Deciphering the Plasma Lipidome: Is it Obesity or Insulin Resistance with Steatosis that Matters?
December 26, 2025
October 8, 2025
Obesity and metabolic imbalance are driven not only by excess energy intake but also by chronic adipose tissue inflammation and dysregulated adipokine signaling, which worsen insulin resistance, lipid dysregulation, and systemic metabolic health.
In a late 2025 publication in Obesities, Nakamura and colleagues explore how L-fucose, a monosaccharide sourced from brown seaweed, influences adipocyte inflammation and metabolism. Their work addresses a critical gap in the field, moving beyond traditional pharmacological and lifestyle interventions to investigate innovative, food-based metabolic regulators.
This paper explores how L-fucose (a monosaccharide) affects metabolic and inflammatory responses in adipocytes — key cells in regulating systemic metabolic balance.
The study shows that L-fucose significantly suppresses pro-inflammatory mediators induced by inflammatory stimulation (e.g., LPS, TNF-α, IFN-γ) in 3T3-L1 adipocytes. This indicates reduced activation of inflammation pathways that are known contributors to metabolic dysregulation in obesity.
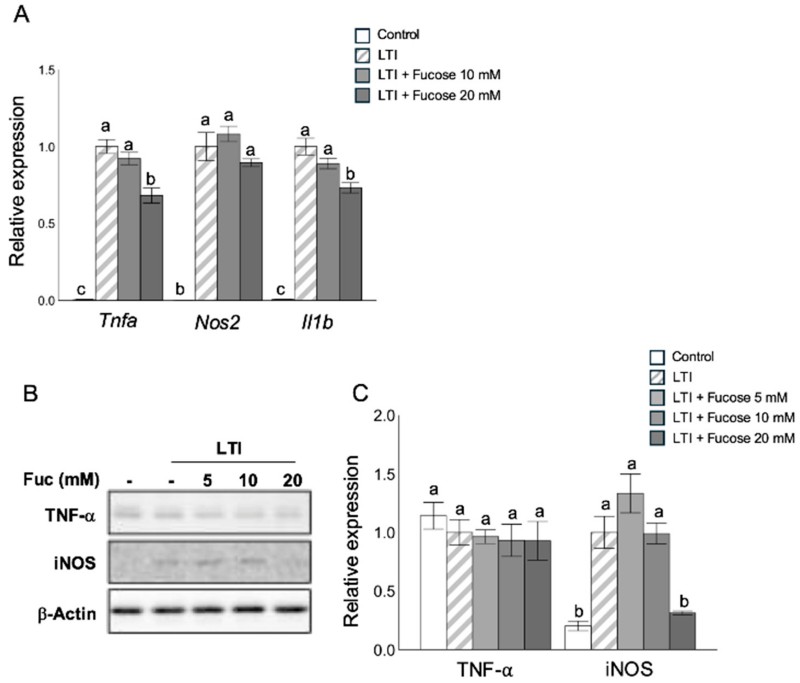 Fig.1 L-fucose reduces pro-inflammatory gene and protein expression in adipocytes treated with inflammatory stimuli. (Nakamura, et al., 2025)
Fig.1 L-fucose reduces pro-inflammatory gene and protein expression in adipocytes treated with inflammatory stimuli. (Nakamura, et al., 2025)
In the presence of inflammatory stimuli, adipocytes exhibited a substantial production of reactive oxygen species (ROS), a defining feature of oxidative stress that is intimately connected to metabolic instability and insulin insensitivity. The administration of L-fucose effectively diminished ROS accumulation, highlighting its capacity to recalibrate the intracellular redox environment independently of its effects on inflammatory transcription. This antioxidant property likely plays a pivotal role in mitigating the secondary metabolic damage triggered by persistent adipose inflammation in obese states.
