Rethinking Obesity as Ponderostat Dysregulation Central Neuroendocrine Control of Energy Balance
October 15, 2025
December 23, 2025
Managing schizophrenia often presents a cruel paradox where the most effective antipsychotic drugs (APDs), such as clozapine and olanzapine, are also the most likely to cause severe metabolic side effects, including rapid weight gain and type 2 diabetes. Published in Reviews in Endocrine and Metabolic Disorders (2025), this comprehensive review by an expert team explores why these essential medications disrupt energy balance so profoundly.
While the therapeutic benefits of APDs are well-documented, the neurobiological mechanisms driving their metabolic "cost" remain a significant challenge in psychiatry, as weight-related health risks often lead to treatment non-adherence and clinical relapse. The study identifies a critical research gap: while we know APDs cause weight gain, we have only recently begun to map the specific hypothalamic circuits and intracellular sensors, like AMPK, that translate drug-receptor interactions into systemic metabolic dysfunction.
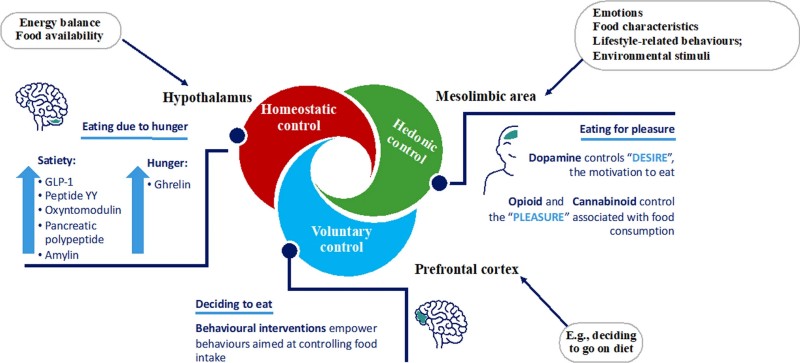
The core of the research details how APDs disrupt specific hypothalamic circuits to drive hyperphagia (excessive eating) and reduced energy expenditure.
The researchers analyze how olanzapine acts as a potent antagonist at H1 and 5HT2C receptors within the ARC. This action suppresses POMC/CART neurons (the "anorexigenic" signal) while simultaneously activating AgRP/NPY neurons (the "orexigenic" signal). This "dual-threat" mechanism effectively tricks the brain into a state of starvation. Recent data also highlights the role of the transcription factor Nr5a2, which increases AgRP expression, and the upregulation of mu-opioid receptors (MORs) in the ARC, potentially driving the intense cravings for high-fat foods seen in patients.
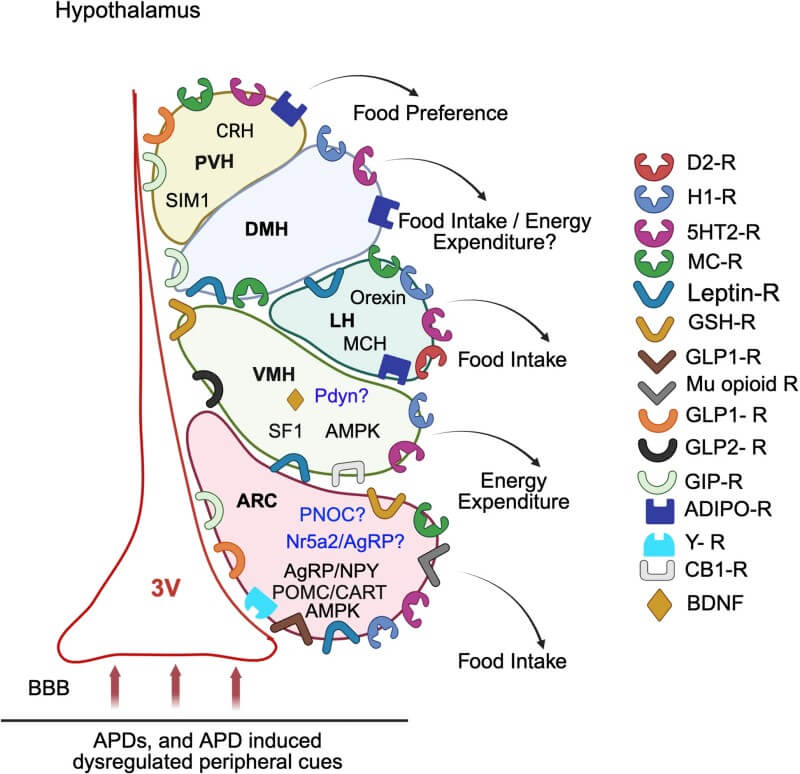 Fig.1 Key hypothalamic nuclei regulate energy balance and respond to APDs and peripheral signals influencing obesity. (Mukherjee, et al., 2025)
Fig.1 Key hypothalamic nuclei regulate energy balance and respond to APDs and peripheral signals influencing obesity. (Mukherjee, et al., 2025)
The study describes how APDs inhibit MC4R-expressing neurons in the PVH. This inhibition doesn't just increase caloric intake; it alters dietary preference. By inhibiting these neurons, APDs promote a shift toward carbohydrate and fat-heavy foods. The review notes that co-treatment with MC4R agonists like setmelanotide can reverse this hyperphagia, confirming the PVH's role as a critical gatekeeper in APD-induced obesity.
