- Home
- Anti-Obesity Drug Interventions and Novel Drug Development
- Unlocking the Anti-Obesity Potential of 6,7-Dihydroxy-4-methylcoumarin: Insights into Adipogenesis Inhibition
Unlocking the Anti-Obesity Potential of 6,7-Dihydroxy-4-methylcoumarin: Insights into Adipogenesis Inhibition
November 7, 2025
Obesity has emerged as a significant global clinical challenge in modern society, characterized by the excessive accumulation of adipose tissue, which substantially increases the risk of type 2 diabetes and cardiovascular diseases. In a landmark study published in December 2024 in the journal Pharmaceuticals, researchers led by authors such as Ji-Hye Kim and others conducted a comprehensive investigation titled 6,7-Dihydroxy-4-methylcoumarin Suppresses Adipogenesis via AMPK and MAPK Signaling with In Silico Analysis of Adipogenic Proteins. While previous pharmacological treatments like orlistat or phentermine have been effective, they often carry adverse effects such as gastrointestinal discomfort or cardiovascular risks, leaving a critical gap for safer, natural product-based alternatives. This research focuses on 4-methylcoumarin (4MC), a versatile scaffold found in plants like Angelica archangelica, to address the insufficient understanding of its anti-obesity mechanisms. By utilizing mouse embryonic fibroblasts known as 3T3-L1 preadipocytes (3T3-L1), the team sought to identify specific derivatives that could effectively modulate the molecular signals governing fat cell formation.
Highlights
- Superior Pharmacokinetic Profile: Through advanced computational screening, 6,7-dihydroxy-4-methylcoumarin (6,7DH-4MC) demonstrated a superior pharmacokinetic profile. With a human intestinal absorption rate of 93.81% and strict adherence to established drug-likeness criteria, this derivative presents a more promising oral therapeutic window than the standard reference compound epigallocatechin gallate (EGCG).
- Dual-Pathway Modulation: Unlike many agents that target a single enzyme, 6,7DH-4MC simultaneously activates energy-sensing pathways while suppressing growth-related signals.
- Integrated In Silico Validation: The use of molecular docking and 100ns molecular dynamics simulations (MD simulations) provided a structural basis for how the compound interacts with multiple adipogenic regulators.
- Safety-First Design: The study confirmed that 6,7DH-4MC inhibits fat accumulation without inducing cytotoxicity, showing a moderate clearance and longer half-life (1.662 h) suitable for sustained systemic exposure.
Key Findings
The experimental results provide a detailed roadmap of how 6,7DH-4MC disrupts the transformation of preadipocytes into mature, fat-storing cells.
- Inhibition of Lipid Accumulation and Cellular Viability
Initial assessments using the MTT assay (MTT) ensured that the concentrations used (up to 100 μM) did not harm the cells, with 6,7DH-4MC actually showing a modest metabolic activation. The methylisobutylxanthine-dexamethasone-insulin (MDI) cocktail induced a fivefold increase in lipid droplets, but treatment with 6,7DH-4MC resulted in a robust dose-dependent reduction. Specifically, at 100 μM, 6,7DH-4MC inhibited lipid accumulation by a striking 66.2%, outperforming the positive control EGCG, which provided a 53.3% reduction at 10 μM.
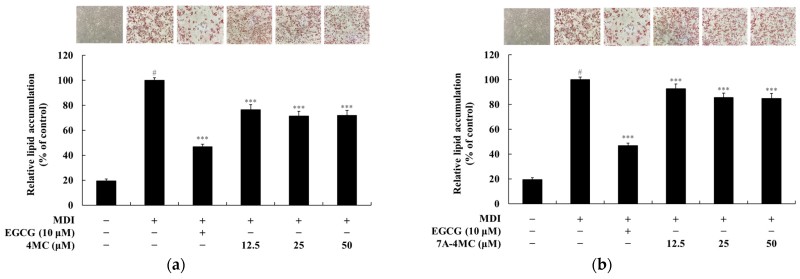 Fig.1 4MC and its derivatives inhibit lipid deposition in 3T3-L1 adipocytes to varying degrees. (Lee, et al., 2025)
Fig.1 4MC and its derivatives inhibit lipid deposition in 3T3-L1 adipocytes to varying degrees. (Lee, et al., 2025)
- Downregulation of Master Adipogenic Transcription Factors
Western blotting (WB) analysis revealed that 6,7DH-4MC directly targets the "master switches" of fat cell development. The expression of peroxisome proliferator-activated receptor gamma (PPAR-γ) was inhibited by 97.4% at the highest dose, while CCAAT/enhancer binding protein alpha (C/EBPα) and sterol regulatory element-binding protein 1c (SREBP-1c) showed reductions of 81.3% and 41.3%, respectively. Furthermore, fatty acid-binding protein 4 (FABP4), a key marker for late-stage lipid storage, was suppressed by 94.7%, suggesting that 6,7DH-4MC prevents the physical formation of lipid droplets.