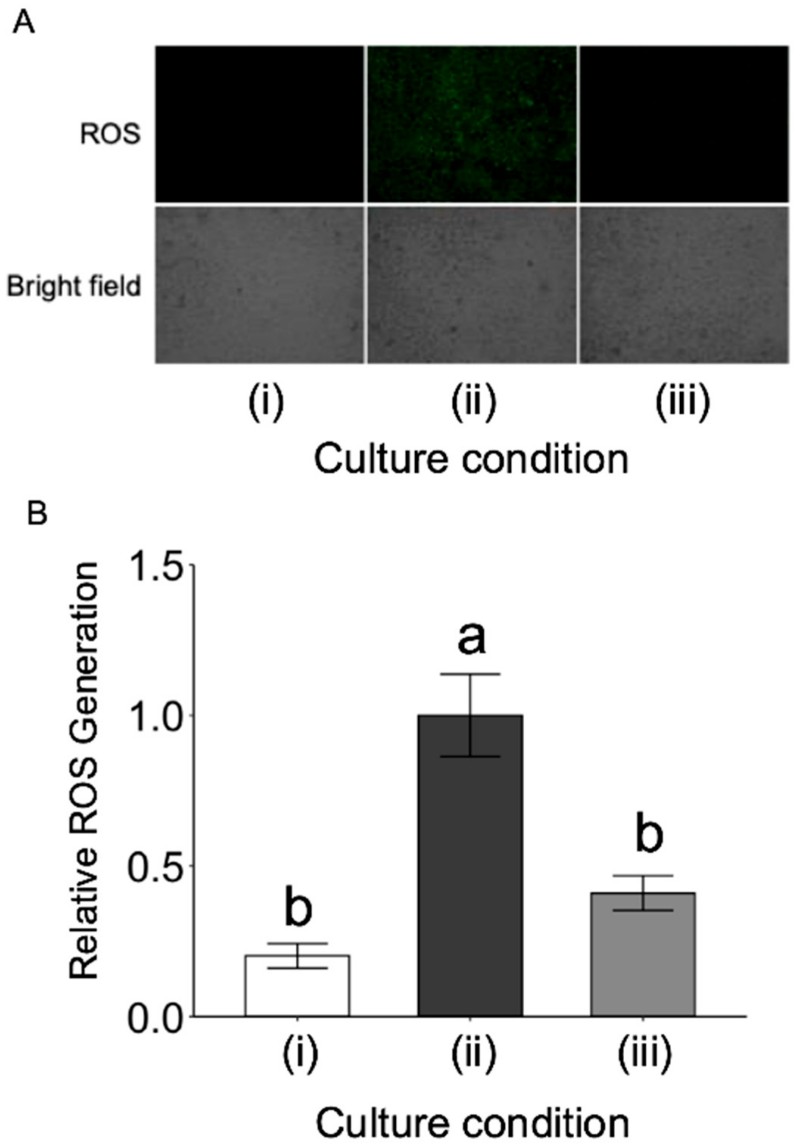 Fig.2 L-fucose lowers oxidative stress levels in adipocytes exposed to inflammatory challenge. (Nakamura, et al., 2025)
Fig.2 L-fucose lowers oxidative stress levels in adipocytes exposed to inflammatory challenge. (Nakamura, et al., 2025)
Although treatment with L-fucose caused a slight reduction in total adiponectin expression in 3T3-L1 adipocytes under inflammatory conditions, it significantly enhanced the formation and secretion of HMW adiponectin, the biologically active multimer that is most relevant for insulin-sensitizing and anti-inflammatory effects. Specifically, the HMW/total adiponectin ratio increased, indicating that L-fucose promotes the preferential assembly or stabilization of HMW adiponectin despite the modest decrease in overall adiponectin levels.
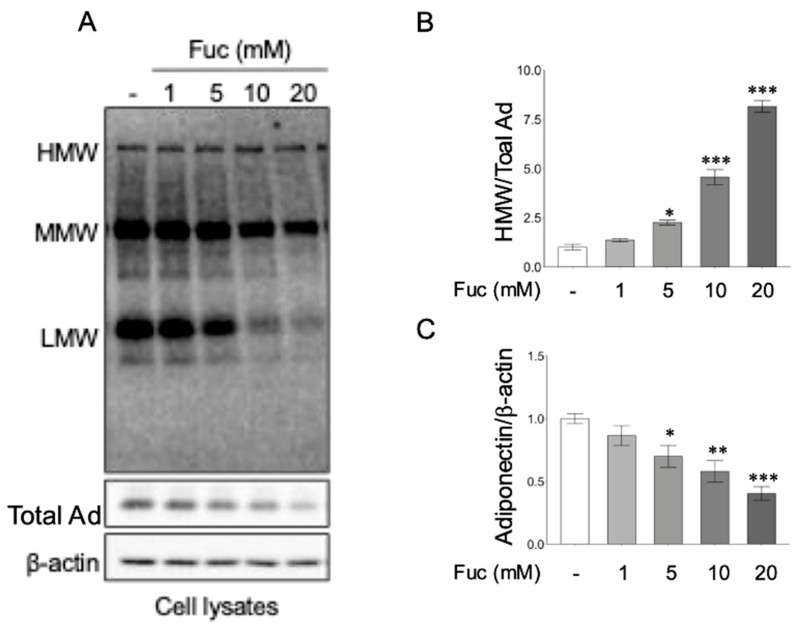 Fig.3 L-fucose increases the ratio of HMW to total adiponectin in adipocytes. (Nakamura, et al., 2025)
Fig.3 L-fucose increases the ratio of HMW to total adiponectin in adipocytes. (Nakamura, et al., 2025)
The anti-inflammatory effects of L-fucose are likely mediated through the inhibition of MAPK (mitogen-activated protein kinase) and NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) signaling pathways, which play central roles in regulating cellular stress and inflammatory responses in adipocytes. Under inflammatory stimuli, these pathways normally trigger the transcription of pro-inflammatory cytokines such as TNF-α, IL-1β, and chemokines that exacerbate adipose tissue inflammation. L-fucose reduces the phosphorylation of key components in these pathways, thereby attenuating their activation and downstream signaling. By dampening MAPK and NF-κB activity, L-fucose helps suppress chronic low-grade inflammation, a hallmark of obesity and metabolic disorders, which in turn may improve insulin signaling, lipid metabolism, and overall adipocyte function.