A significant portion of the analysis is dedicated to AMP-activated protein kinase (AMPK). APDs like clozapine are shown to over-activate hypothalamic AMPK. In the ARC, this activation promotes food intake, while in the VMH, its effects are more complex, potentially interfering with thermogenesis and "turning down the furnace" of the body's energy expenditure. The study suggests that targeting AMPK signaling specifically in the hypothalamus could be a "magic bullet" for preventing weight gain without affecting the drug's therapeutic action on dopamine receptors.
The review synthesizes recent clinical and preclinical data on GLP-1 receptor agonists (like semaglutide) and the emerging role of GLP-2. While GLP-1 agonists show great promise in inducing weight loss in psychiatric populations, the review highlights a fascinating preclinical finding: the GLP-2 analog teduglutide can mitigate olanzapine-induced obesity by activating 5HT2C receptors in the VMH. This adds a new layer to our understanding of the gut-brain-metabolism axis.
The latter half of the review discusses how we might deliver treatments directly to the hypothalamus. Gold nanoclusters (AuNCs) have shown success in rats by ameliorating dysfunctional H1R-AMPK signaling. Furthermore, the use of small extracellular vesicles (sEVs) to deliver dominant-negative AMPK constructs specifically to the ARC or VMH represents a pinpoint-accurate method of metabolic control. These "natural" nanocarriers cross the blood-brain barrier effectively and carry a lower risk of immune rejection than synthetic alternatives.
The findings underscore that antipsychotic-induced weight gain is not a simple matter of "willpower" but a complex neuroendocrine disruption centered in the hypothalamus. By identifying the specific receptors and signaling molecules (like MC4R, AMPK, and MORs) involved, this research opens the door for "co-therapy" strategies. For instance, combining APDs with MC4R agonists, GLP-1/GLP-2 analogs, or utilizing nano-delivery systems for metabolic regulators could potentially preserve the life-saving psychiatric benefits of these drugs while neutralizing their metabolic toxicity. This shifts the paradigm from treating obesity after it occurs to preventing it through precise, circuit-based neuropharmacology and bio-compatible nanotechnology.
Protheragen provides specialized R&D services tailored for neuroendocrinology and obesity research, delivering robust data support for the discovery and validation of novel Anti-obesity Therapies. Our platform leverages advanced tools such as gene editing technology and microvesicle delivery systems to enhance experimental design and enable high-precision metabolic phenotyping, empowering researchers to achieve innovative breakthroughs.
Reference
Copyright © Protheragen. All rights reserves.