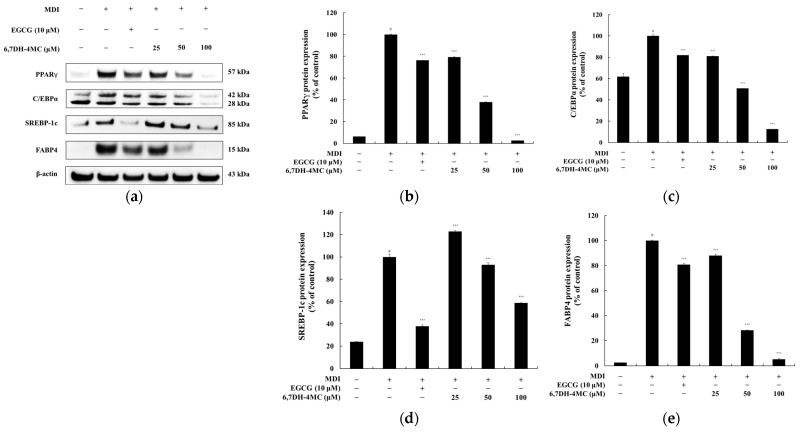 Fig.2 6,7DH-4MC significantly downregulates the expression levels of adipocyte differentiation-related marker proteins. (Lee, et al., 2025)
Fig.2 6,7DH-4MC significantly downregulates the expression levels of adipocyte differentiation-related marker proteins. (Lee, et al., 2025)
- Modulation of Upstream Signaling Pathways
The study delved deeper into the early stages of adipogenesis, focusing on the mitogen-activated protein kinase (MAPK) and AMP-activated protein kinase (AMPK) pathways. 6,7DH-4MC significantly inhibited the phosphorylation of ERK1/2 and p38 MAPK, which are essential for the initial mitotic clonal expansion of preadipocytes. Concurrently, the compound acted as a potent AMPK activator, increasing AMPK phosphorylation (P-AMPK) by 135.6% at 100 μM. This shift promotes fatty acid oxidation and inhibits the synthesis of new fats. Interestingly, while the compound reduced the phosphorylation of cyclic AMP-responsive element-binding protein (CREB) by 54.8%, it had minimal impact on the AKT pathway.
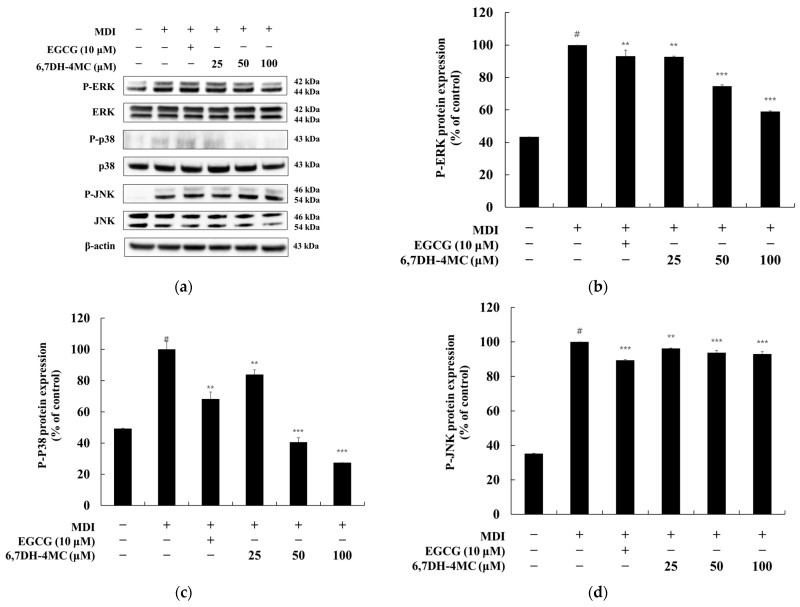 Fig.3 6,7DH-4MC primarily regulates adipogenesis signaling by inhibiting phosphorylation in the ERK and p38 pathways. (Lee, et al., 2025)
Fig.3 6,7DH-4MC primarily regulates adipogenesis signaling by inhibiting phosphorylation in the ERK and p38 pathways. (Lee, et al., 2025)
- Pharmacokinetics and Structural Stability
Absorption, distribution, metabolism, and excretion (ADME) analysis highlighted that 6,7DH-4MC is not a P-glycoprotein (P-gp) inhibitor and shows minimal central nervous system exposure, indicating high oral bioavailability and safety. Molecular docking revealed low binding energies across targets like ERK1/2 (-6.5 kcal/mol) and PPAR-γ (-6.4 kcal/mol). Further 100ns MD simulations confirmed that the protein-ligand complexes are thermodynamically stable, driven primarily by van der Waals and hydrophobic interactions with key residues like TYR36 and PHE57.