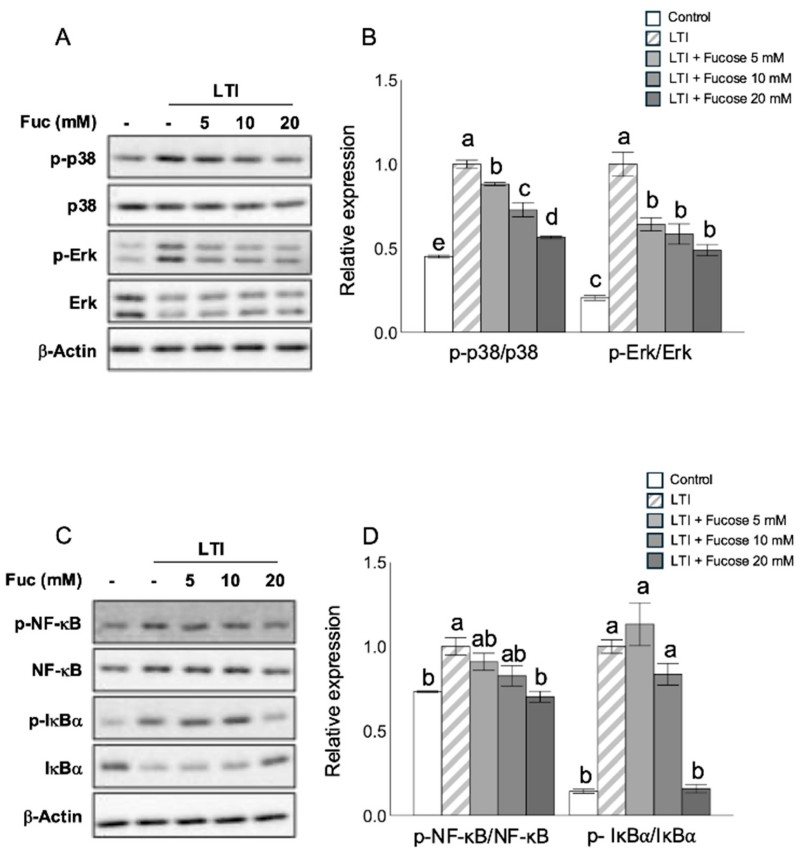 Fig.4 L-fucose decreases phosphorylation of MAPK and NF-κB signaling proteins in adipocytes. (Nakamura, et al., 2025)
Fig.4 L-fucose decreases phosphorylation of MAPK and NF-κB signaling proteins in adipocytes. (Nakamura, et al., 2025)
This research offers significant mechanistic clarity by demonstrating that L-fucose alleviates oxidative stress and adipose tissue inflammation, both of which are central indicators of metabolic imbalance in obese phenotypes. Through the enhancement of anti-inflammatory adipokine secretions like HMW adiponectin and the inhibition of inflammatory signals, L-fucose facilitates the restoration of metabolic equilibrium and glucose sensitivity. Upon successful clinical validation, these metabolic regulators could serve as adjuncts to conventional obesity treatments by addressing underlying redox instability and inflammatory pathways rather than focusing solely on mass reduction.
Fundamentally, pursuing metabolic stability via nutritional and biochemical interventions, such as the use of bioactive food ingredients like L-fucose, represents a potent strategy for comprehensive metabolic disease control, particularly in mitigating persistent inflammation and adipose tissue impairment.
At Protheragen, for investigators seeking to decode metabolic imbalances, adipocyte signaling networks, and inflammatory cascades, or the bioactivity of functional nutrients, our platform delivers End-to-end Preclinical and Translational Support. These offerings encompass comprehensive metabolic phenotyping, adipocyte-based functional assays, redox profiling, and systems-level pathway analysis to facilitate robust experimental design and biological validation.
Reference
Copyright © Protheragen. All rights reserves.