNA methylation: seven specific sites on the AQP7 locus showed a negative correlation with expression, three of which were positively linked to BMI. This suggests that epigenetic "silencing" of AQP7 serves as a molecular interface between environmental factors and metabolic health.
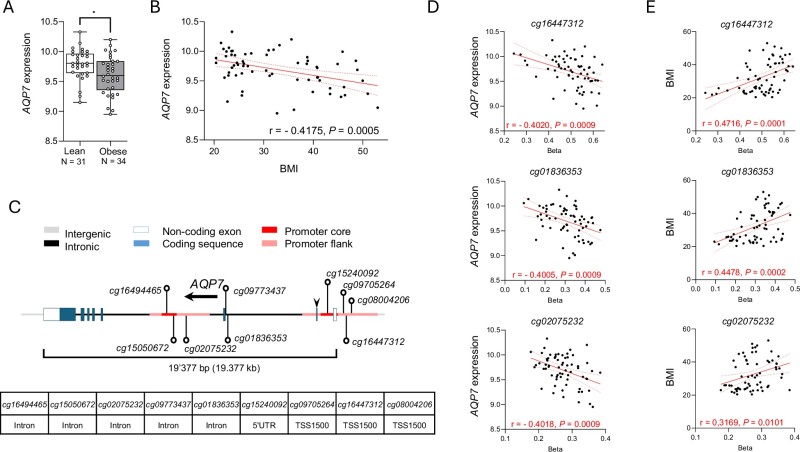 Fig.1 AQP7 expression in adipose tissue decreases with increasing BMI and is closely correlated with DNA methylation levels at its gene locus. (Costa, et al., 2025)
Fig.1 AQP7 expression in adipose tissue decreases with increasing BMI and is closely correlated with DNA methylation levels at its gene locus. (Costa, et al., 2025)
- Systemic Dysfunction and Metabolic Shifts
In mice, the absence of AQP7 led to a dose-dependent increase in body weight, hyperglycemia, and hyperinsulinemia. Micro-CT scans confirmed excessive VAT accumulation, while histological examination revealed a "hypertrophic" phenotype: fewer but much larger adipocytes. Mechanistically, AQP7 deficiency triggers a metabolic shift characterized by reduced lipolytic activity and increased expression of enzymes involved in lipogenesis, such as fatty acid synthase (Fasn). When exposed to a high-glucose diet, these mice showed even more severe ectopic fat deposition in the liver and skeletal muscle, likely because hypertrophic adipocytes lose their capacity to mobilize lipids effectively.
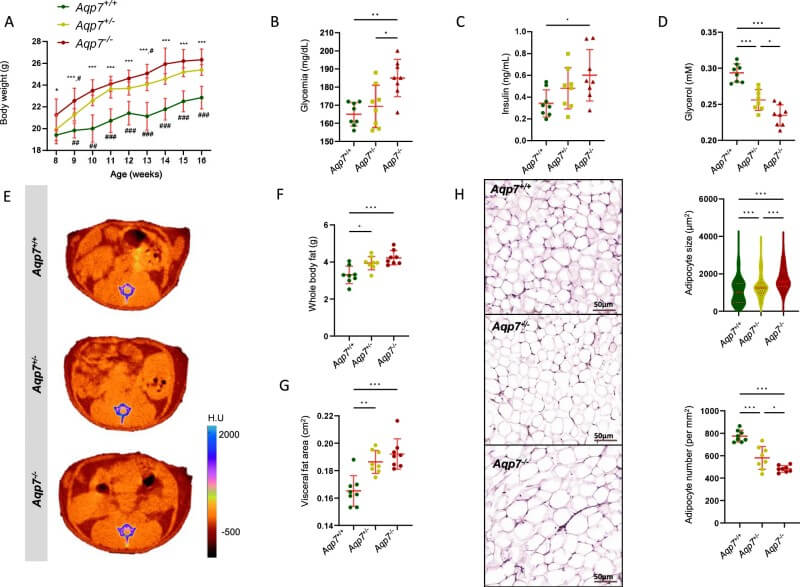 Fig.2 AQP7-deficient mice exhibit weight gain, enhanced fat accumulation, and impaired glucose and insulin metabolism. (Costa, et al., 2025)
Fig.2 AQP7-deficient mice exhibit weight gain, enhanced fat accumulation, and impaired glucose and insulin metabolism. (Costa, et al., 2025)
- Tissue Remodeling and Peritoneal Impairment
The study provides striking evidence that AQP7 deficiency transforms the VAT into a pro-inflammatory and fibrotic environment. RT-qPCR showed the upregulation of fibrotic markers (Col1a, Col3a, and Tgfβ1), indicating excessive ECM accumulation that contributes to tissue stiffening. This "fibrotic remodeling" likely impairs oxygen diffusion, leading to localized hypoxia—a key driver of further dysfunction. The researchers also observed pathological vascular remodeling, with increased capillary density (CD31) and thickened vascular walls (α-SMA). Functional tests revealed that this remodeled VAT significantly reduced water transport (ultrafiltration) across the peritoneal membrane, identifying AQP7 as a protector of peritoneal membrane integrity.