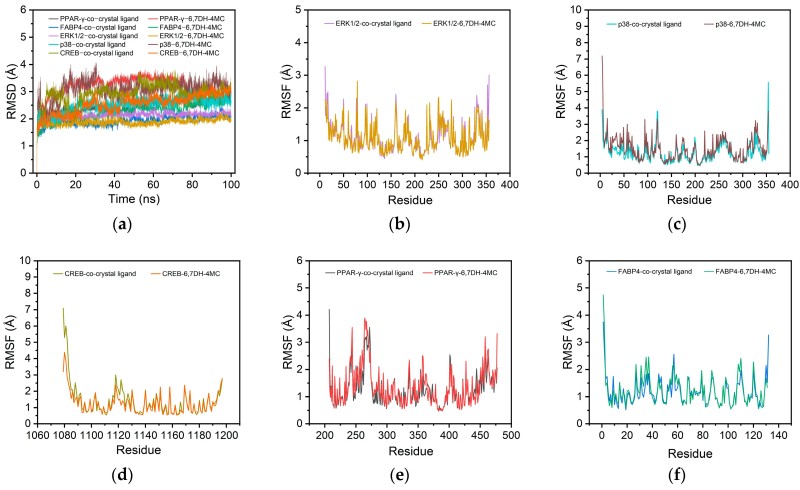 Fig.4 Molecular dynamics simulations reveal that 6,7DH-4MC exhibits excellent structural stability and binding properties with multiple target protein complexes. (Lee, et al., 2025)
Fig.4 Molecular dynamics simulations reveal that 6,7DH-4MC exhibits excellent structural stability and binding properties with multiple target protein complexes. (Lee, et al., 2025)
Interpretation & Translational Value
The findings of this study significantly advance our understanding of how coumarin derivatives can be repurposed as metabolic regulators by demonstrating that 6,7DH-4MC operates through a sophisticated "dual-action" mechanism that both prevents the birth of new fat cells and shifts existing cellular metabolism toward energy expenditure. The high intestinal absorption and adherence to Lipinski's "Rule of Five" make 6,7DH-4MC a far more "drug-like" candidate than complex polyphenols like EGCG, which often suffer from poor permeability. This pleiotropic approach is particularly valuable for drug development because targeting multiple nodes in the adipogenic signaling network reduces the likelihood of cellular compensation and increases overall efficacy in complex metabolic environments. Beyond its direct anti-obesity potential, the activation of AMPK suggests broader applications in treating related conditions such as non-alcoholic fatty liver disease (NAFLD) and insulin resistance. Future research should prioritize in vivo animal models to confirm these systemic benefits and explore the potential for oral administration in humans.
Research Support
For researchers interested in exploring the mechanisms of adipogenesis or conducting high-throughput screening of natural compounds, Protheragen provides comprehensive Preclinical Services. We offer specialized 3T3-L1 Cell Models, advanced western blotting (WB) quantification, and integrated in silico molecular docking and molecular dynamics (MD) analysis to accelerate your drug discovery and metabolic health studies.
Reference
- Lee, Y.J.; et al. 6, 7-Dihydroxy-4-methylcoumarin suppresses adipogenesis via AMPK and MAPK signaling with in silico analysis of adipogenic proteins. Pharmaceuticals. 2025, 18(12): 1780. (CC BY 4.0)