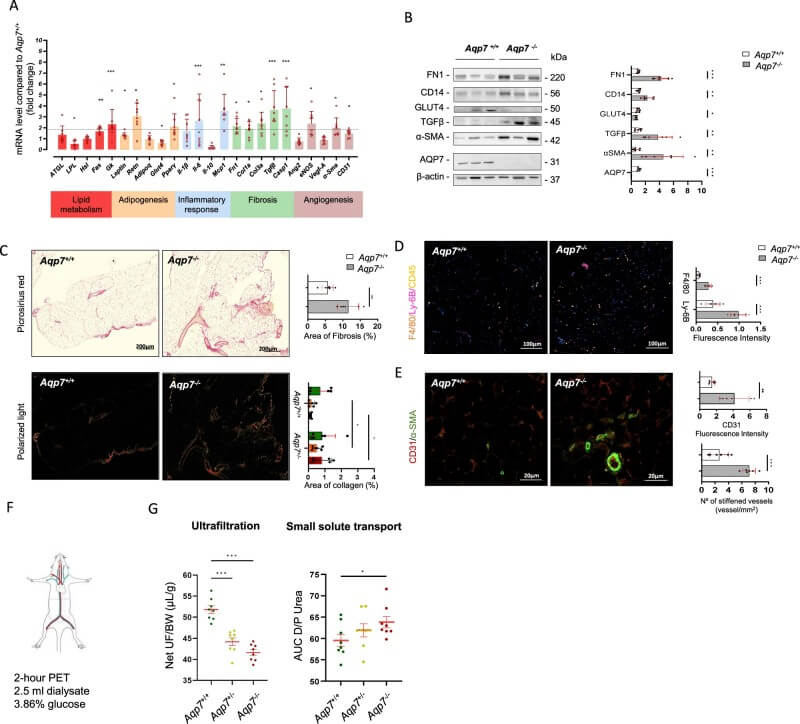 Fig.3 AQP7 deficiency promotes adipose tissue fibrosis, inflammatory responses, and vascular remodeling, impairing tissue functional status. (Costa, et al., 2025)
Fig.3 AQP7 deficiency promotes adipose tissue fibrosis, inflammatory responses, and vascular remodeling, impairing tissue functional status. (Costa, et al., 2025)
- Mechanistic Insights via Cell Culture and Secretomics
Using mouse embryonic fibroblasts (MEFs), the team showed that while AQP7 is not required for adipocyte differentiation, its absence leads to functionally defective, oversized adipocytes. These cells showed a dramatic shift in their "secretome," with proteomic analysis identifying 1761 murine proteins. AQP7-null (Aqp7-/-) adipocytes activated pathways related to mitochondrial dysfunction and hypoxia while deactivating insulin signaling. Crucially, the secretome of Aqp7-/- adipocytes was found to promote fibroblast activation, creating a pro-fibrotic microenvironment characterized by enhanced TGF-β production.
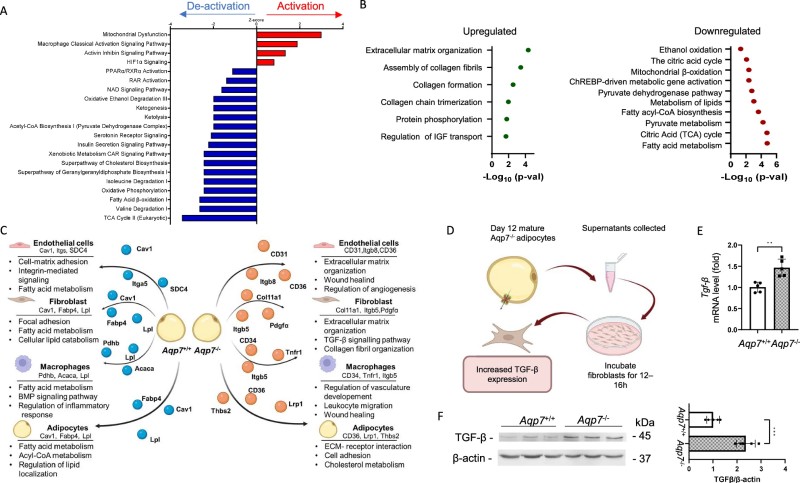 Fig.4 AQP7 deficiency alters adipocyte secretion profiles, activates inflammatory and fibrotic pathways, and inhibits metabolic and insulin signaling pathways. (Costa, et al., 2025)
Fig.4 AQP7 deficiency alters adipocyte secretion profiles, activates inflammatory and fibrotic pathways, and inhibits metabolic and insulin signaling pathways. (Costa, et al., 2025)
Interpretation & Translational Value
These findings redefine AQP7 as a central regulator of adipose tissue homeostasis and metabolic resilience. The study moves beyond the "glycerol channel" narrative, positioning AQP7 as an essential manager of the adipocyte secretome and tissue architecture. By demonstrating that AQP7 deficiency promotes a shift toward fibrosis, inflammation, and pathological vascular remodeling, the research identifies a major pathway for obesity-related complications. Translating this to the clinic, targeting AQP7 expression or its downstream fibrotic signals offers a novel therapeutic strategy. This is especially vital for patients on peritoneal dialysis, where daily glucose exposure puts visceral fat at risk. Preserving AQP7 function may be the key to preventing the metabolic deterioration and peritoneal membrane failure that often complicates long-term dialysis.
Research Support
For researchers interested in exploring the complexities of adipose tissue remodeling, Protheragen provides a full suite of preclinical services. From CpG Methylation Analysis and body composition scanning to advanced TMT-based secretome proteomics, our platform is designed to support high-impact metabolic research. If you are investigating how adipocyte hypertrophy influences systemic inflammation or seeking to characterize novel mouse models, we offer the analytical tools and expertise to accelerate your discovery.
Reference
- Costa, I.P.D.; et al. AQP7 deficiency drives adipose tissue remodeling and disrupts homeostasis. npj metabolic health and disease. 2025, 3(1): 44. (CC BY 4.0)